代谢相关脂肪性肝病的早期筛查策略
DOI: 10.12449/JCH260223
-
摘要: 代谢相关脂肪性肝病(MAFLD)是一种全球范围内高发的慢性肝脏疾病,及时精准干预可延缓病程,显著降低肝纤维化、肝硬化及肝癌等严重并发症的发生风险。传统活检技术结合代谢指标虽为金标准,但是作为一项有创检查,可能引发疼痛、出血等并发症,该现状促使科学研究将研究重点转向无创评估体系的构建。近年来,基于多维度检测策略的无创诊断技术不断更新,包括血清学模型、影像技术和临床算法等。本文系统综述了MAFLD在纤维化F1~F3期的筛查方法,重点探讨结合人工智能的深度学习模型,旨在为MAFLD的早期筛查提供思路,并为优化疾病管理策略提供科学参考。Abstract: Metabolic associated fatty liver disease (MAFLD) is a common chronic liver disease worldwide, and timely and precise intervention can delay disease progression and significantly reduce the risk of serious complications such as liver fibrosis, liver cirrhosis, and liver cancer. Although traditional liver biopsy combined with metabolic markers is the gold standard, it may cause complications such as pain and bleeding as an invasive examination, which has promoted scientific research to shift its focus to the construction of noninvasive assessment systems. In recent years, noninvasive diagnostic technologies based on multi-dimensional detection strategies have been continuously updated, including serological models, imaging techniques, and clinical algorithms. This article systematically reviews the screening methods for MAFLD during the fibrotic stages F1—F3, especially deep learning models based on artificial intelligence, in order to provide ideas for the early screening of MAFLD, as well as a scientific reference for optimizing disease management strategies.
-
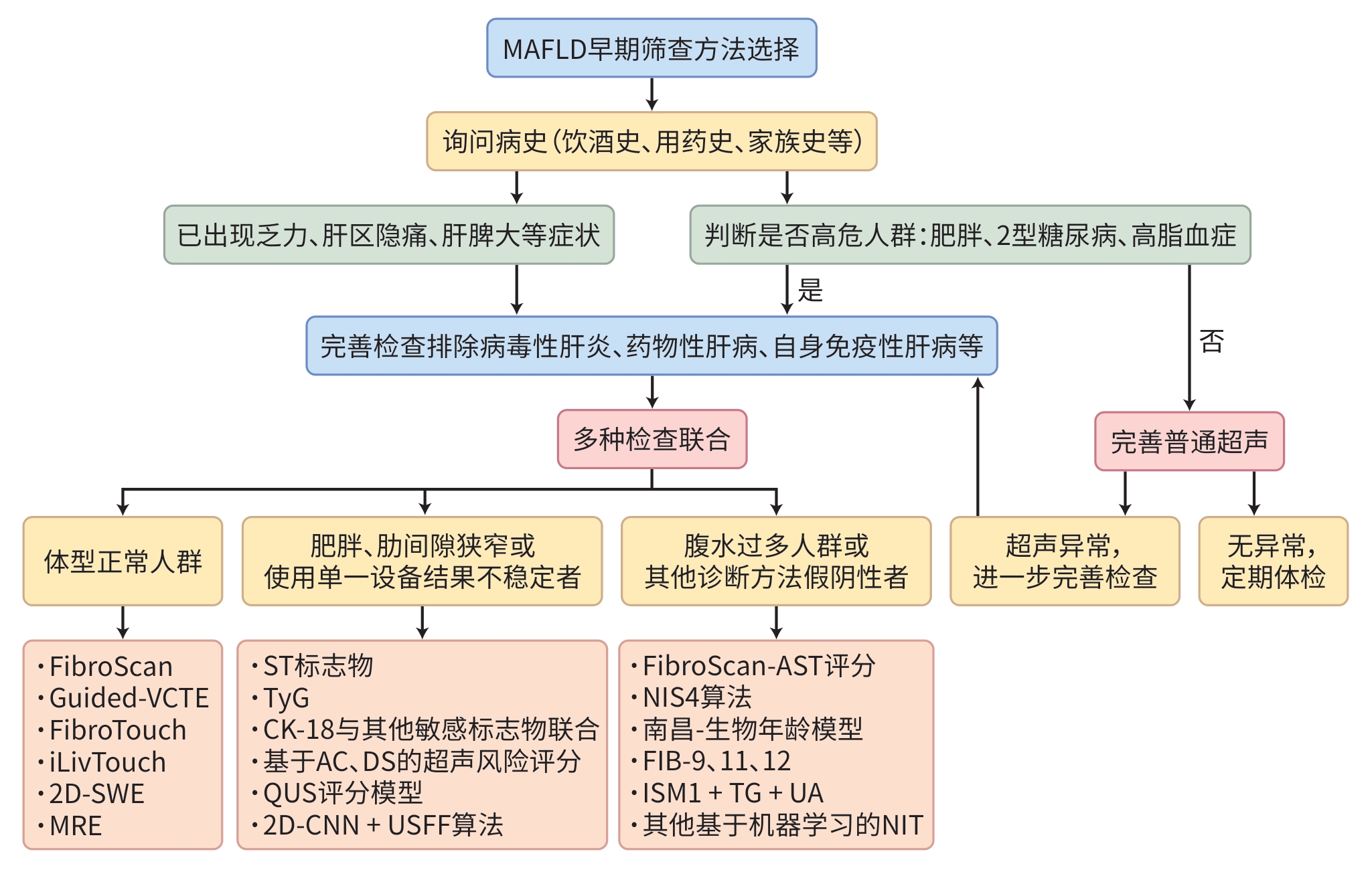
表 1 基于影像学早期筛查MAFLD的方法
Table 1. Early screening of MAFLD based on imaging methods
方法 优点 缺点 诊断效能差异 CT 易获得,清楚显示肝脏的形态、
结构及其与周围组织的关系存在辐射,孕妇及儿童等不适用 对轻度MAFLD诊断效能不佳 MRE 取样均一,无辐射,精准量化肝
脂肪含量,适合长期随访检查费用较高,植入心脏起搏器者不
适用特异度与敏感度均高,适用于初筛后
进一步确诊FibroScan 出现最早,占据欧美市场,不断
更新受肥胖、腹水和肋间隙狭窄等影响 适用于晚期肝炎与肝纤维化的评估 FibroTouch 无需根据肥胖程度选择探头型
号,仅需1个探头成本更高,个别特殊人群,如植入起
搏器患者、大量腹水患者等不适用对于轻中度脂肪变性更敏感 iLivTouch 检测效率更高,操作难度下降 普及率低,测量结果稳定性不高,在
腹水患者中效果同样不佳对于早期纤维化的筛查效果更佳 SWE 对肥胖患者准确性较高,可动
态监测病变区域设备昂贵,操作复杂 适用于专科医院对于肝纤维化精准
分期,不适合早期筛查MAFLDQUS 在不同亚组人群中均展现出稳
定的适用性依赖后处理算法,处理时间较长 各阶段诊断效能尚可 2D-CNN+
USFF算法适合MAFLD早期筛查 无法评估潜在的混杂因素,例如炎症
或纤维化对于临床疑似MAFLD患者诊断效能
较高FibroScan-AST 对体重、腹水等干扰因素适应
性更强基层医院缺少计算资源 在肝纤维化中晚期(≥F3)的诊断效能
优于早期Noureddin团队
机器学习模型能够减少代谢指标干扰 难以解释预测逻辑,接受度低 对于≥F2的患者敏感度和特异性平
衡较好注:CT,计算机体层成像;MRE,磁共振弹性成像;SWE,剪切波弹性成像技术;QUS,定量超声;USFF,超声脂肪分数;MAFLD,代谢相关脂肪性肝病。
-
[1] IMAI J, TAKASHIMIZU S, SUZUKI N, et al. Comparative study of MAFLD as a predictor of metabolic disease treatment for NAFLD[J]. Sci Rep, 2024, 14( 1): 13411. DOI: 10.1038/s41598-024-64301-3. [2] ZHANG L, MAO YL. Artificial intelligence in NAFLD: Will liver biopsy still be necessary in the future[J]. Healthcare, 2022, 11( 1): 117. DOI: 10.3390/healthcare11010117. [3] ZHANG JM, HUANG XC, DONG LY, et al. Epidemiology of lean/non-obese nonalcoholic fatty liver disease in China: A systematic review and meta-analysis[J]. Saudi Med J, 2023, 44( 9): 848- 863. DOI: 10.15537/smj.2023.44.9.20230021. [4] WANG YJ, WANG Q, ZHANG J, et al. The characteristics of lean non-alcoholic fatty liver disease in physical examination[J/CD]. Chin J Liver Dis: Electronic Edition, 2025, 17( 1): 57- 63. DOI: 10.3969/j.issn.1674-7380.2025.01.009.王援军, 王倩, 张婕, 等. 体检痩型非酒精性脂肪性肝病人群特征分析[J/CD]. 中国肝脏病杂志(电子版), 2025, 17( 1): 57- 63. DOI: 10.3969/j.issn.1674-7380.2025.01.009. [5] DE A, BHAGAT N, MEHTA M, et al. Central obesity is an independent determinant of advanced fibrosis in lean patients with nonalcoholic fatty liver disease[J]. J Clin Exp Hepatol, 2025, 15( 1): 102400. DOI: 10.1016/j.jceh.2024.102400. [6] LI YF, WANG XT, ZHANG J, et al. Applications of artificial intelligence(AI) in researches on non-alcoholic fatty liver disease(NAFLD): A systematic review[J]. Rev Endocr Metab Disord, 2022, 23( 3): 387- 400. DOI: 10.1007/s11154-021-09681-x. [7] ZHENG WW, YU H, ZHENG H. Application value of liver hardness measurement by two-dimensional ultrasonic shear wave elastography in nonalcoholic fatty liver disease[J]. J Clin Exp Med, 2024, 23( 7): 764- 767. DOI: 10.3969/j.issn.1671-4695.2024.07.024.郑伟伟, 俞慧, 郑慧. 二维超声剪切波弹性成像技术肝脏硬度测量在非酒精性脂肪性肝病疾病严重程度预测中的应用价值[J]. 临床和实验医学杂志, 2024, 23( 7): 764- 767. DOI: 10.3969/j.issn.1671-4695.2024.07.024. [8] MALLET M, SILAGHI CA, SULTANIK P, et al. Current challenges and future perspectives in treating patients with NAFLD-related cirrhosis[J]. Hepatology, 2024, 80( 5): 1270- 1290. DOI: 10.1097/HEP.0000000000000456. [9] AHMED N, KUMARI A, MURTY RS. FibroScan's evolution: a critical 20-year review[J]. J Ultrasound, 2025, 28( 2): 295- 304. DOI: 10.1007/s40477-024-00971-z. [10] BASTARD C, AUDIÈRE S, FOUCQUIER J, et al. Guided-VCTE: An enhanced FibroScan examination with improved guidance and applicability[J]. Ultrasound Med Biol, 2025, 51( 4): 628- 637. DOI: 10.1016/j.ultrasmedbio.2024.12.007. [11] SONG SJ, NOGAMI A, LIANG LY, et al. Performance of continuous controlled attenuation parameter and liver stiffness measurement by the novel SmartExam in metabolic dysfunction-associated steatotic liver disease[J]. Liver Int, 2024, 44( 5): 1167- 1175. DOI: 10.1111/liv.15862. [12] QU Y, SONG YY, CHEN CW, et al. Diagnostic performance of FibroTouch ultrasound attenuation parameter and liver stiffness measurement in assessing hepatic steatosis and fibrosis in patients with nonalcoholic fatty liver disease[J]. Clin Transl Gastroenterol, 2021, 12( 4): e00323. DOI: 10.14309/ctg.0000000000000323. [13] SUKARAM T, MAUNG ST, CHONGPISON Y, et al. Diagnostic performance of FibroTouch® in assessing hepatic steatosis and fibrosis in patients with metabolic dysfunction-associated steatotic liver disease: An Asian experience[J]. Ann Hepatol, 2025, 30( 1): 101753. DOI: 10.1016/j.aohep.2024.101753. [14] ÖZERCAN M, MELEKOĞLU ELLIK Z, PARMAKSıZ A, et al. Liver stiffness and steatosis measurements with iLivTouch and FibroScan: A comparative study[J]. Turk J Gastroenterol, 2024, 35( 8): 634- 642. DOI: 10.5152/tjg.2024.23531. [15] YAO MJ, WEN XJ, WANG LJ, et al. Establishment of a model for evaluating the severity of nonalcoholic fatty liver disease based on transient elastography parameters[J]. J Clin Hepatol, 2021, 37( 7): 1614- 1618. DOI: 10.3969/j.issn.1001-5256.2021.07.027.姚明解, 文夏杰, 王雷婕, 等. 基于瞬时弹性成像技术检测参数的非酒精性脂肪性肝病进展评估模型的建立[J]. 临床肝胆病杂志, 2021, 37( 7): 1614- 1618. DOI: 10.3969/j.issn.1001-5256.2021.07.027. [16] LINDVIG KP, THORHAUGE KH, HANSEN JK, et al. Development, validation, and prognostic evaluation of LiverPRO for the prediction of significant liver fibrosis in primary care: A prospective cohort study[J]. Lancet Gastroenterol Hepatol, 2025, 10( 1): 55- 67. DOI: 10.1016/S2468-1253(24)00274-7. [17] LIU FY, BI MS, JING X, et al. Multiparametric US for identifying metabolic dysfunction-associated steatohepatitis: A prospective multicenter study[J]. Radiology, 2024, 310( 3): e232416. DOI: 10.1148/radiol.232416. [18] YAMAGUCHI R, ODA T, NAGASHIMA K. Comparison of the diagnostic accuracy of shear wave elastography with transient elastography in adult nonalcoholic fatty liver disease: A systematic review and network meta-analysis of diagnostic test accuracy[J]. Abdom Radiol, 2025, 50( 2): 734- 746. DOI: 10.1007/s00261-024-04546-8. [19] SELVARAJ EA, MÓZES FE, JAYASWAL ANA, et al. Diagnostic accuracy of elastography and magnetic resonance imaging in patients with NAFLD: A systematic review and meta-analysis[J]. J Hepatol, 2021, 75( 4): 770- 785. DOI: 10.1016/j.jhep.2021.04.044. [20] MA QF, GONG LF, MA LY, et al. Application of ultrasound attenuation imaging and shear wave elastography in the diagnosis of metabolic fatty liver disease[J]. Biotechnol Genet Eng Rev, 2024, 40( 3): 2830- 2841. DOI: 10.1080/02648725.2023.2202537. [21] PAN LX, TIAN W, HUANG ZH, et al. Identification of a liver fibrosis and disease progression-related transcriptome signature in non-alcoholic fatty liver disease[J]. Int J Biochem Cell Biol, 2025, 180: 106751. DOI: 10.1016/j.biocel.2025.106751. [22] LIGUORI A, ESPOSTO G, AINORA ME, et al. Liver elastography for liver fibrosis stratification: A comparison of three techniques in a biopsy-controlled MASLD cohort[J]. Biomedicines, 2025, 13( 1): 138. DOI: 10.3390/biomedicines13010138. [23] JIANG H, QIN C, XU YM. Feasibility of shear wave elastography for assessing steatosis in early-stage non-alcoholic fatty liver disease[J]. PLoS One, 2025, 20( 5): e0324637. DOI: 10.1371/journal.pone.0324637. [24] JANG JK, LEE ES, SEO JW, et al. Two-dimensional shear-wave elastography and US attenuation imaging for nonalcoholic steatohepatitis diagnosis: A cross-sectional, multicenter study[J]. Radiology, 2022, 305( 1): 118- 126. DOI: 10.1148/radiol.220220. [25] ABDELHAMEED F, KITE C, LAGOJDA L, et al. Non-invasive scores and serum biomarkers for fatty liver in the era of metabolic dysfunction-associated steatotic liver disease(MASLD): A comprehensive review from NAFLD to MAFLD and MASLD[J]. Curr Obes Rep, 2024, 13( 3): 510- 531. DOI: 10.1007/s13679-024-00574-z. [26] GAO F, HE Q, LI G, et al. A novel quantitative ultrasound technique for identifying non-alcoholic steatohepatitis[J]. Liver Int, 2022, 42( 1): 80- 91. DOI: 10.1111/liv.15064. [27] JEON SK, LEE JM, JOO I, et al. Two-dimensional convolutional neural network using quantitative US for noninvasive assessment of hepatic steatosis in NAFLD[J]. Radiology, 2023, 307( 1): e221510. DOI: 10.1148/radiol.221510. [28] KURODA H, OGURI T, KAMIYAMA N, et al. Multivariable quantitative US parameters for assessing hepatic steatosis[J]. Radiology, 2023, 309( 1): e230341. DOI: 10.1148/radiol.230341. [29] RAVAIOLI F, DAJTI E, MANTOVANI A, et al. Diagnostic accuracy of FibroScan-AST(FAST) score for the non-invasive identification of patients with fibrotic non-alcoholic steatohepatitis: A systematic review and meta-analysis[J]. Gut, 2023, 72( 7): 1399- 1409. DOI: 10.1136/gutjnl-2022-328689. [30] CHANG D, TRUONG E, MENA EA, et al. Machine learning models are superior to noninvasive tests in identifying clinically significant stages of NAFLD and NAFLD-related cirrhosis[J]. Hepatology, 2023, 77( 2): 546- 557. DOI: 10.1002/hep.32655. [31] XIAO JF, ZHANG XX, CHANG LN, et al. Associations of four surrogate insulin resistance indexes with non-alcoholic steatohepatitis in Chinese patients with obesity: A cross-sectional study[J]. Endocrine, 2024, 86( 2): 546- 555. DOI: 10.1007/s12020-024-03888-z. [32] ZHANG F, HAN Y, WU YF, et al. Association between triglyceride glucose-body mass index and the staging of non-alcoholic steatohepatitis and fibrosis in patients with non-alcoholic fatty liver disease[J]. Ann Med, 2024, 56( 1): 2409342. DOI: 10.1080/07853890.2024.2409342. [33] BENDE R, HEREDEA D, RAŢIU I, et al. Association between visceral adiposity and the prediction of hepatic steatosis and fibrosis in patients with metabolic dysfunction-associated steatotic liver disease(MASLD)[J]. J Clin Med, 2025, 14( 10): 3405. DOI: 10.3390/jcm14103405. [34] ZHANG YX, WANG Y, YOU CL, et al. Analysis of related factors of abnormal liver function in patients with non-alcoholic fatty liver disease[J]. Chin J Med Offic, 2025, 53( 5): 522- 524, 528. DOI: 10.16680/j.1671-3826.2025.05.21.张月霞, 王宇, 尤丛蕾, 等. 非酒精性脂肪肝患者肝功能异常相关因素分析[J]. 临床军医杂志, 2025, 53( 5): 522- 524, 528. DOI: 10.16680/j.1671-3826.2025.05.21. [35] WU Y, ZHOU J, ZHANG J, et al. Cytokeratin 18 in nonalcoholic fatty liver disease: Value and application[J]. Expert Rev Mol Diagn, 2024, 24( 11): 1009- 1022. DOI: 10.1080/14737159.2024.2413941. [36] MOGNA-PELÁEZ P, ROMO-HUALDE A, RIEZU-BOJ JI, et al. Isoliquiritigenin in combination with visceral adipose tissue and related markers as a predictive tool for nonalcoholic fatty liver disease[J]. J Physiol Biochem, 2024, 80( 3): 639- 653. DOI: 10.1007/s13105-023-00998-6. [37] AKTAS G, KOCAK MZ, BILGIN S, et al. Uric acid to HDL cholesterol ratio is a strong predictor of diabetic control in men with type 2 diabetes mellitus[J]. Aging Male, 2020, 23( 5): 1098- 1102. DOI: 10.1080/13685538.2019.1678126. [38] LEI XH, CHEN HY, XU YX, et al. Serum isthmin-1 is a potential biomarker for metabolic dysfunction associated fatty liver disease in patients with metabolic syndrome and type 2 diabetes mellitus[J]. BMJ Open Diabetes Res Care, 2024, 12( 5): e004514. DOI: 10.1136/bmjdrc-2024-004514. [39] NDUMA BN, AL-AJLOUNI YA, NJEI B. The application of artificial intelligence(AI)-based ultrasound for the diagnosis of fatty liver disease: A systematic review[J]. Cureus, 2023, 15( 12): e50601. DOI: 10.7759/cureus.50601. [40] STEFANAKIS K, MINGRONE G, GEORGE J, et al. Accurate non-invasive detection of MASH with fibrosis F2-F3 using a lightweight machine learning model with minimal clinical and metabolomic variables[J]. Metabolism, 2025, 163: 156082. DOI: 10.1016/j.metabol.2024.156082. [41] GINTER-MATUSZEWSKA B, ADAMEK A, MAJCHRZAK M, et al. FibrAIm- The machine learning approach to identify the early stage of liver fibrosis and steatosis[J]. Int J Med Inform, 2025, 197: 105837. DOI: 10.1016/j.ijmedinf.2025.105837. [42] MATBOLI M, EL-ATTAR NE, ABDELBAKY I, et al. Unveiling NLR pathway signatures: EP300 and CPN60 markers integrated with clinical data and machine learning for precision NASH diagnosis[J]. Cytokine, 2025, 188: 156882. DOI: 10.1016/j.cyto.2025.156882. [43] DENG LR, HUANG J, YUAN H, et al. Biological age prediction and NAFLD risk assessment: A machine learning model based on a multicenter population in Nanchang, Jiangxi, China[J]. BMC Gastroenterol, 2025, 25( 1): 172. DOI: 10.1186/s12876-025-03752-y. [44] HARRISON SA, RATZIU V, BOURSIER J, et al. A blood-based biomarker panel(NIS4) for non-invasive diagnosis of non-alcoholic steatohepatitis and liver fibrosis: A prospective derivation and global validation study[J]. Lancet Gastroenterol Hepatol, 2020, 5( 11): 970- 985. DOI: 10.1016/S2468-1253(20)30252-1. [45] SANYAL AJ, SHANKAR SS, YATES KP, et al. Diagnostic performance of circulating biomarkers for non-alcoholic steatohepatitis[J]. Nat Med, 2023, 29( 10): 2656- 2664. DOI: 10.1038/s41591-023-02539-6. [46] XU K, ZHENG KI, ZHENG MH. External validation of the nonalcoholic steatohepatitis scoring system in patients with biopsy-proven nonalcoholic fatty liver disease in China[J]. Clin Gastroenterol Hepatol, 2021, 19( 2): 412- 413. DOI: 10.1016/j.cgh.2020.04.009. [47] RATZIU V, HARRISON SA, HAJJI Y, et al. NIS2+TM as a screening tool to optimize patient selection in metabolic dysfunction-associated steatohepatitis clinical trials[J]. J Hepatol, 2024, 80( 2): 209- 219. DOI: 10.1016/j.jhep.2023.10.038. [48] CALÈS P, CANIVET CM, COSTENTIN C, et al. A new generation of non-invasive tests of liver fibrosis with improved accuracy in MASLD[J]. J Hepatol, 2025, 82( 5): 794- 804. DOI: 10.1016/j.jhep.2024.11.049. [49] GE JY, WANG ZY, DENG Y, et al. Near-infrared lipid droplets polarity fluorescent probe for early diagnosis of nonalcoholic fatty liver disease[J]. Spectrochim Acta A Mol Biomol Spectrosc, 2024, 318: 124479. DOI: 10.1016/j.saa.2024.124479. [50] OH TG, KIM SM, CAUSSY C, et al. A universal gut-microbiome-derived signature predicts cirrhosis[J]. Cell Metab, 2020, 32( 5): 901. DOI: 10.1016/j.cmet.2020.10.015. [51] BAO SRL, DU XX, GE HY. Value of Helicobacter pylori infection combined with traditional risk factors in predicting the risk of metabolic associated fatty liver disease[J]. J Clin Hepatol, 2023, 39( 6): 1318- 1324. DOI: 10.3969/j.issn.1001-5256.2023.06.011.包萨如拉, 杜晓旭, 戈宏焱. 幽门螺杆菌感染联合传统危险因素预测代谢相关脂肪性肝病发生风险的价值分析[J]. 临床肝胆病杂志, 2023, 39( 6): 1318- 1324. DOI: 10.3969/j.issn.1001-5256.2023.06.011. [52] RIAZI K, AZHARI H, CHARETTE JH, et al. The prevalence and incidence of NAFLD worldwide: A systematic review and meta-analysis[J]. Lancet Gastroenterol Hepatol, 2022, 7( 9): 851- 861. DOI: 10.1016/S2468-1253(22)00165-0. [53] PREECHATHAMMAWONG N, CHAROENPITAKCHAI M, WONGSASON N, et al. Development of a diagnostic support system for the fibrosis of nonalcoholic fatty liver disease using artificial intelligence and deep learning[J]. Kaohsiung J Med Sci, 2024, 40( 8): 757- 765. DOI: 10.1002/kjm2.12850. [54] FORLANO R, MULLISH BH, GIANNAKEAS N, et al. High-throughput, machine learning-based quantification of steatosis, inflammation, ballooning, and fibrosis in biopsies from patients with nonalcoholic fatty liver disease[J]. Clin Gastroenterol Hepatol, 2020, 18( 9): 2081- 2090.e9. DOI: 10.1016/j.cgh.2019.12.025. -



 PDF下载 ( 760 KB)
PDF下载 ( 760 KB)

 下载:
下载:


