丁型肝炎病毒治疗药物的研究进展
DOI: 10.12449/JCH260205
Research advances in antiviral drugs for the treatment of hepatitis D virus infection
-
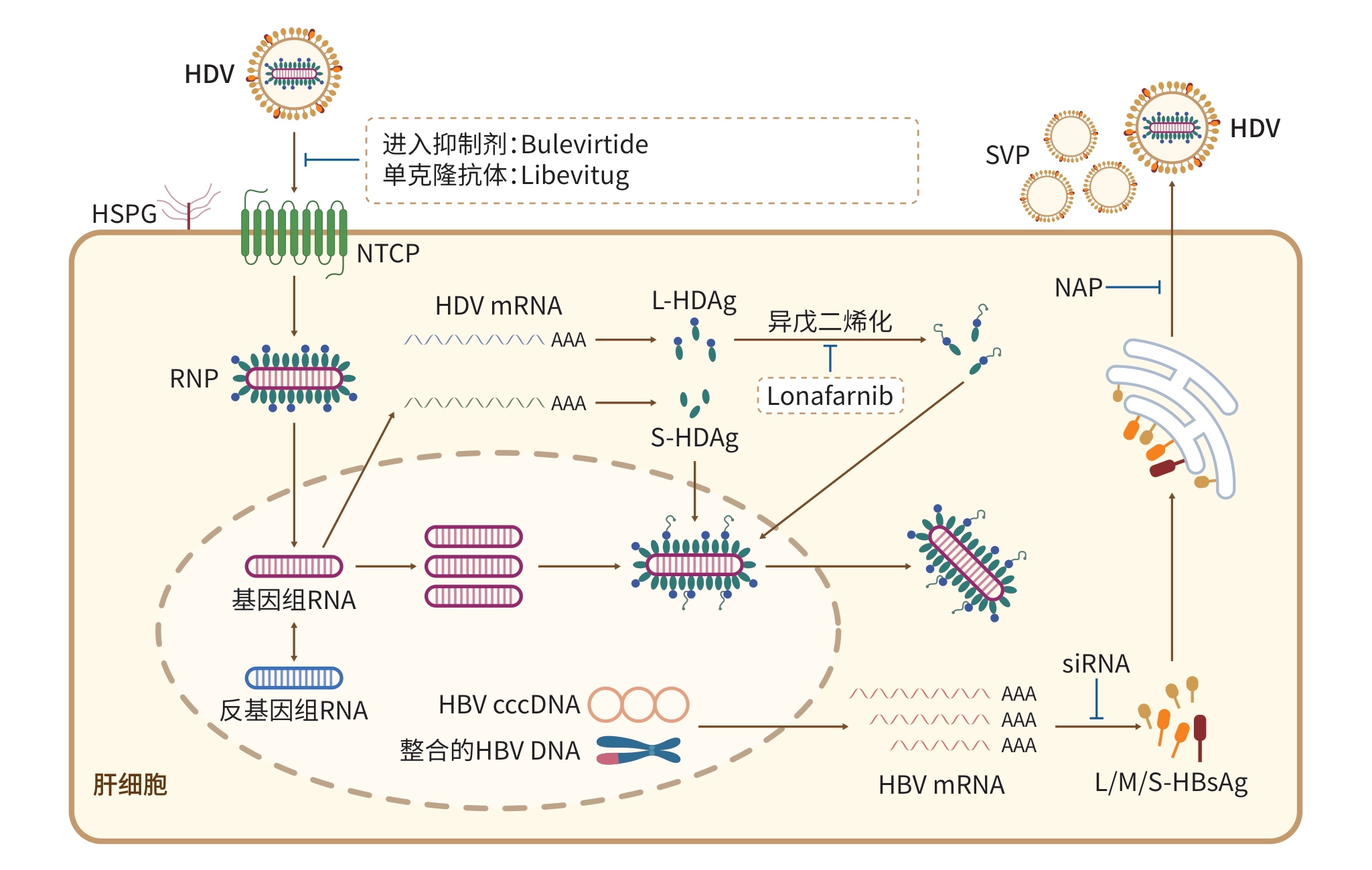
摘要: 在病毒性肝炎中,丁型肝炎病毒(HDV)合并乙型肝炎病毒(HBV)感染是最为严重的类型,患者肝脏疾病进展快速,发生肝硬化与肝细胞癌的风险显著增加。治疗HBV的核苷(酸)类似物对HDV感染无效,亟需开发特异有效的抗HDV病毒治疗手段。近年来,针对HDV的特异性抗病毒药物研发取得重要进展,其中以靶向病毒入侵阶段的病毒进入抑制剂(布来韦肽)和单克隆抗体药物(立贝韦塔单抗)为代表的新药,为HDV感染治疗带来突破性进展。本综述简述HDV治疗药物的最新研究进展,重点介绍新近获批的HDV治疗新药的作用机制和临床研究数据,并探讨HDV治疗领域内仍待解决的问题,以期为了解丁型肝炎的治疗现状提供参考。Abstract: Co-infection of hepatitis D virus (HDV) and hepatitis B virus (HBV) is the most severe form of viral hepatitis and is associated with accelerated progression of liver disease and a significant increase in the risk of liver cirrhosis and hepatocellular carcinoma. Nucleo(s)tide analogues for HBV treatment are ineffective against HDV infection, necessitating the urgent need for developing specific and effective antiviral therapies for HDV. In recent years, significant advances have been made in the research and development of specific antiviral drugs against HDV, including entry inhibitors targeting viral entry (Bulevirtide) and monoclonal antibody drugs (Libevitug), which bring ground-breaking advances in the treatment of HDV infection. This article briefly reviews the latest research advances in therapeutic drugs for HDV, introduces the mechanism of action and clinical research data of new drugs recently approved for the treatment of HDV, and discusses the challenges that need to be solved in the field of HDV treatment, in order to provide a reference for understanding the current status of hepatitis D treatment.
-
Key words:
- Hepatitis Delta Virus /
- Antiviral Agents /
- Bulevirtide /
- Libevitug
-
表 1 HDV治疗药物研究进展汇总
Table 1. Summary of research advances in HDV therapeutics
药物名称 药物类型/靶点 作用机制 开发阶段 Bulevirtide 多肽/NTCP 竞争性阻断病毒和受体结合,抑制病毒入侵 欧洲等地区上市 Libevitug 单克隆抗体/PreS1 通过特异性结合HBV/HDV表面的PreS1区域,阻断病毒和其受
体NTCP的结合,从而阻止病毒感染或再感染肝细胞中国附条件上市/临床
Ⅲ期Lonafarnib 小分子药物/法尼基转移酶 抑制L-HDAg的异戊二烯化修饰,阻断病毒组装 临床Ⅲ期/停止开发 Tobevibart 单克隆抗体/HBsAg 中和病毒感染,阻断病毒进入肝细胞 临床Ⅲ期 Brelovitug 单克隆抗体/HBsAg 中和病毒感染,阻断病毒进入肝细胞 临床Ⅲ期 Elebsiran siRNA/HBV X基因 沉默HBV mRNA,降低HBsAg表达水平,抑制HDV病毒组装 临床Ⅲ期 JNJ-3989 siRNA/HBV X基因 沉默HBV mRNA,降低HBsAg表达水平,抑制HDV病毒组装 临床Ⅱ期 RBD1016 siRNA/HBV X基因 沉默HBV mRNA,降低HBsAg表达水平,抑制HDV病毒组装 临床Ⅱ期 贺普拉肽 多肽/NTCP 竞争性阻断病毒和受体结合,抑制病毒入侵 临床Ⅱ期 REP 2139 核酸聚合物/HBsAg HBsAg分泌抑制剂,抑制病毒颗粒组装和释放 临床Ⅱ期 ABI-6250 小分子药物/NTCP 抑制病毒入侵 临床Ⅰ期 HH-1270 小分子药物/NTCP 抑制病毒入侵 临床前 注:Bulevirtide,布来韦肽;Libevitug,立贝韦塔单抗;Tobevibart,托韦拜单抗;NTCP,钠离子牛磺胆酸共转运蛋白;PreS1,乙型肝炎病毒前S1抗原;HBsAg,乙型肝炎表面抗原;siRNA,小干扰RNA;HBV X,乙型肝炎病毒X基因;HBV,乙型肝炎病毒;HDV,丁型肝炎病毒;L-HDAg,大丁型肝炎病毒δ抗原;mRNA,信使RNA。
-
[1] MIAO ZJ, ZHANG SS, OU XM, et al. Estimating the global prevalence, disease progression, and clinical outcome of hepatitis delta virus infection[J]. J Infect Dis, 2020, 221( 10): 1677- 1687. DOI: 10.1093/infdis/jiz633. [2] ASSELAH T, RIZZETTO M. Hepatitis D virus infection[J]. N Engl J Med, 2023, 389( 1): 58- 70. DOI: 10.1056/nejmra2212151. [3] World Health Organization. Global hepatitis report 2024: Action for access in low- and middle-income countries[R]. Geneva: World Health Organization, 2024. [4] ZHANG ZF, URBAN S. New insights into HDV persistence: The role of interferon response and implications for upcoming novel therapies[J]. J Hepatol, 2021, 74( 3): 686- 699. DOI: 10.1016/j.jhep.2020.11.032. [5] WEDEMEYER H, YURDAYDÌN C, DALEKOS GN, et al. Peginterferon plus adefovir versus either drug alone for hepatitis delta[J]. N Engl J Med, 2011, 364( 4): 322- 331. DOI: 10.1056/NEJMoa0912696. [6] FARCI P, MANDAS A, COIANA A, et al. Treatment of chronic hepatitis D with interferon Alfa-2a[J]. N Engl J Med, 1994, 330( 2): 88- 94. DOI: 10.1056/NEJM199401133300202. [7] CASTELNAU C, LE GAL F, RIPAULT MP, et al. Efficacy of peginterferon alpha-2b in chronic hepatitis delta: Relevance of quantitative RT-PCR for follow-up[J]. Hepatology, 2006, 44( 3): 728- 735. DOI: 10.1002/hep.21325. [8] HEIDRICH B, YURDAYDıN C, KABAÇAM G, et al. Late HDV RNA relapse after peginterferon alpha-based therapy of chronic hepatitis delta[J]. Hepatology, 2014, 60( 1): 87- 97. DOI: 10.1002/hep.27102. [9] WRANKE A, HARDTKE S, HEIDRICH B, et al. Ten-year follow-up of a randomized controlled clinical trial in chronic hepatitis delta[J]. J Viral Hepat, 2020, 27( 12): 1359- 1368. DOI: 10.1111/jvh.13366. [10] RIZZETTO M, CANESE MG, ARICÒ S, et al. Immunofluorescence detection of new antigen-antibody system(delta/anti-delta) associated to hepatitis B virus in liver and in serum of HBsAg carriers[J]. Gut, 1977, 18( 12): 997- 1003. DOI: 10.1136/gut.18.12.997. [11] FLORES R, OWENS RA, TAYLOR J. Pathogenesis by subviral agents: Viroids and hepatitis delta virus[J]. Curr Opin Virol, 2016, 17: 87- 94. DOI: 10.1016/j.coviro.2016.01.022. [12] URBAN S, NEUMANN-HAEFELIN C, LAMPERTICO P. Hepatitis D virus in 2021: Virology, immunology and new treatment approaches for a difficult-to-treat disease[J]. Gut, 2021, 70( 9): 1782- 1794. DOI: 10.1136/gutjnl-2020-323888. [13] HOYER B, BONINO F, PONZETTO A, et al. Properties of delta-associated ribonucleic acid[J]. Prog Clin Biol Res, 1983, 143: 91- 97. [14] YAN H, ZHONG GC, XU GW, et al. Sodium taurocholate cotransporting polypeptide is a functional receptor for human hepatitis B and D virus[J]. eLife, 2012, 1: e00049. DOI: 10.7554/eLife.00049. [15] CHAO M, HSIEH SY, TAYLOR J. Role of two forms of hepatitis delta virus antigen: Evidence for a mechanism of self-limiting genome replication[J]. J Virol, 1990, 64( 10): 5066- 5069. DOI: 10.1128/JVI.64.10.5066-5069.1990. [16] SUREAU C, GUERRA B, LANFORD RE. Role of the large hepatitis B virus envelope protein in infectivity of the hepatitis delta virion[J]. J Virol, 1993, 67( 1): 366- 372. DOI: 10.1128/JVI.67.1.366-372.1993. [17] SUREAU C, NEGRO F. The hepatitis delta virus: Replication and pathogenesis[J]. J Hepatol, 2016, 64( 1 Suppl): S102- S116. DOI: 10.1016/j.jhep.2016.02.013. [18] STOCKDALE AJ, KREUELS B, HENRION MYR, et al. The global prevalence of hepatitis D virus infection: Systematic review and meta-analysis[J]. J Hepatol, 2020, 73( 3): 523- 532. DOI: 10.1016/j.jhep.2020.04.008. [19] PALOM A, RODRÍGUEZ-TAJES S, NAVASCUÉS CA, et al. Long-term clinical outcomes in patients with chronic hepatitis delta: The role of persistent viraemia[J]. Aliment Pharmacol Ther, 2020, 51( 1): 158- 166. DOI: 10.1111/apt.15521. [20] RIZZETTO M. Chronic hepatitis D; at a standstill[J]. Dig Dis, 2016, 34( 4): 303- 307. DOI: 10.1159/000444467. [21] YURDAYDIN C, ABBAS Z, BUTI M, et al. Treating chronic hepatitis delta: The need for surrogate markers of treatment efficacy[J]. J Hepatol, 2019, 70( 5): 1008- 1015. DOI: 10.1016/j.jhep.2018.12.022. [22] KARAIVAZOGLOU K, ICONOMOU G, TRIANTOS C, et al. Fatigue and depressive symptoms associated with chronic viral hepatitis patients’ health-related quality of life(HRQOL)[J]. Ann Hepatol, 2010, 9( 4): 419- 427. DOI: 10.1016/S1665-2681(19)31618-7. [23] ROMEO R, FOGLIENI B, CASAZZA G, et al. High serum levels of HDV RNA are predictors of cirrhosis and liver cancer in patients with chronic hepatitis delta[J]. PLoS One, 2014, 9( 3): e92062. DOI: 10.1371/journal.pone.0092062. [24] FATTOVICH G, BOSCARO S, NOVENTA F, et al. Influence of hepatitis delta virus infection on progression to cirrhosis in chronic hepatitis type B[J]. J Infect Dis, 1987, 155( 5): 931- 935. DOI: 10.1093/infdis/155.5.931. [25] ROMEO R, DEL NINNO E, RUMI M, et al. A 28-year study of the course of hepatitis Delta infection: A risk factor for cirrhosis and hepatocellular carcinoma[J]. Gastroenterology, 2009, 136( 5): 1629- 1638. DOI: 10.1053/j.gastro.2009.01.052. [26] European Association for the Study of the Liver. EASL clinical practice guidelines on the management of hepatitis B virus infection[J]. J Hepatol, 2025, 83( 2): 502- 583. DOI: 10.1016/j.jhep.2025.03.018. [27] ZUO SN, ZHAO JZ. Current status and considerations of clinical development of new drugs for the treatment of hepatitis D[J]. Chin J Clin Pharmacol, 2024, 40( 3): 464- 471. DOI: 10.13699/j.cnki.1001-6821.2024.03.033.左书凝, 赵建中. 丁型病毒性肝炎治疗新药临床研发现状及考量[J]. 中国临床药理学杂志, 2024, 40( 3): 464- 471. DOI: 10.13699/j.cnki.1001-6821.2024.03.033. [28] GHANY MG, BUTI M, LAMPERTICO P, et al. Guidance on treatment endpoints and study design for clinical trials aiming to achieve cure in chronic hepatitis B and D: Report from the 2022 AASLD-EASL HBV-HDV Treatment Endpoints Conference[J]. Hepatology, 2023, 78( 5): 1654- 1673. DOI: 10.1097/HEP.0000000000000431. [29] GRIPON P, CANNIE I, URBAN S. Efficient inhibition of hepatitis B virus infection by acylated peptides derived from the large viral surface protein[J]. J Virol, 2005, 79( 3): 1613- 1622. DOI: 10.1128/JVI.79.3.1613-1622.2005. [30] ENGELKE M, MILLS K, SEITZ S, et al. Characterization of a hepatitis B and hepatitis delta virus receptor binding site[J]. Hepatology, 2006, 43( 4): 750- 760. DOI: 10.1002/hep.21112. [31] European Association for the Study of the Liver. EASL clinical practice guidelines on hepatitis delta virus[J]. J Hepatol, 2023, 79( 2): 433- 460. DOI: 10.1016/j.jhep.2023.05.001. [32] WEDEMEYER H, ALEMAN S, BRUNETTO MR, et al. A phase 3, randomized trial of bulevirtide in chronic hepatitis D[J]. N Engl J Med, 2023, 389( 1): 22- 32. DOI: 10.1056/NEJMoa2213429. [33] LAMPERTICO P, ALEMAN S, BRUNETTO M, et al. P135 Efficacy and safety of 144 weeks of bulevirtide 2 mg or 10 mg monotherapy from the ongoing phase 3 study, MYR301[J]. Gut, 2024, 73( 3): A96– A97. DOI: 10.1136/gutjnl-2024-BASL.137. [34] WEDEMEYER H, ALEMAN S, BLANK A, et al. P158 Final results of MYR301: A randomised phase 3 study evaluating the efficacy and safety of up to 144 weeks of bulevirtide monotherapy for chronic hepatitis delta and 96 weeks of posttreatment follow-up[J]. Gut, 2025, 74( 4): A117. DOI: 10.1136/gutjnl-2025-BASL.168. [35] DEGASPERI E, ANOLLI MP, JACHS M, et al. Real-world effectiveness and safety of bulevirtide monotherapy for up to 96 weeks in patients with HDV-related cirrhosis[J]. J Hepatol, 2025, 82( 6): 1012- 1022. DOI: 10.1016/j.jhep.2024.12.044. [36] DIETZ-FRICKE C, DEGASPERI E, JACHS M, et al. Safety and efficacy of off-label bulevirtide monotherapy in patients with HDV with decompensated Child-B cirrhosis-a real-world case series[J]. Hepatology, 2024, 80( 3): 664- 673. DOI: 10.1097/HEP.0000000000000847. [37] ANOLLI MP, DEGASPERI E, ALLWEISS L, et al. A 3-year course of bulevirtide monotherapy may cure hdv infection in cirrhotics[J]. J Hepatol, 2023, 78( 4): 876- 880. DOI: 10.1016/j.jhep.2022.12.023. [38] LOGLIO A, FERENCI P, UCEDA RENTERIA SC, et al. Excellent safety and effectiveness of high-dose myrcludex-B monotherapy administered for 48 weeks in HDV-related compensated cirrhosis: A case report of 3 patients[J]. J Hepatol, 2019, 71( 4): 834- 839. DOI: 10.1016/j.jhep.2019.07.003. [39] ASSELAH T, LAMPERTICO P, ALEMAN S, et al. Bulevirtide monotherapy is safe and well tolerated in chronic hepatitis delta: An integrated safety analysis of bulevirtide clinical trials at week 48[J]. Liver Int, 2025, 45( 4): e16174. DOI: 10.1111/liv.16174. [40] WINDISCH MP, UNCHWANIWALA N, LI JC, et al. LBP-001 Preclinical profiling of ABI-6250, a first-in-class oral therapeutic candidate for chronic hepatitis[J]. J Hepatol, 2025, 82: S70. DOI: 10.1016/s0168-8278(25)00420-9. [41] LIU Y, ZHOU J, MAO F, et al. A novel oral NTCP inhibitor HH-1270 exhibits favorable properties as a clinical candidate for the treatment of HBV/HDV infection[C]// Proceedings of the 2024 International HBV Meeting. Chicago: 2024. [42] GRIPON P, LE SEYEC J, RUMIN S, et al. Myristylation of the hepatitis B virus large surface protein is essential for viral infectivity[J]. Virology, 1995, 213( 2): 292- 299. DOI: 10.1006/viro.1995.0002. [43] LE SEYEC J, CHOUTEAU P, CANNIE I, et al. Infection process of the hepatitis B virus depends on the presence of a defined sequence in the pre-S1 domain[J]. J Virol, 1999, 73( 3): 2052- 2057. DOI: 10.1128/JVI.73.3.2052-2057.1999. [44] CHOUTEAU P, LE SEYEC J, CANNIE I, et al. A short N-proximal region in the large envelope protein harbors a determinant that contributes to the species specificity of human hepatitis B virus[J]. J Virol, 2001, 75( 23): 11565- 11572. DOI: 10.1128/JVI.75.23.11565-11572.2001. [45] BLANCHET M, SUREAU C. Infectivity determinants of the hepatitis B virus pre-S domain are confined to the N-terminal 75 amino acid residues[J]. J Virol, 2007, 81( 11): 5841- 5849. DOI: 10.1128/JVI.00096-07. [46] LE DUFF Y, BLANCHET M, SUREAU C. The pre-S1 and antigenic loop infectivity determinants of the hepatitis B virus envelope proteins are functionally independent[J]. J Virol, 2009, 83( 23): 12443- 12451. DOI: 10.1128/JVI.01594-09. [47] SUREAU C, SALISSE J. A conformational heparan sulfate binding site essential to infectivity overlaps with the conserved hepatitis B virus a-determinant[J]. Hepatology, 2013, 57( 3): 985- 994. DOI: 10.1002/hep.26125. [48] LI D, HE WH, LIU XM, et al. A potent human neutralizing antibody Fc-dependently reduces established HBV infections[J]. eLife, 2017, 6: e26738. DOI: 10.7554/eLife.26738. [49] WANG XR, CHI XM, ZHANG YY, et al. LBP-24 Safety and efficacy of anti-pre-S1 domain monoclonal antibody(HH-003) treatment in patients with co-infection of chronic hepatitis B virus(HBV) and hepatitis D virus(HDV): A single center, open-label, phase 2 trial[J]. J Hepatol, 2023, 78: S117. DOI: 10.1016/S0168-8278(23)00606-2. [50] SAEED SH, YAO X, ZAIGHAM A, et al. Efficacy and safety of libevitug(HH-003) in patients with chronic hepatitis D virus infection: An international, multicenter, randomized, controlled, open-label phase 2 b Study(H H003- 204)[C]// Proceedings of The Liver Meeting, American Association for the Study of Liver Diseases. Washington, DC: 2025. [51] LEMPP FA, VOLZ T, CAMERONI E, et al. Potent broadly neutralizing antibody VIR-3434 controls hepatitis B and D virus infection and reduces HBsAg in humanized mice[J]. J Hepatol, 2023, 79( 5): 1129- 1138. DOI: 10.1016/j.jhep.2023.07.003. [52] RAMOS H, ALCALA R, MICHELET M, et al. WED-366 Characterization of BJT-778, an anti-HBsAg neutralizing monoclonal antibody for treatment of hepatitis B virus and hepatitis D virus infections[J]. J Hepatol, 2024, 80: S808. DOI: 10.1016/S0168-8278(24)02233-5. [53] ASSELAH T, CHATTERGOON MA, JUCOV A, et al. A phase 2 trial of tobevibart plus elebsiran in hepatitis D[J]. N Engl J Med, 2026, 394( 4): 343- 353. DOI: 10.1056/nejmoa2508827. [54] AGARWAL K, DOBRYANSKA M, JUCOV A, et al. BJT-778, anti-HBsAg monoclonal antibody, achieved 100% virologic response in subjects with chronic hepatitis D(CHD): Phase 2 study results[C]// Proceedings of The Liver Meeting, American Association for the Study of Liver Diseases. San Diego, CA: 2024. [55] AGARWAL K, JUCOV A, DOBRYANSKA M, et al. Brelovitug(BJT-778) monotherapy achieved 100% virologic response in patients with chronic hepatitis D: On treatment week 48 phase 2 study results[C]// Proceedings of The Liver Meeting, American Association for the Study of Liver Diseases. Washington, DC: 2025. [56] RIZZETTO M, HOYER B, CANESE MG, et al. Delta Agent: Association of delta antigen with hepatitis B surface antigen and RNA in serum of delta-infected chimpanzees[J]. Proc Natl Acad Sci USA, 1980, 77( 10): 6124- 6128. DOI: 10.1073/pnas.77.10.6124. [57] WANG CJ, CHEN PJ, WU JC, et al. Small-form hepatitis B surface antigen is sufficient to help in the assembly of hepatitis delta virus-like particles[J]. J Virol, 1991, 65( 12): 6630- 6636. DOI: 10.1128/JVI.65.12.6630-6636.1991. [58] YURDAYDIN C, KESKIN O, KALKAN Ç, et al. Optimizing lonafarnib treatment for the management of chronic delta hepatitis: The LOWR HDV-1 study[J]. Hepatology, 2018, 67( 4): 1224- 1236. DOI: 10.1002/hep.29658. [59] ETZION O, HAMID SS, ASSELAH T, et al. GS-012 Week 48 results of the phase 3 D-LIVR study, a randomized double-blind, placebo-controlled trial evaluating the safety and efficacy of Lonafarnib-boosted with Ritonavir with or without Peginterferon Alfa in patients with chronic hepatitis delta[J]. J Hepatol, 2023, 78: S10. DOI: 10.1016/S0168-8278(23)00451-8. [60] VAILLANT A. Oligonucleotide-based therapies for chronic HBV infection: A primer on biochemistry, mechanisms and antiviral effects[J]. Viruses, 2022, 14( 9): 2052. DOI: 10.3390/v14092052. [61] ASSELAH T, STREINU-CERCEL A, JUCOV A, et al. OS-127 Efficacy and safety of tobevibart(VIR-3434) alone or in combination with elebsiran(VIR-2218) in participants with chronic hepatitis delta virus infection: Preliminary results from the phase 2 SOLSTICE trial in non-cirrhotic and compensated cirrhotic participants[J]. J Hepatol, 2024, 80: S75- S76. DOI: 10.1016/S0168-8278(24)00565-8. [62] WOODDELL C, ZHU R, HAMILTON H, et al. Development of subcutaneously administered RNAi therapeutic ARO-HBV for chronic hepatitis B virus infection[J]. J Hepatol, 2018, 68: S18- S19. DOI: 10.1016/s0168-8278(18)30255-1. [63] SETO WK, LIANG ZC, GAN LM, et al. SAT-172 Safety and antiviral activity of RBD1016, a RNAi therapeutic, in Chinese subjects with chronic hepatitis B virus(HBV) infection[J]. J Hepatol, 2023, 78: S1152. DOI: 10.1016/S0168-8278(23)03287-7. [64] WEDEMEYER H, LAMPERTICO P, GANE EJ, et al. LBP-044 Robust reduction of HBsAg and HDV RNA levels with low risk for ALT elevations in JNJ-73763989 treated patients with chronic hepatitis D(CHD) and baseline HBsAg levels below 10, 000 IU/mL: Part 2 of the REEF-D study[J]. J Hepatol, 2024, 80: S100. DOI: 10.1016/s0168-8278(24)00611-1. [65] BAZINET M, PÂNTEA V, PLACINTA G, et al. Safety and efficacy of 48 weeks REP 2139 or REP 2165, tenofovir disoproxil, and pegylated interferon Alfa-2a in patients with chronic HBV infection Naïve to nucleos(t)ide therapy[J]. Gastroenterology, 2020, 158( 8): 2180- 2194. DOI: 10.1053/j.gastro.2020.02.058. [66] BAZINET M, PÂNTEA V, CEBOTARESCU V, et al. Safety and efficacy of REP 2139 and pegylated interferon Alfa-2a for treatment-naive patients with chronic hepatitis B virus and hepatitis D virus co-infection(REP 301 and REP 301-LTF): A non-randomised, open-label, phase 2 trial[J]. Lancet Gastroenterol Hepatol, 2017, 2( 12): 877- 889. DOI: 10.1016/S2468-1253(17)30288-1. [67] BAZINET M, PÂNTEA V, CEBOTARESCU V, et al. Persistent control of hepatitis B virus and hepatitis delta virus infection following REP 2139-Ca and pegylated interferon therapy in chronic hepatitis B virus/hepatitis delta virus coinfection[J]. Hepatol Commun, 2020, 5( 2): 189- 202. DOI: 10.1002/hep4.1633. -



 PDF下载 ( 767 KB)
PDF下载 ( 767 KB)

 下载:
下载:


