BCHE基因复合杂合突变引起胆碱酯酶极度降低1例报告
DOI: 10.12449/JCH241023
伦理学声明:本例报告已获得患者知情同意。
利益冲突声明:本文不存在任何利益冲突。
作者贡献声明:吕飒负责课题设计,资料分析,撰写论文;朱冰、徐天娇参与收集数据,修改论文;游绍莉负责拟定写作思路,指导撰写文章并最后定稿。
The extreme reduction in cholinesterase caused by compound heterozygous mutations in the BCHE gene: A case report
-
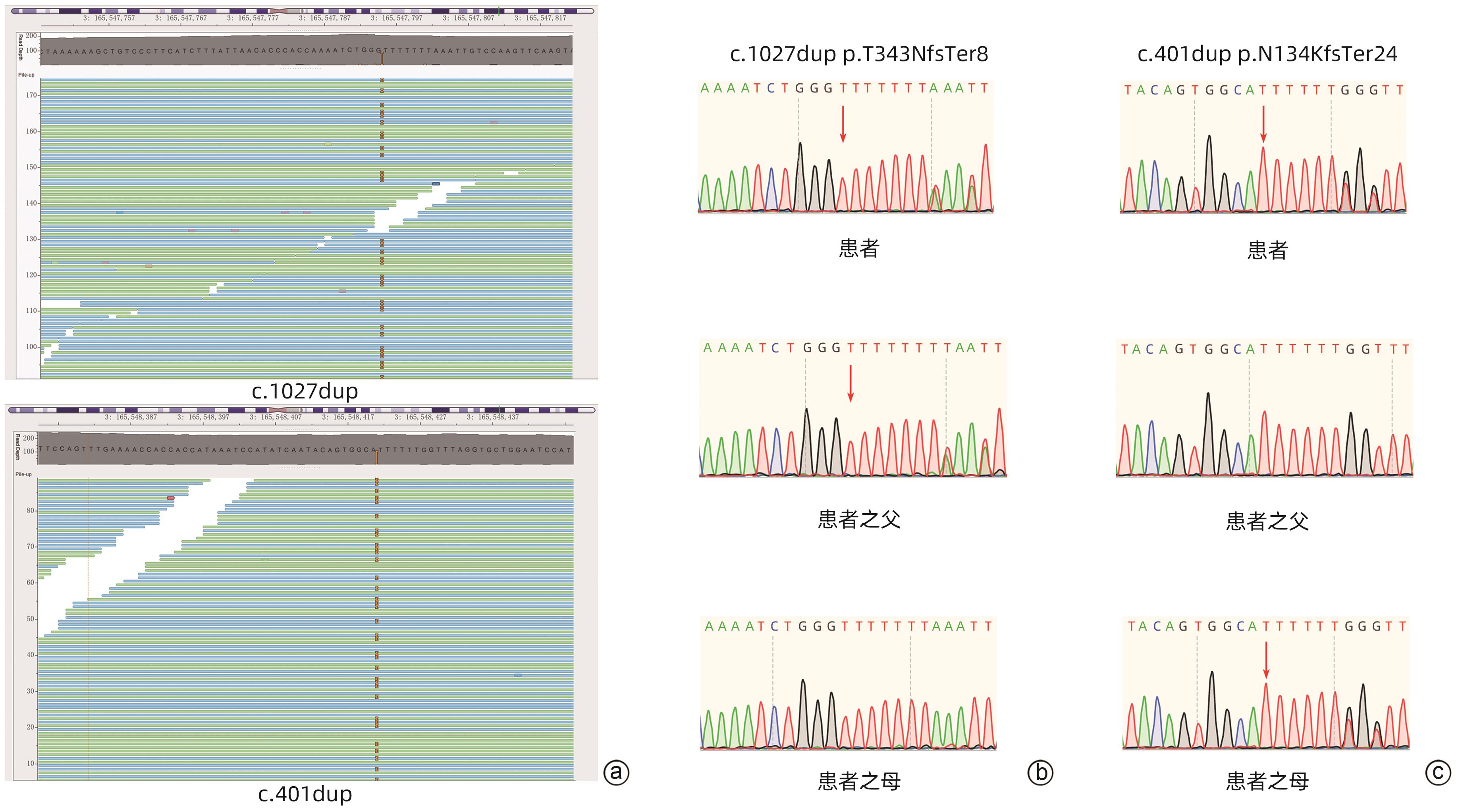
摘要: 血清胆碱酯酶(ChE)水平对于肝脏疾病、中毒性疾病等多种疾病的诊断、预后判断有重要意义,丁酰胆碱酯酶是其中重要的组成成分。BCHE基因突变可引起丁酰胆碱酯酶水平明显下降,在欧美人群报道较多,但在东方尤其中国报道较少。本研究描述了1例35岁男性因ChE水平极度降低被误诊为有机磷农药中毒并给予解毒治疗,但经过多项生化检查排除,最终通过基因全外显子测序及Sanger测序,确认为BCHE基因2号外显子存在c.1027dup和c.401dup两处复合杂合突变,所致的遗传性丁酰胆碱酯酶缺乏症是其外周血ChE水平极度降低的原因。
-
关键词:
- 丁酰胆碱酯酶 /
- BCHE基因 /
- 遗传性疾病, 先天性 /
- 突变
Abstract: Serum cholinesterase (ChE) level is important for the diagnosis and prognostic evaluation of various diseases such as liver diseases and toxic diseases, and butyrylcholinesterase (BuChE) is an important component of ChE. Mutations in the BCHE gene can cause a significant reduction in the level of BuChE, with extensive reports in European and American populations and relatively few reports in Eastern countries, particularly China. This study describes a male patient, aged 35 years, who was misdiagnosed with organophosphorus pesticide poisoning due to an extreme reduction in ChE level and was given detoxification therapy, but such diagnosis was excluded by various biochemical examinations. Finally whole-exome sequencing and Sanger sequencing revealed the complex heterozygous mutations of c.1027dup and c.401dup at exon 2 of the BCHE gene, and hereditary BuChE deficiency due to these mutations is the cause of the extreme reduction in ChE level.-
Key words:
- Butyrylcholinesterase /
- BCHE gene /
- Genetic Diseases, Inborn /
- Mutation
-
[1] LOCKRIDGE O. Review of human butyrylcholinesterase structure, function, genetic variants, history of use in the clinic, and potential therapeutic uses[J]. Pharmacol Ther, 2015, 148: 34- 46. DOI: 10.1016/j.pharmthera.2014.11.011. [2] ZHU GD, DAWSON E, HUSKEY A, et al. Genetic testing for BCHE variants identifies patients at risk of prolonged neuromuscular blockade in response to succinylcholine[J]. Pharmgenomics Pers Med, 2020, 13: 405- 414. DOI: 10.2147/PGPM.S263741. [3] IIDA S, KINOSHITA M, FUJII H, et al. Mutations of human butyrylcholinesterase gene in a family with hypocholinesterasemia[J]. Hum Mutat, 1995, 6( 4): 349- 351. DOI: 10.1002/humu.1380060411. [4] YEN T, NIGHTINGALE BN, BURNS JC, et al. Butyrylcholinesterase(BCHE) genotyping for post-succinylcholine apnea in an Australian population[J]. Clin Chem, 2003, 49( 8): 1297- 1308. DOI: 10.1373/49.8.1297. [5] YU RT, GUO YZ, DAN YJ, et al. A novel mutation in the BCHE gene and phenotype identified in a child with low butyrylcholinesterase activity: A case report[J]. BMC Med Genet, 2018, 19( 1): 58. DOI: 10.1186/s12881-018-0561-5. [6] MANOHARAN I, BOOPATHY R, DARVESH S, et al. A medical health report on individuals with silent butyrylcholinesterase in the Vysya community of India[J]. Clin Chim Acta, 2007, 378( 1-2): 128- 135. DOI: 10.1016/j.cca.2006.11.005. [7] LOCKRIDGE O, NORGREN RB Jr, JOHNSON RC, et al. Naturally occurring genetic variants of human acetylcholinesterase and butyrylcholinesterase and their potential impact on the risk of toxicity from cholinesterase inhibitors[J]. Chem Res Toxicol, 2016, 29( 9): 1381- 1392. DOI: 10.1021/acs.chemrestox.6b00228. [8] NICOLET Y, LOCKRIDGE O, MASSON P, et al. Crystal structure of human butyrylcholinesterase and of its complexes with substrate and products[J]. J Biol Chem, 2003, 278( 42): 41141- 41147. DOI: 10.1074/jbc.M210241200. [9] AMMUNDSEN HB, SØRENSEN MK, GÄTKE MR. Succinylcholine resistance[J]. Br J Anaesth, 2015, 115( 6): 818- 821. DOI: 10.1093/bja/aev228. [10] WICHMANN S, FÆRK G, BUNDGAARD JR, et al. Patients with prolonged effect of succinylcholine or mivacurium had novel mutations in the butyrylcholinesterase gene[J]. Pharmacogenet Genomics, 2016, 26( 7): 351- 356. DOI: 10.1097/FPC.0000000000000221. [11] ANDERSSON ML, MØLLER AM, WILDGAARD K. Butyrylcholinesterase deficiency and its clinical importance in anaesthesia: A systematic review[J]. Anaesthesia, 2019, 74( 4): 518- 528. DOI: 10.1111/anae.14545. [12] BRIMIJOIN S, TYE S. Favorable impact on stress-related behaviors by modulating plasma butyrylcholinesterase[J]. Cell Mol Neurobiol, 2018, 38( 1): 7- 12. DOI: 10.1007/s10571-017-0523-z. [13] LI Q, YANG HY, CHEN Y, et al. Recent progress in the identification of selective butyrylcholinesterase inhibitors for Alzheimer’s disease[J]. Eur J Med Chem, 2017, 132: 294- 309. DOI: 10.1016/j.ejmech.2017.03.062. [14] SANTARPIA L, GRANDONE I, CONTALDO F, et al. Butyrylcholinesterase as a prognostic marker: A review of the literature[J]. J Cachexia Sarcopenia Muscle, 2013, 4( 1): 31- 39. DOI: 10.1007/s13539-012-0083-5. [15] SATO T, YAMAUCHI H, SUZUKI S, et al. Serum cholinesterase is an important prognostic factor in chronic heart failure[J]. Heart Vessels, 2015, 30( 2): 204- 210. DOI: 10.1007/s00380-014-0469-8. [16] GOLIASCH G, KLEBER ME, RICHTER B, et al. Routinely available biomarkers improve prediction of long-term mortality in stable coronary artery disease: The Vienna and Ludwigshafen Coronary Artery Disease(VILCAD) risk score[J]. Eur Heart J, 2012, 33( 18): 2282- 2289. DOI: 10.1093/eurheartj/ehs164. [17] YUAN M, HAN B, XIA YP, et al. Augmentation of peripheral lymphocyte-derived cholinergic activity in patients with acute ischemic stroke[J]. BMC Neurol, 2019, 19( 1): 236. DOI: 10.1186/s12883-019-1481-5. [18] GREMMEL T, WADOWSKI PP, MUELLER M, et al. Serum cholinesterase levels are associated with 2-year ischemic outcomes after angioplasty and stenting for peripheral artery disease[J]. J Endovasc Ther, 2016, 23( 5): 738- 743. DOI: 10.1177/1526602816655521. [19] FENG WM, TANG CW, GUO HH, et al. Prognostic value of serum cholinesterase activities in sepsis patients[J]. Hepatogastroenterology, 2013, 60( 125): 1001- 1005. DOI: 10.5754/hge13141. [20] ZIVKOVIC AR, TOURELLE KM, BRENNER T, et al. Reduced serum cholinesterase activity indicates splenic modulation of the sterile inflammation[J]. J Surg Res, 2017, 220: 275- 283. DOI: 10.1016/j.jss.2017.07.024. [21] KLOCKER EV, BARTH DA, RIEDL JM, et al. Decreased activity of circulating butyrylcholinesterase in blood is an independent prognostic marker in pancreatic cancer patients[J]. Cancers(Basel), 2020, 12( 5): 1154. DOI: 10.3390/cancers12051154. [22] YAMAMOTO M, SAITO H, UEJIMA C, et al. Combination of serum albumin and cholinesterase levels as prognostic indicator in patients ith colorectal cancer[J]. Anticancer Res, 2019, 39( 2): 1085- 1090. DOI: 10.21873/anticanres.13217. [23] SOUZA RL, MIKAMI LR, MAEGAWA RO, et al. Four new mutations in the BCHE gene of human butyrylcholinesterase in a Brazilian blood donor sample[J]. Mol Genet Metab, 2005, 84( 4): 349- 353. DOI: 10.1016/j.ymgme.2004.12.005. [24] ON-KEI CHAN A, LAM CW, TONG SF, et al. Novel mutations in the BCHE gene in patients with no butyrylcholinesterase activity[J]. Clin Chim Acta, 2005, 351( 1-2): 155- 159. DOI: 10.1016/j.cccn.2004.09.004. [25] LV HY, YANG LH, BU LN, et al. A case report of primary neonatal hypocholinesterase caused by homozygous frameshift mutation of the utyrylcholinesterase(BCHE) gene and review of literature[J]. Clin Lab, 2019, 65( 7). DOI: 10.7754/Clin.Lab.2019.181254. -



 PDF下载 ( 1397 KB)
PDF下载 ( 1397 KB)

 下载:
下载:


