血清肠型脂肪酸结合蛋白(I-FABP)在慢加急性肝衰竭发生发展中的预测价值
DOI: 10.12449/JCH240820
Value of intestinal fatty acid binding protein in predicting the development and progression of acute-on-chronic liver failure
-
摘要:
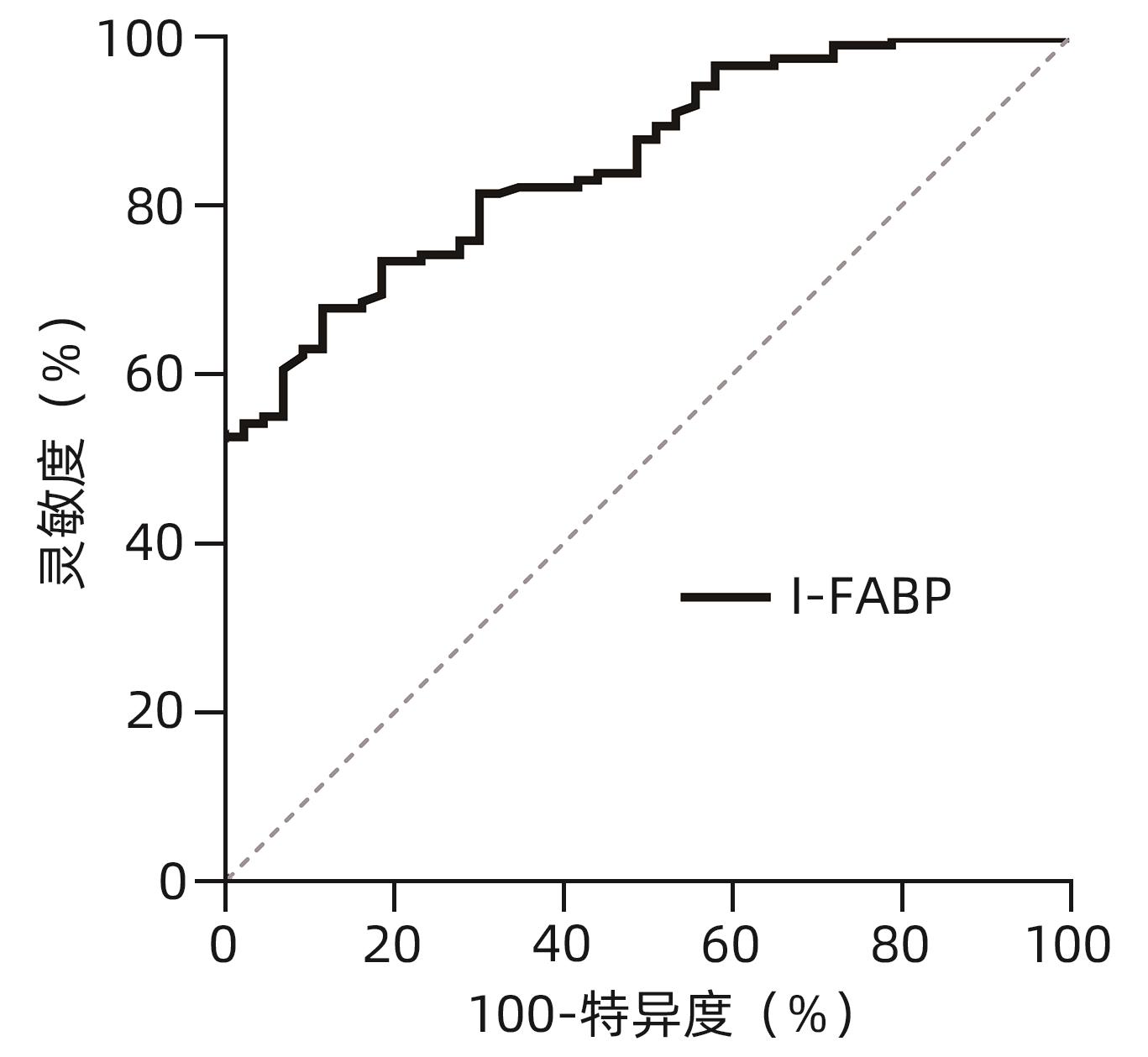
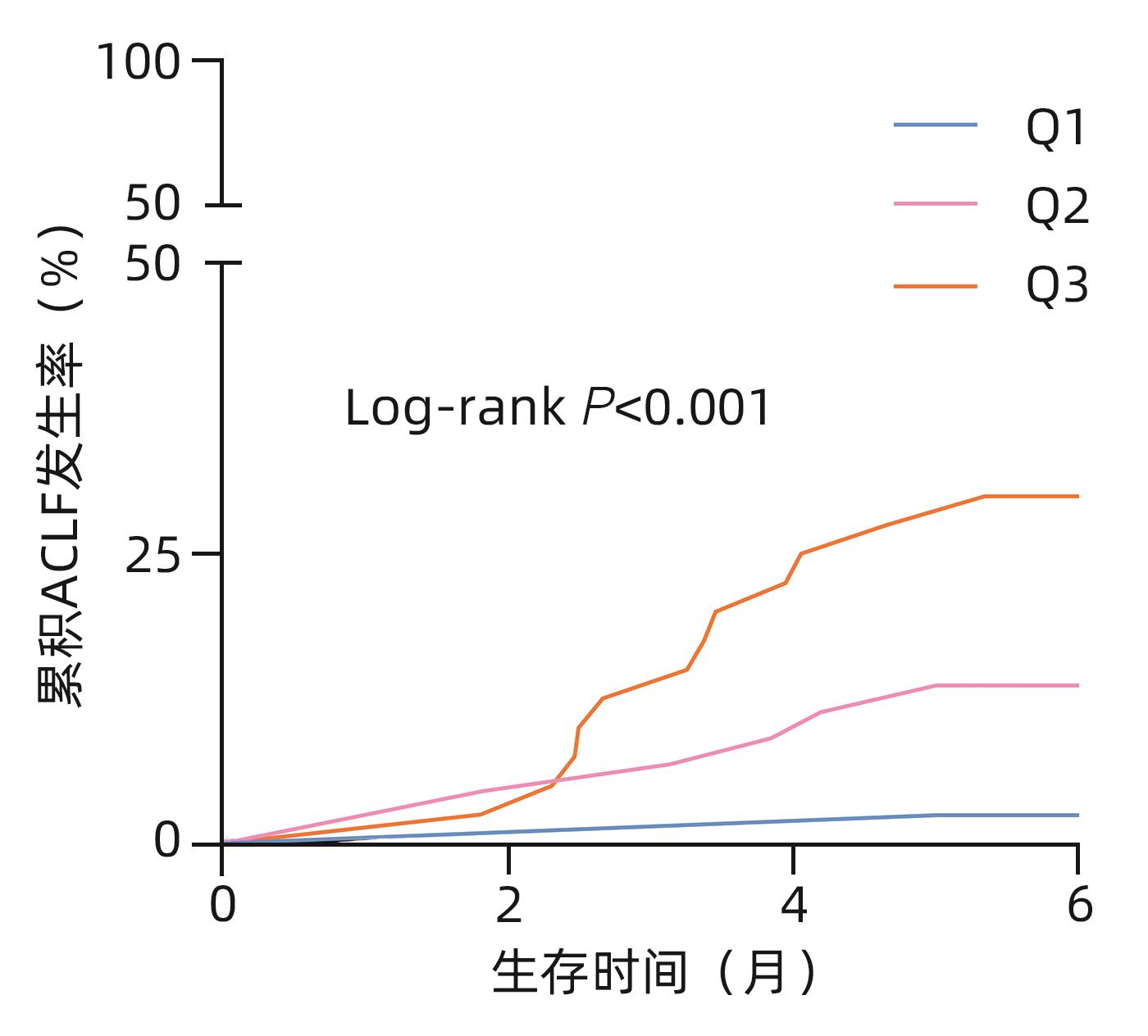
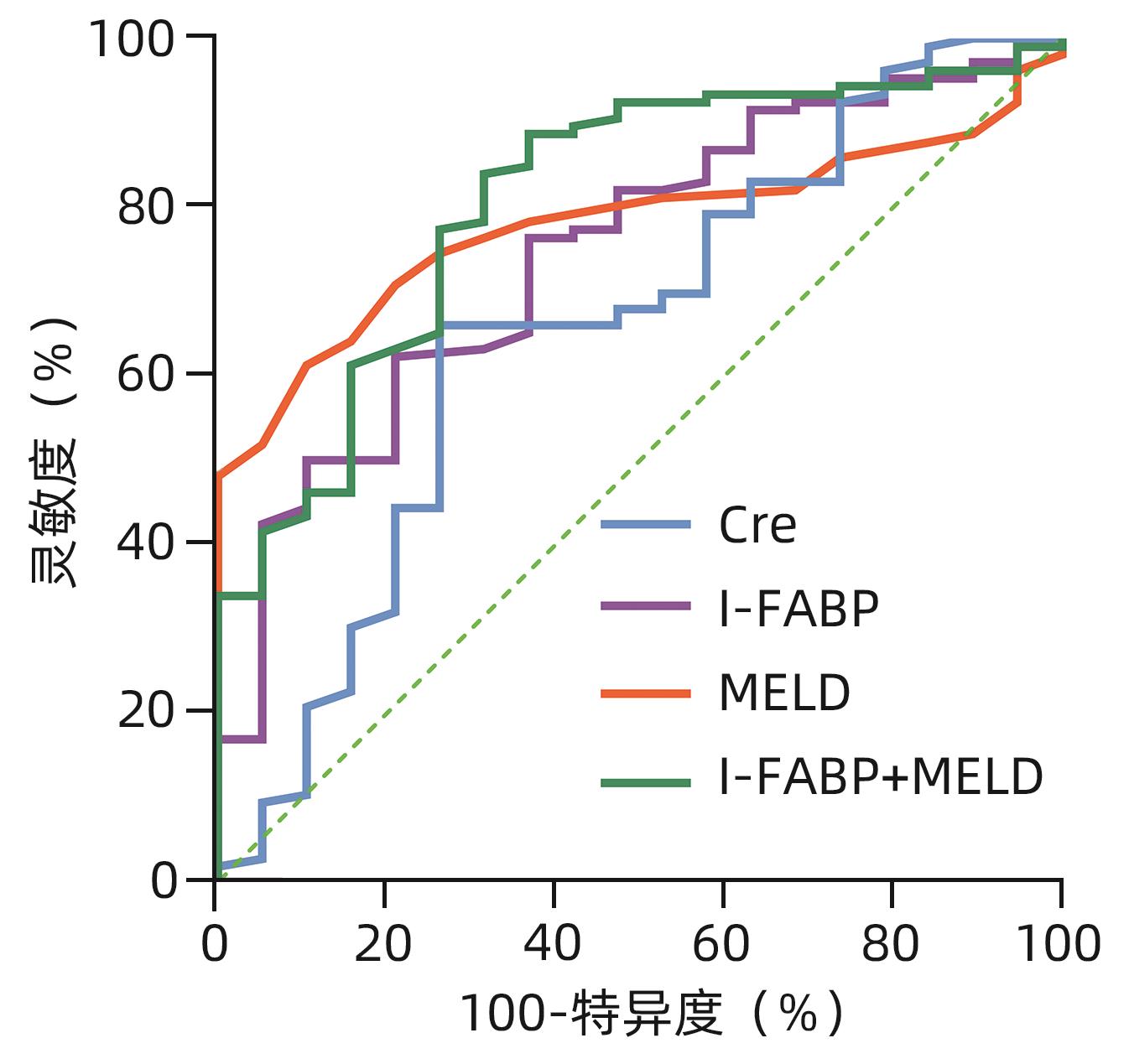
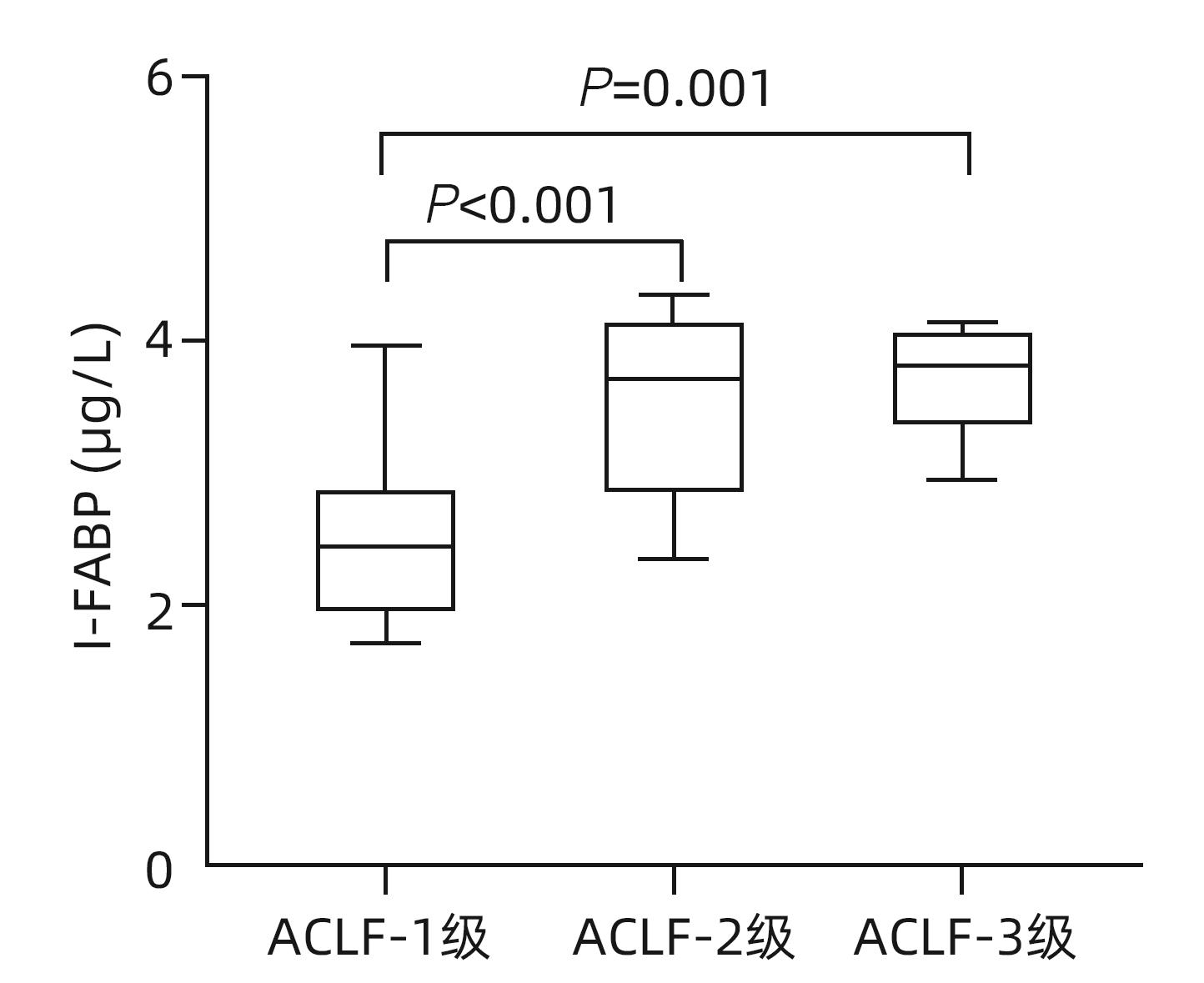
目的 探讨血清肠型脂肪酸结合蛋白(I-FABP)对慢加急性肝衰竭(ACLF)发生发展的预测作用。 方法 回顾性分析2020年9月—2023年3月延边大学附属医院收治的168例肝硬化失代偿患者的临床资料,观察入院合并ACLF的患者情况和随访6个月新发ACLF事件。采用ELISA法测定患者入院血清I-FABP水平。非正态分布计量资料两组间比较采用Mann-Whitney U检验,多组间比较采用Kruskal-Wallis H秩和检验。计数资料组间比较采用χ2检验。趋势性分析采用Jonckheere-Terpstra检验。采用Spearman相关分析两变量间相关性。多变量Cox回归法分析随访期间新发ACLF的影响因素。Kaplan-Meier曲线分析不同组间ACLF发生情况,并采用Log-rank检验评估差异。采用受试者工作特征曲线(ROC曲线)和曲线下面积评估I-FABP对ACLF发生、发展的预测性能。 结果 入组168例患者中43例合并ACLF,125例无ACLF的患者在随访期间新发ACLF19例。纳入时合并ACLF患者的I-FABP水平高于无ACLF者,差异有统计学意义(Z=4.359,P<0.001)。新发ACLF患者的I-FABP水平均高于未发生ACLF者,差异有统计学意义(Z=3.414,P<0.001)。I-FABP随ACLF分级增加而升高(H=17.385,P<0.001,P趋势<0.001)。多因素分析显示I-FABP与随访期间新发ACLF独立相关(HR=2.138,95%CI:1.297~3.525,P=0.003),且I-FABP三分位数显示出良好的区分能力(χ2=12.16,P<0.001)。ROC曲线显示I-FABP对ACLF发生和发展具有较好的预测效能,ROC曲线下面积分别为0.854(95%CI:0.791~0.903)和0.747(95%CI:0.661~0.820),最佳截断值分别为2.07 μg/L和1.86 μg/L。 结论 I-FABP是预测ACLF发生和发展的生物标志物,有助于临床识别高危患者,改善临床管理。 -
关键词:
- 慢加急性肝功能衰竭 /
- 肠脂肪酸结合蛋白质类 /
- 生物标记
Abstract:Objective To investigate the value of intestinal fatty acid binding protein (I-FABP) in predicting the development and progression of acute-on-chronic liver failure (ACLF). Methods A retrospective analysis was performed for the clinical data of 168 patients with decompensated liver cirrhosis who were admitted to The Affiliated Hospital of Yanbian University from September 2020 to March 2023. The conditions of the patients with ACLF on admission were observed, and the patients were followed up for 6 months to identify new-onset ACLF cases. ELISA was used to measure the serum level of I-FABP on admission. The Mann-Whitney U test was used for comparison of non-normally distributed continuous data between two groups, and the Kruskal-Wallis H rank sum test was used for comparison between multiple groups; the chi-square test was used for comparison of categorical data between groups; the Jonckheere-Terpstra test was used for trend analysis. The Spearman correlation analysis was used to investigate the correlation between two variables, and the multivariate Cox regression analysis was used to investigate the influencing factors for new-onset ACLF during follow-up. The Kaplan-Meier curve was used to analyze the onset of ACLF in different groups, and the log-rank test was used for the analysis of such differences. The receiver operating characteristic (ROC) curve and the area under the ROC curve (AUC) were used to investigate the performance of I-FABP in predicting the development and progression of ACLF. Results Among the 168 patients enrolled in this study, there were 43 patients with ACLF and 125 patients without ACLF, among whom 19 developed ACLF during follow-up. The patients with ACLF on admission had a significantly higher level of I-FABP than those without ACLF (Z=4.359, P<0.001). The patients with new-onset ACLF had a significantly higher level of I-FABP than those without new-onset ACLF (Z=3.414, P<0.001). The level of I-FABP increased with the increase in ACLF severity grade (H=17.385, P<0.001,Ptrend<0.001). The multivariate Cox regression analysis showed that I-FABP was independently associated with new-onset ACLF during follow-up (hazard ratio=2.138, 95% confidence interval [CI]: 1.297 — 3.525, P=0.003), and the tertile of I-FABP showed a good discriminatory ability (χ2=12.16, P<0.001). The ROC curve showed that I-FABP had a good performance in predicting the development and progression of ACLF, with an area under the ROC curve of 0.854 (95%CI: 0.791 — 0.903) and 0.747 (95%CI: 0.661 — 0.820), respectively, and an optimal cut-off value of 2.07 μg/L and 1.86 μg/L, respectively. Conclusion I-FABP can be used as a biomarker to predict the development and progression of ACLF, and it may help to identify high-risk patients and improve clinical management. -
Key words:
- Acute-On-Chronic Liver Failure /
- Fatty Acid-Binding Proteins /
- Biomarkers
-
表 1 研究人群基线特征
Table 1. Baseline characteristics of study population
指标 非ACLF组(n=125) ACLF组(n=43) 统计值 P值 年龄(岁) 54(42~64) 55(48~67) Z=1.795 0.073 男性[例(%)] 83(66.4) 32(74.4) χ2=0.953 0.327 WBC(109/L) 6.39(5.20~9.35) 8.13(6.61~10.12) Z=3.140 0.002 CRP(mg/L) 13.9(8.3~40.6) 29.9(17.9~51.3) Z=3.800 <0.001 Alb(g/L) 33(29~37) 28(27~31) Z=5.054 <0.001 Cre(μmol/L) 85(75~106) 123(107~144) Z=6.066 <0.001 TBil(μmol/L) 39.8(27.2~67.8) 171.5(116.7~233.1) Z=8.915 <0.001 INR 1.30(1.04~1.74) 2.01(1.73~2.24) Z=6.749 <0.001 MELD评分 15(11~21) 29(25~33) Z=7.776 <0.001 I-FABP(μg/L) 1.51(0.87~2.55) 3.09(2.51~3.95) Z=4.359 <0.001 表 2 随访期间ACLF发展情况的临床资料比较
Table 2. Comparison of clinical data on the development of ACLF during the follow-up
指标 未发生ACLF组(n=106) 新发ACLF组(n=19) 统计值 P值 年龄(岁) 54(40~64) 53(47~62) Z=0.678 0.498 男性[例(%)] 70(66.0) 13(68.4) χ2=0.041 0.839 WBC(109/L) 6.35(5.20~9.35) 6.69(5.45~10.22) Z=0.589 0.556 CRP(mg/L) 13.2(7.7~40.7) 20.5(12.6~33.8) Z=1.241 0.214 Alb(g/L) 34(30~37) 31(25~35) Z=1.069 0.285 Cre(μmol/L) 84(75~105) 96(86~130) Z=2.156 0.031 TBil(μmol/L) 37.4(26.0~67.8) 48.0(40.5~61.1) Z=2.029 0.042 INR 1.26(1.02~1.61) 1.68(1.25~2.03) Z=2.219 0.027 MELD评分 14(11~19) 21(18~23) Z=3.867 <0.001 I-FABP(μg/L) 1.34(0.85~2.13) 2.69(1.91~3.14) Z=3.414 <0.001 表 3 多因素Cox回归分析随访期间发生ACLF的影响因素
Table 3. Multivariate Cox regression analyzed the factors of developing ACLF during follow-up
因素 β值 Wald HR(95%CI) P值 Cre 0.017 10.278 1.017(1.007~1.027) 0.001 I-FABP 0.760 8.880 2.138(1.297~3.525) 0.003 MELD评分 0.096 5.495 1.101(1.016~1.194) 0.019 表 4 各指标对随访期间新发ACLF的预测性能
Table 4. Predictive performance of the study parameters on new-onset ACLF during follow-up
指标 AUC(95%CI) Cut-off值 灵敏度(%) 特异度(%) Z值 P值 Cre 0.656(0.565~0.738) 92.00 73.68 66.04 2.100 0.036 I-FABP 0.747(0.661~0.820) 1.86 78.95 62.26 4.275 <0.001 MELD评分 0.779(0.696~0.848) 16.00 89.47 61.32 6.617 <0.001 I-FABP+MELD评分 0.810(0.731~0.875) 73.68 80.19 5.955 <0.001 -
[1] MOREAU R, JALAN R, GINES P, et al. Acute-on-chronic liver failure is a distinct syndrome that develops in patients with acute decompensation of cirrhosis[J]. Gastroenterology, 2013, 144( 7): 1426- 1437. e1- 1437. e9. DOI: 10.1053/j.gastro.2013.02.042. [2] KAMATH PS, KIM WR, GROUP ALDS. The model for end-stage liver disease(MELD)[J]. Hepatology, 2007, 45( 3): 797- 805. DOI: 10.1002/hep.21563. [3] JALAN R, SALIBA F, PAVESI M, et al. Development and validation of a prognostic score to predict mortality in patients with acute-on-chronic liver failure[J]. J Hepatol, 2014, 61( 5): 1038- 1047. DOI: 10.1016/j.jhep.2014.06.012. [4] CHEN MJ, LI X, TANG SH. Research progress on multidimensional evaluation of liver function in the prognosis of liver failure patients[J]. Clin J Med Offic, 2023, 51( 9): 901- 903, 907. DOI: 10.16680/j.1671-3826.2023.09.05.陈美娟, 李雪, 汤善宏. 多维度评估肝功能在肝衰竭患者预后中研究进展[J]. 临床军医杂志, 2023, 51( 9): 901- 903, 907. DOI: 10.16680/j.1671-3826.2023.09.05. [5] WANG P, ZHANG YJ, LI YR, et al. A correlation between gastrointestinal dysfunction and cirrhosis severity[J]. Medicine(Baltimore), 2018, 97( 37): e12070. DOI: 10.1097/MD.0000000000012070. [6] KALAITZAKIS E. Gastrointestinal dysfunction in liver cirrhosis[J]. World J Gastroenterol, 2014, 20( 40): 14686- 14695. DOI: 10.3748/wjg.v20.i40.14686. [7] BLASER AR, PREISER JC, FRUHWALD S, et al. Gastrointestinal dysfunction in the critically ill: A systematic scoping review and research agenda proposed by the Section of Metabolism, Endocrinology and Nutrition of the European Society of Intensive Care Medicine[J]. Crit Care, 2020, 24( 1): 224. DOI: 10.1186/s13054-020-02889-4. [8] LOGAN M, MACKINDER M, CLARK CM, et al. Intestinal fatty acid binding protein is a disease biomarker in paediatric coeliac disease and Crohn’s disease[J]. BMC Gastroenterol, 2022, 22( 1): 260. DOI: 10.1186/s12876-022-02334-6. [9] BLASER A, PADAR M, TANG J, et al. Citrulline and intestinal fatty acid-binding protein as biomarkers for gastrointestinal dysfunction in the critically ill[J]. Anaesthesiol Intensive Ther, 2019, 51( 3): 230- 239. DOI: 10.5114/ait.2019.86049. [10] TYSZKO M, LIPIŃ SKA-GEDIGA M, LEMAŃ SKA-PEREK A, et al. Intestinal fatty acid binding protein(I-FABP) as a prognostic marker in critically ill COVID-19 patients[J]. Pathogens, 2022, 11( 12): 1526. DOI: 10.3390/pathogens11121526. [11] SEILITZ J, EDSTRÖM M, KASIM A, et al. Intestinal fatty acid-binding protein and acute gastrointestinal injury grade in postoperative cardiac surgery patients[J]. J Card Surg, 2021, 36( 6): 1850- 1857. DOI: 10.1111/jocs.15430. [12] HAN CJ, WU ZX, HUANG Y, et al. Correlation between intestinal fatty acid-binding protein and bacterial infection as well as poor prognosis in patients with liver cirrhosis[J]. Chin J Infect Contr, 2023, 22( 3): 315- 321. DOI: 10.12138/j.issn.1671-9638.20233630.韩才均, 吴政燮, 黄媛, 等. 肠脂肪酸结合蛋白与肝硬化患者细菌感染和不良预后的相关性研究[J]. 中国感染控制杂志, 2023, 22( 3): 315- 321. DOI: 10.12138/j.issn.1671-9638.20233630. [13] Chinese Society of Hepatology, Chinese Medical Association. Chinese guidelines on the management of liver cirrhosis[J]. J Clin Hepatol, 2019, 35( 11): 2408- 2425. DOI: 10.3969/j.issn.1001-5256.2019.11.006.中华医学会肝病学分会. 肝硬化诊治指南[J]. 临床肝胆病杂志, 2019, 35( 11): 2408- 2425. DOI: 10.3969/j.issn.1001-5256.2019.11.006. [14] FAN Q, LI Z. Liver transplantation for acute-on-chronic liver failure[J]. Ogran Transplant, 2022, 13( 3): 333- 337. DOI: 10.3969/j.issn.1674-7445.2022.03.008.范祺, 李照. 慢加急性肝衰竭的肝移植治疗[J]. 器官移植, 2022, 13( 3): 333- 337. DOI: 10.3969/j.issn.1674-7445.2022.03.008. [15] Liver Failure and ArtificiaI Liver Group, Chinese Society of Infectious Diseases, Chinese Medical Association; Severe Liver Disease and ArtificiaI Liver Group, Chinese Society of Hepatology, Chinese Medical Association. Guideline for diagnosis and treatment of Iiver failure(2018)[J]. J Clin Hepatol, 2019, 35( 1): 38- 44. DOI: 10.3969/j.issn.1001-5256.2019.01.007.中华医学会感染病学分会肝衰竭与人工肝学组, 中华医学会肝病学分会重型肝病与人工肝学组. 肝衰竭诊治指南(2018年版)[J]. 临床肝胆病杂志, 2019, 35( 1): 38- 44. DOI: 10.3969/j.issn.1001-5256.2019.01.007. [16] HAN CJ, JIN X, WU ZX, et al. Predicting the prognosis of patients with HBV-related acute liver failure by combining the ratio of neutrophils to lymphocytes and the ratio of C-reactive protein to albumin[J]. Chin J Clin Lab Sci, 2022, 40( 4): 281- 285. DOI: 10.13602/j.cnki.jcls.2022.04.10.韩才均, 金星, 吴政燮, 等. 联合中性粒细胞与淋巴细胞比值和C反应蛋白与清蛋白比值预测HBV相关慢加急性肝衰竭患者预后[J]. 临床检验杂志, 2022, 40( 4): 281- 285. DOI: 10.13602/j.cnki.jcls.2022.04.10. [17] FUKUI H. Leaky gut and gut-liver axis in liver cirrhosis: Clinical studies update[J]. Gut Liver, 2021, 15( 5): 666- 676. DOI: 10.5009/gnl20032. [18] CHOPYK DM, GRAKOUI A. Contribution of the intestinal microbiome and gut barrier to hepatic disorders[J]. Gastroenterology, 2020, 159( 3): 849- 863. DOI: 10.1053/j.gastro.2020.04.077. [19] OKADA K, SEKINO M, FUNAOKA H, et al. Intestinal fatty acid-binding protein levels in patients with chronic renal failure[J]. J Surg Res, 2018, 230: 94- 100. DOI: 10.1016/j.jss.2018.04.057. [20] TSAI IT, WU CC, HUNG WC, et al. FABP1 and FABP2 as markers of diabetic nephropathy[J]. Int J Med Sci, 2020, 17( 15): 2338- 2345. DOI: 10.7150/ijms.49078. -



 PDF下载 ( 902 KB)
PDF下载 ( 902 KB)

 下载:
下载: