慢性乙型肝炎患者聚乙二醇干扰素α-2b治疗过程中不良反应的监测与管理
DOI: 10.12449/JCH260222
利益冲突声明:本文不存在任何利益冲突。
作者贡献声明:李欣怡负责设计论文框架,资料分析、撰写论文;陈丹蕾、张珈伟参与收集数据、论文修改;叶卫江负责拟定写作思路,指导撰写文章并最后定稿。
Monitoring and management of adverse reactions to pegylated interferon alpha-2b for treatment of chronic hepatitis B
-
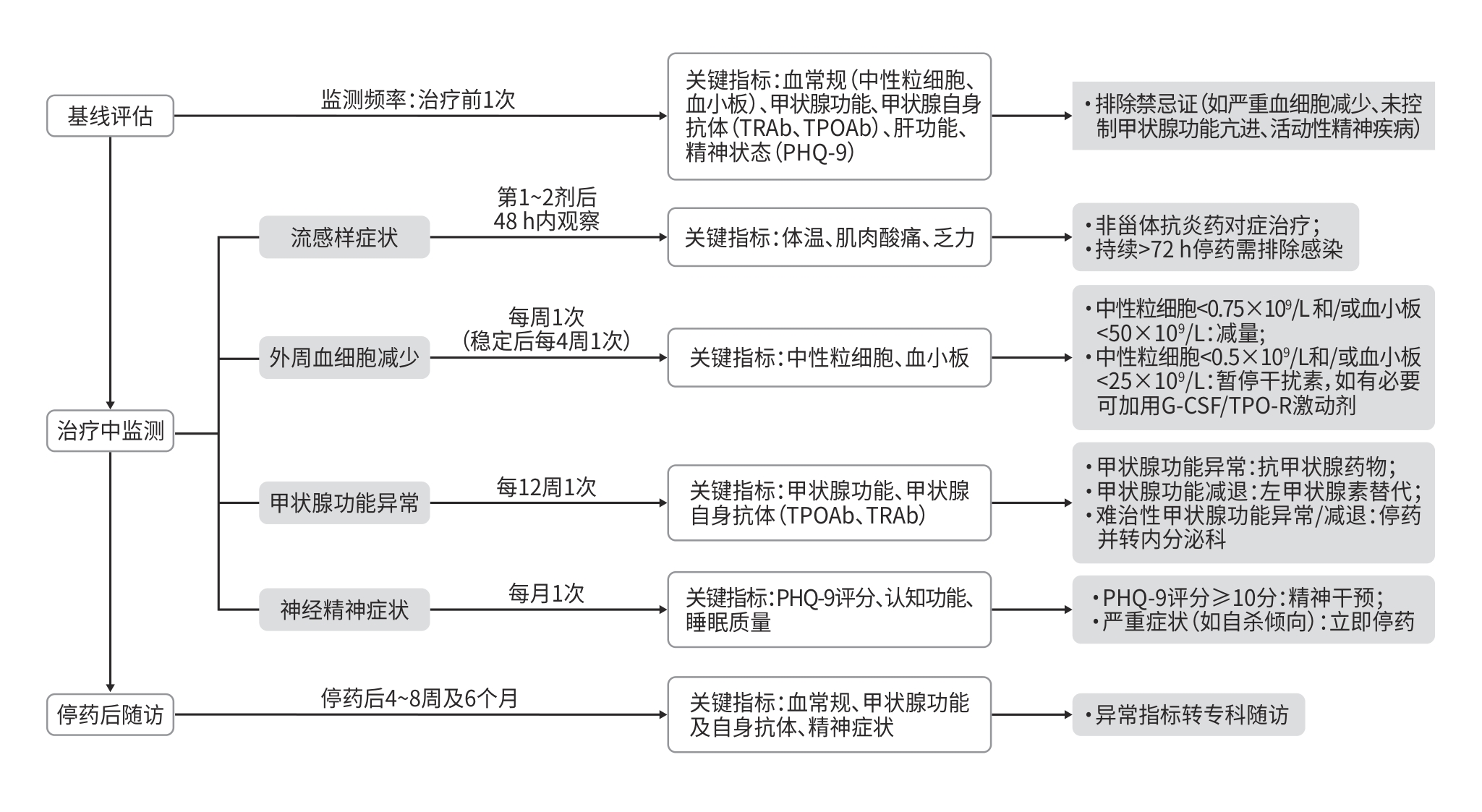
摘要: 聚乙二醇干扰素α-2b是当前治疗慢性乙型肝炎的一线药物,在临床中应用广泛。该药物具有抑制病毒复制、调节免疫、改善肝功能等多重作用,部分患者可实现临床治愈。然而,其在治疗过程中常伴随多种不良反应,这些不良反应常成为影响患者依从性、降低治疗效果的重要因素。本文系统综述了干扰素治疗慢性乙型肝炎时的不良反应及其发生机制,包括流感样症状、外周血细胞减少、甲状腺功能异常及神经精神症状等,并总结了相应的监测与管理策略,旨在为临床决策提供循证依据,同时强调个体化治疗的重要性。
-
关键词:
- 聚乙二醇干扰素 /
- 慢性乙型肝炎 /
- 药物相关性副作用和不良反应
Abstract: Pegylated interferon α-2b (PEG-IFN-α-2b) is currently a first-line drug for the treatment of chronic hepatitis B virus infection and is widely used in clinical practice. This drug has multiple effects of inhibiting viral replication, regulating immunity, and improving liver function, and some patients can achieve clinical cure. However, it often causes various adverse reactions during treatment, which are important factors for compromising treatment compliance and efficacy. This article systematically reviews the adverse reactions and their mechanisms during PEG-IFN-α-2b therapy for chronic hepatitis B, including influenza-like symptoms, peripheral blood cytopenia, thyroid dysfunction, and neuropsychiatric symptoms, and it also summarizes the monitoring and management strategies for these adverse reactions, in order to provide evidence-based guidance for clinical decision-making and emphasize the importance of individualized treatment. -
[1] Polaris Observatory Collaborators. Global prevalence, cascade of care, and prophylaxis coverage of hepatitis B in 2022: A modelling study[J]. Lancet Gastroenterol Hepatol, 2023, 8( 10): 879- 907. DOI: 10.1016/S2468-1253(23)00197-8. [2] QI JL, LI ML, WANG LJ, et al. National and subnational trends in cancer burden in China, 2005-20: An analysis of national mortality surveillance data[J]. Lancet Public Health, 2023, 8( 12): e943- e955. DOI: 10.1016/S2468-2667(23)00211-6. [3] CHEN J, QI M, FAN XG, et al. Efficacy of peginterferon Alfa-2b in nucleoside analogue experienced patients with negative HBeAg and low HBsAg: A non-randomized clinical trial[J]. Infect Dis Ther, 2021, 10( 4): 2259- 2270. DOI: 10.1007/s40121-021-00497-5. [4] JIANG SW, GUO SM, HUANG Y, et al. Interim analysis of the PARADISE study: Benefits of add-on peginterferon-α in NA-treated patients with CHB[J]. Antiviral Res, 2024, 226: 105892. DOI: 10.1016/j.antiviral.2024.105892. [5] YOSHIDA K, ENOMOTO M, TAMORI A, et al. Combination of entecavir or tenofovir with pegylated interferon-α for long-term reduction in hepatitis B surface antigen levels: Simultaneous, sequential, or add-on combination therapy[J]. Int J Mol Sci, 2021, 22( 3): 1456. DOI: 10.3390/ijms22031456. [6] ZHANG Y, LI YS, WANG J. Effect of ganfule capsule, tenofovir amibufenamide tablets combined with polyethylene glycol interferon α-2b in patients with chronic hepatitis B and its influence on immune function[J]. J Clin Exp Med, 2024, 23( 21): 2268- 2271. DOI: 10.3969/j.issn.1671-4695.2024.21.008.张阳, 李永胜, 王婧. 肝复乐胶囊、艾米替诺福韦片联合聚乙二醇干扰素α-2b治疗慢性乙型病毒性肝炎的临床疗效及对免疫功能的影响[J]. 临床和实验医学杂志, 2024, 23( 21): 2268- 2271. DOI: 10.3969/j.issn.1671-4695.2024.21.008. [7] Chinese Society of Hepatology, Chinese Medical Association; Chinese Society of Infectious Diseases, Chinese Medical Association. Guidelines for prevention and treatment of chronic hepatitis B[J]. Infect Dis Info, 2023, 36( 1): 1- 17. DOI: 10.3969/j.issn.1007-8134.2023.01.01.中华医学会肝病学分会, 中华医学会感染病学分会. 慢性乙型肝炎防治指南(2022年版)[J]. 传染病信息, 2023, 36( 1): 1- 17. DOI: 10.3969/j.issn.1007-8134.2023.01.01. [8] World Health Organization. Guidelines for the prevention, diagnosis, care and treatment for people with chronic hepatitis B infection[R/OL].( 2024-03-29)[ 2024-07-03]. https://www.who.int/publications/i/item/9789240090903. https: //www.who.int/publications/i/item/9789240090903 [9] QIN X, HOU ZY, MA L. Adverse reactions of interferon therapy in 63 patients with chronic hepatitis B and its countermeasures[J]. Heilongjiang Med J, 2018, 42( 6): 530- 531, 536. DOI: 10.3969/j.issn.1004-5775.2018.06.004.秦旭, 侯志云, 马丽. 63例慢性乙型肝炎患者应用干扰素治疗的不良反应及对策[J]. 黑龙江医学, 2018, 42( 6): 530- 531, 536. DOI: 10.3969/j.issn.1004-5775.2018.06.004. [10] GHERLAN GS, LAZAR SD, CULINESCU A, et al. Results of response-guided therapy with pegylated interferon alpha 2a in chronic hepatitis B and D[J]. Trop Med Infect Dis, 2024, 9( 4): 73. DOI: 10.3390/tropicalmed9040073. [11] SU ZZ, CHEN J, ZHANG JL, et al. Circulating IL-1β, IL-17, and IP-10 as potential predictors of hepatitis B virus infection prognosis[J]. J Immunol Res, 2022, 2022: 5202898. DOI: 10.1155/2022/5202898. [12] LI MH, LU HH, CHEN QQ, et al. Changes in the cytokine profiles of patients with chronic hepatitis B during antiviral therapy[J]. Biomed Environ Sci, 2021, 34( 6): 443- 453. DOI: 10.3967/bes2021.061. [13] WANG WX, JIA R, JIN XY, et al. Serum cytokine change profile associated with HBsAg loss during combination therapy with PEG-IFN-α in NAs-suppressed chronic hepatitis B patients[J]. Front Immunol, 2023, 14: 1121778. DOI: 10.3389/fimmu.2023.1121778. [14] LI ZZ, WU SL, ZHANG L, et al. Changes of peripheral blood during the antiviral treatment with Peg-IFNα-2 a for patients with chronic hepatitis B and the relationship between the changes and effect[J]. Chin J Exp Clin Virol, 2017, 31( 5): 446- 449. DOI: 10.3760/cma.j.issn.1003-9279.2017.05.015.李真真, 吴淑玲, 张璐, 等. 慢性乙型病毒性肝炎干扰素抗病毒治疗外周血的变化特征及与疗效关系[J]. 中华实验和临床病毒学杂志, 2017, 31( 5): 446- 449. DOI: 10.3760/cma.j.issn.1003-9279.2017.05.015. [15] Chinese Society of Hepatology, Chinese Medical Association; Chinese Society of Infectious Diseases, Chinese Medical Association. The guideline of prevention and treatment for chronic hepatitis B(2019 version)[J]. Chin J Infect Dis, 2005, 23( 6): 421- 431. DOI: 10.3760/j.issn:1000-6680.2005.06.021.中华医学会肝病学分会, 中华医学会感染病学分会. 慢性乙型肝炎防治指南[J]. 中华传染病杂志, 2005, 23( 6): 421- 431. DOI: 10.3760/j.issn:1000-6680.2005.06.021. [16] SATA M, YANO Y, YOSHIYAMA Y, et al. Mechanisms of thrombocytopenia induced by interferon therapy for chronic hepatitis B[J]. J Gastroenterol, 1997, 32( 2): 206- 210. DOI: 10.1007/BF02936369. [17] LIU YL, DU XF, CHEN XY, et al. Bone marrow suppression or active proliferation An analysis of neutropenia after pegylated interferon treatment of patients with chronic hepatitis C[J]. Scand J Infect Dis, 2013, 45( 12): 939- 943. DOI: 10.3109/00365548.2013.835067. [18] MELIA MT, BRÄU N, POORDAD F, et al. Infections during peginterferon/ribavirin therapy are associated with the magnitude of decline in absolute lymphocyte count: Results of the IDEAL study[J]. Clin Infect Dis, 2014, 58( 7): 960- 969. DOI: 10.1093/cid/ciu009. [19] HERMOS JA, QUACH L, GAGNON DR, et al. Incident severe thrombocytopenia in veterans treated with pegylated interferon plus ribavirin for chronic hepatitis C infection[J]. Pharmacoepidemiol Drug Saf, 2014, 23( 5): 480- 488. DOI: 10.1002/pds.3585. [20] MEI SQ, FENG Y, CUI LL, et al. Thrombotic thrombocytopenic Purpura developed after pegylated interferon treatment for hepatitis B infection[J]. BMC Nephrol, 2022, 23( 1): 400. DOI: 10.1186/s12882-022-03034-9. [21] WANG XR, XIAO L, GENG AW, et al. Pegylated interferon for treatment of chronic hepatitis B-induced thrombotic thrombocytopenic Purpura: A case report[J]. J Clin Hepatol, 2020, 36( 11): 2534- 2536. DOI: 10.3969/j.issn.1001-5256.2020.11.028.王欣茹, 肖丽, 耿爱文, 等. 聚乙二醇干扰素治疗慢性乙型肝炎诱发血栓性血小板减少性紫癜1例报告[J]. 临床肝胆病杂志, 2020, 36( 11): 2534- 2536. DOI: 10.3969/j.issn.1001-5256.2020.11.028. [22] DING L, QIAN C, ZHANG YH, et al. Hepatitis B complicated with thrombotic thrombocytopenic Purpura after interferon treatment: A case report[J]. Chin J Hematol, 2016, 37( 12): 1037. DOI: 10.3760/cma.j.issn.0253-2727.2016.12.005.丁磊, 钱琤, 张宇浩, 等. 乙型肝炎干扰素治疗期间合并血栓性血小板减少性紫癜一例[J]. 中华血液学杂志, 2016, 37( 12): 1037. DOI: 10.3760/cma.j.issn.0253-2727.2016.12.005. [23] JIA HY, THELWELL C, DILGER P, et al. Endothelial cell functions impaired by interferon in vitro: Insights into the molecular mechanism of thrombotic microangiopathy associated with interferon therapy[J]. Thromb Res, 2018, 163: 105- 116. DOI: 10.1016/j.thromres.2018.01.039. [24] LIAN JS, KUANG W, JIA HY, et al. Pegylated interferon-α-2b combined with tenofovir disoproxil fumarate, granulocyte-macrophage colony-stimulating factor, and hepatitis B vaccine treatment for naïve HBeAg-positive chronic hepatitis B patients: A prospective, multicenter, randomized controlled study[J]. J Med Virol, 2022, 94( 11): 5475- 5483. DOI: 10.1002/jmv.28003. [25] National Clinical Research Center for Infectious Diseases; Society of Hepatology, Beijing Medical Association; Translational Medicine Branch, Chinese Association of Gerontology and Geriatrics. Chinese expert consensus on clinical management of hepatopathy-related thrombocytopenia[J]. J Clin Hepatol, 2023, 39( 10): 2307- 2320. DOI: 10.3969/j.issn.1001-5256.2023.10.007.国家感染性疾病临床医学研究中心, 北京医学会肝病学分会, 中国老年学和老年医学学会转化医学分会. 肝病相关血小板减少症临床管理中国专家共识[J]. 临床肝胆病杂志, 2023, 39( 10): 2307- 2320. DOI: 10.3969/j.issn.1001-5256.2023.10.007. [26] KOLANIS S, VASILEIOU E, HATZIPANTELIS E, et al. Safety and efficacy of eltrombopag in children and adults with immune thrombocytopenia: A systematic review and meta-analysis[J]. Cardiovasc Hematol Agents Med Chem, 2021, 19( 1): 83- 92. DOI: 10.2174/1871525718666200910-161540. [27] HUANGFU T, LIN YM, RAO KM, et al. Effect of α-interferon in treating chronic hepatitis B on liver function, hepatitis B surface antigen and thyroid function[J]. Lab Med Clin, 2024, 21( 19): 2920- 2924. DOI: 10.3969/j.issn.1672-9455.2024.19.027.皇甫彤, 蔺咏梅, 饶珂萌, 等. α-干扰素治疗慢性乙型肝炎对患者肝功能、乙型肝炎表面抗原及甲状腺功能的影响[J]. 检验医学与临床, 2024, 21( 19): 2920- 2924. DOI: 10.3969/j.issn.1672-9455.2024.19.027. [28] LUO WF, WU S, CHEN HJ, et al. Thyroid dysfunction is associated with the loss of hepatitis B surface antigen in patients with chronic hepatitis B undergoing treatment with α-interferon[J]. J Int Med Res, 2021, 49( 6): 03000605211025139. DOI: 10.1177/03000605211025139. [29] MA ZX, QIN YL, JIA YD, et al. Thyroid dysfunction incidence and risk factors in Chinese chronic hepatitis B patients treated with pegylated interferon alpha: A long-term follow-up study[J]. J Viral Hepat, 2022, 29( 6): 412- 419. DOI: 10.1111/jvh.13667. [30] WANG LH, LI BQ, ZHAO H, et al. A systematic review and meta-analysis of endocrine-related adverse events associated with interferon[J]. Front Endocrinol, 2022, 13: 949003. DOI: 10.3389/fendo.2022.949003. [31] HASHAM A, ZHANG WJ, LOTAY V, et al. Genetic analysis of interferon induced thyroiditis(IIT): Evidence for a key role for MHC and apoptosis related genes and pathways[J]. J Autoimmun, 2013, 44: 61- 70. DOI: 10.1016/j.jaut.2013.04.002. [32] MAZZIOTTI G, SORVILLO F, PISCOPO M, et al. Innate and acquired immune system in patients developing interferon-alpha-related autoimmune thyroiditis: A prospective study[J]. J Clin Endocrinol Metab, 2005, 90( 7): 4138- 4144. DOI: 10.1210/jc.2005-0093. [33] CARACCIO N, GIANNINI R, CUCCATO S, et al. Type I interferons modulate the expression of thyroid peroxidase, sodium/iodide symporter, and thyroglobulin genes in primary human thyrocyte cultures[J]. J Clin Endocrinol Metab, 2005, 90( 2): 1156- 1162. DOI: 10.1210/jc.2004-1173. [34] CARACCIO N, CUCCATO S, PRATESI F, et al. Effect of type I interferon(s) on cell viability and apoptosis in primary human thyrocyte cultures[J]. Thyroid, 2009, 19( 2): 149- 155. DOI: 10.1089/thy.2008.0290. [35] LIU Q, LI JF, ZONG QY, et al. Interferon-induced polarization of M1 macrophages mediates antiviral activity against the hepatitis B virus via the hepcidin-ferroportin axis[J]. Int Immunopharmacol, 2024, 134: 112219. DOI: 10.1016/j.intimp.2024.112219. [36] KOTKOWSKA A, SEWERYNEK E, DOMAŃSKA D, et al. Single nucleotide polymorphisms in the STAT3 gene influence AITD susceptibility, thyroid autoantibody levels, and IL6 and IL17 secretion[J]. Cell Mol Biol Lett, 2015, 20( 1): 88- 101. DOI: 0.1515/cmble-2015-0004. [37] LIU YS, ZHENG YH, LIN X, et al. Analysis of clinical characteristics of thyroid disorders in patients with chronic hepatitis B treated with pegylated-interferon alpha[J]. BMC Endocr Disord, 2023, 23( 1): 115. DOI: 10.1186/s12902-023-01371-w. [38] MU H, XU DQ, LIU CY, et al. Relationship between HBsAG and TSH during interferon treatment in hepatitis B patients with low HBsAg level[J]. Chongqing Med J, 2024, 53( 18): 2826- 2829. DOI: 10.3969/j.issn.1671-8348.2024.18.020.木唤, 许丹青, 刘春云, 等. 低HBsAg水平乙型肝炎患者使用干扰素治疗期间HBsAg与TSH的关系[J]. 重庆医学, 2024, 53( 18): 2826- 2829. DOI: 10.3969/j.issn.1671-8348.2024.18.020. [39] HE QF, MAO CJ, CHEN ZL, et al. Dynamic changes of growth and thyroid function in young children with chronic hepatitis B treated with peginterferon monotherapy[J]. Pediatr Infect Dis J, 2025, 44( 2): 112- 117. DOI: 10.1097/INF.0000000000004567. [40] LAI JY, HO JX, KOW ASF, et al. Interferon therapy and its association with depressive disorders-A review[J]. Front Immunol, 2023, 14: 1048592. DOI: 10.3389/fimmu.2023.1048592. [41] HU CG, YUAN GS, HUANG HP, et al. Depression and anxiety caused by pegylated interferon treatment in patients with chronic hepatitis B and the therapeutic effects of escitalopram and alprazolam[J]. J South Med Univ, 2017, 37( 9): 1201- 1205. DOI: 10.3969/j.issn.1673-4254.2017.09.10.胡承光, 袁国盛, 黄华萍, 等. 长效干扰素治疗慢性乙型肝炎引发的精神心理异常及干预治疗效果分析[J]. 南方医科大学学报, 2017, 37( 9): 1201- 1205. DOI: 10.3969/j.issn.1673-4254.2017.09.10. [42] HU PJ, YU ML, CHEN MY, et al. Long-term risk of depression and the impact of risk factors among chronic hepatitis C patients after successful antiviral therapy: A nationwide real-world Taiwanese cohort(T-COACH)[J]. J Formos Med Assoc, 2025. DOI: 10.1016/j.jfma.2025.05.039.[ Epub ahead of print] [43] CHIU WC, SU YP, SU KP, et al. Recurrence of depressive disorders after interferon-induced depression[J]. Transl Psychiatry, 2017, 7( 2): e1026. DOI: 10.1038/tp.2016.274. [44] ZHANG MD, WAN MJ, WANG W, et al. Effect of interferon therapy on quality of life in patients with chronic hepatitis B[J]. Sci Rep, 2024, 14( 1): 2461. DOI: 10.1038/s41598-024-51292-4. -



 PDF下载 ( 757 KB)
PDF下载 ( 757 KB)

 下载:
下载:


