慢性乙型肝炎低病毒血症患者HBV RNA的表达及临床意义
DOI: 10.12449/JCH260311
Expression and clinical significance of HBV RNA in chronic hepatitis B patients with low-level viremia
-
摘要:
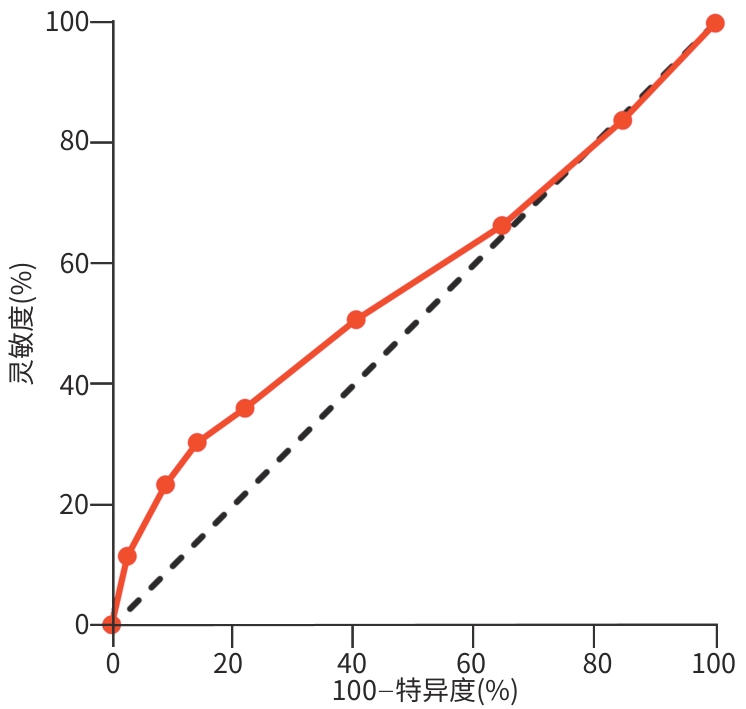
目的 探讨低病毒血症(LLV)患者血清乙型肝炎病毒(HBV)RNA的表达特征,并评估其对LLV的辅助诊断价值。 方法 选取2023年12月—2025年5月在遵义医科大学附属医院就诊的402例慢性乙型肝炎(CHB)患者,根据不同病毒载量分为完全病毒学应答(CVR)组(n=190,HBV DNA<20 IU/mL)和LLV组(n=212,20 IU/mL≤HBV DNA<2 000 IU/mL)。对两组患者年龄、性别、疾病类型、血清HBV RNA、乙型肝炎病毒e抗原(HBeAg)、丙氨酸氨基转移酶(ALT)、天冬氨酸氨基转移酶(AST)及总胆红素(TBil)等指标进行分析,同时根据LLV组中HBeAg状态分为HBeAg阴性组(n=140)和HBeAg阳性组(n=72)。计数资料两组间比较采用χ2检验;计量资料两组间比较采用Mann-Whitney U检验。通过多因素Logistic回归分析CHB患者发生LLV的影响因素。运用Spearman秩相关分析LLV组中HBV RNA与HBV DNA、ALT、AST及TBil的相关性。应用受试者操作特征(ROC)曲线评估HBV RNA对LLV的诊断效能。 结果 与CVR组相比,LLV组患者的血清HBV RNA水平更高[3(1~5) log10 copies/mL vs 2(1~3) log10 copies/mL,Z=-2.346,P=0.019],乙型肝炎肝硬化(31.13% vs 22.11%,χ2=4.155,P=0.042)及肝细胞癌(9.91% vs 4.74%,χ2=3.876,P=0.049)患者占比均较高。多因素Logistic回归分析显示,HBV RNA是CHB患者发生LLV的独立危险因素(比值比=1.163,95%置信区间:1.058~1.278,P=0.002)。在LLV患者中,HBeAg阳性者HBV RNA水平显著高于HBeAg阴性者[6(4~7) log10 copies/mL vs 2(1~3) log10 copies/mL,Z=-9.962,P<0.001]。相关性分析结果显示,LLV患者HBV RNA水平与HBV DNA、ALT、AST及TBil均无显著相关性(P值均>0.05)。ROC曲线分析表明,HBV RNA诊断LLV的曲线下面积为0.567(P=0.021),最佳截断值为4.5 log10 copies/mL,灵敏度为30.7%,特异度为85.8%。 结论 血清HBV RNA水平是CHB患者发生LLV的独立危险因素,尤其在HBeAg阳性患者中表达显著升高,可作为临床风险评估的潜在生物学标志物。 Abstract:Objective To investigate the expression characteristics of serum HBV RNA in patients with low-level viremia (LLV) and its value in the diagnosis of LLV. Methods A total of 402 chronic hepatitis B (CHB) patients who attended Affiliated Hospital of Zunyi Medical University from December 2023 to May 2025 were enrolled, and according to their viral load, they were divided into complete virologic response (CVR) group (190 patients with HBV DNA <20 IU/mL) and LLV group (212 patients with an HBV DNA level of ≥20 IU/mL and <2 000 IU/mL). The two groups were analyzed in terms of age, sex, disease type, serum HBV RNA, HBeAg, alanine aminotransferase (ALT), aspartate aminotransferase (AST), and total bilirubin (TBil), and according to HBeAg status, the patients in the LLV group were further divided into HBeAg-negative group with 140 patients and HBeAg-positive group with 72 patients. The chi-square test was used for comparison of categorical data between two groups, and the Mann-Whitney U test was used for comparison of continuous data between two groups. A multivariate Logistic regression analysis was used to investigate the influencing factors for LLV in CHB patients, and a Spearman’s rank correlation analysis was used to analyze the correlation of HBV RNA with HBV DNA, ALT, AST, and TBil in the LLV group. The receiver operating characteristic (ROC) curve was plotted to evaluate the efficacy of HBV RNA in the diagnosis of LLV. Results Compared with the CVR group, the LLV group had a significantly higher serum level of HBV RNA [3 (1 — 5) log10 copies/mL vs 2 (1 — 3) log10 copies/mL, Z=-2.346, P=0.019] and a significantly higher proportion of patients with hepatitis B cirrhosis (31.13% vs 22.11%, χ2=4.155, P=0.042) or hepatocellular carcinoma (9.91% vs 4.74%, χ2=3.876, P=0.049). The multivariate Logistic regression analysis showed that HBV RNA (odds ratio=1.163, 95% confidence interval: 1.058 — 1.278, P=0.002) was an independent risk factor for the onset of LLV in CHB patients. Among the patients with LLV, HBeAg-positive patients had a significantly higher level of HBV RNA than HBeAg-negative patients [6 (4 — 7) log10 copies/mL vs 2 (1 — 3) log10 copies/mL, Z=-9.962, P<0.001]. The correlation analysis showed that HBV RNA level had no significant correlation with HBV DNA, ALT, AST, or TBil in the patients with LLV (all P>0.05). The ROC curve analysis showed that HBV RNA had an AUC of 0.567 for the diagnosis of LLV (P=0.021), with an optimal cut-off value of 4.5 log10 copies/mL, a sensitivity of 30.7%, and a specificity of 85.8%. Conclusion Serum HBV RNA level is an independent risk factor for the development of LLV in CHB patients, and there is a significant increase in the expression of HBV RNA in HBeAg-positive patients. Therefore, it may serve as a potential biomarker for clinical risk assessment. -
Key words:
- Hepatitis B, Chronic /
- Low-Level Viremia /
- RNA, Viral /
- Diagnosis
-
表 1 CVR组和LLV组患者的基线特征及临床指标比较
Table 1. Comparison of baseline characteristics and clinical indicators between CVR and LLV groups
指标 CVR组(n=190) LLV组(n=212) 统计值 P值 年龄(岁) 42.5(35.0~50.0) 40.0(31.0~50.7) Z=-1.628 0.104 ≥60岁[例(%)] 12(6.32) 13(6.13) χ2 =0.006 0.939 性别[例(%)] χ2 =0.273 0.601 男 116(61.05) 124(58.49) 女 74(38.95) 88(41.51) 乙型肝炎肝硬化[例(%)] 42(22.11) 66(31.13) χ2 =4.155 0.042 乙型肝炎相关肝细胞癌[例(%)] 9(4.74) 21(9.91) χ2 =3.876 0.049 HBV RNA(log10 copies/mL) 2(1~3) 3(1~5) Z=-2.346 0.019 HBeAg阳性[例(%)] 50(26.32) 72(33.96) χ2 =2.772 0.096 ALT(U/L) 29.00(21.00~40.00) 31.00(20.00~46.00) Z=-1.034 0.301 AST(U/L) 29.00(24.00~35.00) 30.00(24.00~40.00) Z=-1.914 0.056 TBil(μmol/L) 14.45(11.17~18.85) 14.30(10.63~19.50) Z=-0.219 0.827 注:CVR,完全病毒学应答;LLV,低病毒血症;HBV,乙型肝炎病毒;HBeAg,乙型肝炎病毒e抗原;ALT,丙氨酸氨基转移酶;AST,天冬氨酸氨基转移酶;TBil,总胆红素。
表 2 CVR组和LLV组患者抗病毒情况
Table 2. Comparison of antiviral regimens between patients in the CVR and LLV groups
指标 CVR组(n=190) LLV组(n=212) 抗病毒药物[例(%)] ETV 80(42.11) 98(46.23) TDF 19(10.00) 46(21.70) TAF 71(37.37) 44(20.75) TMF 20(10.53) 1(0.47) 联合用药 0(0.00) 23(10.85) 抗病毒治疗时间[例(%)] ≥48周~96周 — 145(68.40) ≥96周~144周 — 51(24.06) ≥144周 — 16(7.55) 注:CVR,完全病毒学应答;LLV,低病毒血症;ETV,恩替卡韦;TDF,富马酸替诺福韦二吡呋酯;TAF,富马酸丙酚替诺福韦;TMF,艾米替诺福韦;—表示未记录。
表 3 CHB患者发生LLV的多因素Logistic回归分析
Table 3. Multivariate Logistic analysis of factors associated with LLV occurrence in chronic hepatitis B patients
指标 β值 SE Wald OR(95%CI) P值 肝硬化(无=0,有=1) 0.372 0.260 2.042 1.451(0.871~2.417) 0.153 肝细胞癌(无=0,有=1) 0.505 0.465 1.179 1.657(0.666~4.120) 0.278 HBV RNA(log10 copies/mL) 1.151 0.048 9.721 1.163(1.058~1.278) 0.002 注:CHB,慢性乙型肝炎;LLV,低病毒血症;HBV,乙型肝炎病毒;SE,标准误;OR,比值比;95%CI,95%置信区间。
表 4 HBeAg阴性组和HBeAg阳性组患者的指标比较
Table 4. Comparison of indicators between patients in the HBeAg-negative group and the HBeAg-positive group
指标 HBeAg阴性组
(n=140)
HBeAg阳性组
(n=72)
统计值 P值 乙型肝炎肝硬化[例(%)] 48(34.29) 18(25.00) χ2 =1.912 0.167 乙型肝炎相关肝细胞癌[例(%)] 16(11.43) 5(6.94) χ2 =1.071 0.301 HBV RNA(log10 copies/mL) 2(1~3) 6(4~7) Z=-9.962 <0.001 HBV DNA(log10 IU/mL) 2(1~2) 2(1~2) Z=-0.732 0.464 ALT(U/L) 31.40(19.25~44.75) 31.00(22.00~48.00) Z=-0.900 0.368 AST(U/L) 28.50(24.00~40.75) 30.00(25.00~40.00) Z=-0.124 0.901 TBil(μmol/L) 14.60(11.00~20.13) 13.70(9.75~18.88) Z=-1.202 0.229 注:HBeAg,乙型肝炎病毒e抗原;HBV,乙型肝炎病毒;ALT,丙氨酸氨基转移酶;AST,天冬氨酸氨基转移酶;TBil,总胆红素。
-
[1] KAKALOU C, POLYCHRONIDOU E, DROSOU V, et al. RiskRadar: Development and pilot results of a technical intervention targeting combination prevention regarding HIV, viral hepatitis, sexually transmitted infections and tuberculosis[J]. BMC Infect Dis, 2021, 21( Suppl 2): 866. DOI: 10.1186/s12879-021-06501-0. [2] Korean Association for the Study of the Liver. KASL clinical practice guidelines for management of chronic hepatitis B[J]. Clin Mol Hepatol, 2022, 28( 2): 276- 331. DOI: 10.3350/cmh.2022.0084. [3] ZHANG Q, CAI DC, HU P, et al. Low-level viremia in nucleoside analog-treated chronic hepatitis B patients[J]. Chin Med J, 2021, 134( 23): 2810- 2817. DOI: 10.1097/cm9.0000000000001793. [4] WANG LJ, GU ZQ, XU ZM, et al. A possible mechanism for low-level viremia occurrence in nucleos(t)ide analog-treated chronic hepatitis B patients[J]. Chin J Hepatol, 2021, 29( 12): 1151- 1155. DOI: 10.3760/cma.j.cn501113-20211124-00578.王雷婕, 顾智强, 许梓萌, 等. 核苷(酸)类药物经治慢性乙型肝炎患者低病毒血症发生的可能机制[J]. 中华肝脏病杂志, 2021, 29( 12): 1151- 1155. DOI: 10.3760/cma.j.cn501113-20211124-00578. [5] YIN GQ, LI J, ZHONG B, et al. New therapeutic options for persistent low-level viremia in patients with chronic hepatitis B virus infection: Increase of entecavir dosage[J]. World J Gastroenterol, 2021, 27( 8): 666- 676. DOI: 10.3748/wjg.v27.i8.666. [6] LI ZB, CHEN DD, JIA YF, et al. Risk factors related to low-level viraemia in chronic hepatitis B patients receiving entecavir treatment[J]. Front Cell Infect Microbiol, 2024, 14: 1413589. DOI: 10.3389/fcimb.2024.1413589. [7] YANG J, CHOI WM, SHIM JH, et al. Low level of hepatitis B viremia compared with undetectable viremia increases the risk of hepatocellular carcinoma in patients with untreated compensated cirrhosis[J]. Am J Gastroenterol, 2023, 118( 6): 1010- 1018. DOI: 10.14309/ajg.0000000000002181. [8] CHEN H, FU JJ, LI L, et al. Risk factors of low-level viremia in chronic hepatitis B patients receiving Entecavir monotherapy: A retrospective cohort study[J]. J Gastroenterol Hepatol, 2024, 39( 1): 180- 184. DOI: 10.1111/jgh.16357. [9] SUN YM, WU XN, ZHOU JL, et al. Persistent low level of hepatitis B virus promotes fibrosis progression during therapy[J]. Clin Gastroenterol Hepatol, 2020, 18( 11): 2582- 2591. DOI: 10.1016/j.cgh.2020.03.001. [10] OGAWA E, NOMURA H, NAKAMUTA M, et al. Tenofovir alafenamide after switching from entecavir or nucleos(t)ide combination therapy for patients with chronic hepatitis B[J]. Liver Int, 2020, 40( 7): 1578- 1589. DOI: 10.1111/liv.14482. [11] HERSHKOVICH L, COTLER SJ, SHEKHTMAN L, et al. HBV serum RNA kinetics during nucleic acid polymers based therapy predict functional cure[J]. Antiviral Res, 2025, 234: 106061. DOI: 10.1016/j.antiviral.2024.106061. [12] RIVEIRO-BARCIELA M, PERICÀS JM, BUTI M. How to interpret viral markers in the management of chronic hepatitis B infection[J]. Clin Microbiol Infect, 2022, 28( 3): 355- 361. DOI: 10.1016/j.cmi.2021.10.020. [13] KOSTYUSHEVA A, KOSTYUSHEV D, BREZGIN S, et al. Clinical implications of hepatitis B virus RNA and covalently closed circular DNA in monitoring patients with chronic hepatitis B today with a gaze into the future: The field is unprepared for a sterilizing cure[J]. Genes, 2018, 9( 10): 483. DOI: 10.3390/genes9100483. [14] Chinese Society of Hepatology, Chinese Medical Association; Chinese Society of Infectious Diseases, Chinese Medical Association. Guidelines for the prevention and treatment of chronic hepatitis B(version 2022)[J]. Infect Dis Info, 2023, 36( 1): 1- 17. DOI: 10.3969/j.issn.1007-8134.2023.01.01.中华医学会肝病学分会, 中华医学会感染病学分会. 慢性乙型肝炎防治指南(2022年版)[J]. 传染病信息, 2023, 36( 1): 1- 17. DOI: 10.3969/j.issn.1007-8134.2023.01.01. [15] National Health Commission of the People’s Republic of China. Standard for diagnosis and treatment of primary liver cancer(2024 edition)[J]. J Clin Hepatol, 2024, 40( 5): 893- 918. DOI: 10.12449/JCH240508.中华人民共和国国家卫生健康委员会. 原发性肝癌诊疗指南(2024年版)[J]. 临床肝胆病杂志, 2024, 40( 5): 893- 918. DOI: 10.12449/JCH240508. [16] WONG RJ, KAUFMAN HW, NILES JK, et al. Simplifying treatment criteria in chronic hepatitis B: Reducing barriers to elimination[J]. Clin Infect Dis, 2023, 76( 3): e791- e800. DOI: 10.1093/cid/ciac385. [17] WANG XM, WANG ZF, CHI XM, et al. Efficacy of a combination of HBV RNA and HBeAg in predicting HBeAg seroconversion in patients treated with entecavir for 144 weeks[J]. Int J Infect Dis, 2020, 99: 171- 178. DOI: 10.1016/j.ijid.2020.07.031. [18] JIA W, ZHU MQ, QI X, et al. Serum hepatitis B virus RNA levels as a predictor of HBeAg seroconversion during treatment with peginterferon alfa-2a[J]. Virol J, 2019, 16( 1): 61. DOI: 10.1186/s12985-019-1152-6. [19] TIAN LY, ZHAN AQ, AN Y, et al. Clinical significance of HBV RNA in chronic hepatitis B patients with low-level viremia after NAs treatment[J]. J Mod Med Health, 2025, 41( 5): 1090- 1094. DOI: 10.3969/j.issn.1009-5519.2025.05.008.田丽艳, 詹爱琴, 安轶, 等. HBV RNA在经治慢乙肝低病毒血症患者中的临床意义[J]. 现代医药卫生, 2025, 41( 5): 1090- 1094. DOI: 10.3969/j.issn.1009-5519.2025.05.008. [20] LYU CX, CHEN M, WANG JC, et al. Risk factors and mechanism of low-level viremia in patients with chronic hepatitis B receiving nucleos(t)ide analogues treatment[J]. J Mod Lab Med, 2023, 38( 5): 133- 137, 184. DOI: 10.3969/j.issn.1671-7414.2023.05.025.吕承秀, 陈梅, 王纪传, 等. 慢性乙型肝炎患者接受核苷(酸)类似物治疗后发生低病毒血症的危险因素及机制研究[J]. 现代检验医学杂志, 2023, 38( 5): 133- 137, 184. DOI: 10.3969/j.issn.1671-7414.2023.05.025. [21] CHEN H, FU JJ, LI L, et al. Influencing factors for low-level viremia in chronic hepatitis B patients treated with long-term entecavir antiviral therapy[J]. J Clin Hepatol, 2021, 37( 3): 556- 559. DOI: 10.3969/j.issn.1001-5256.2021.03.011.陈贺, 傅涓涓, 李丽, 等. 长期恩替卡韦经治慢性乙型肝炎患者低病毒血症的相关影响因素[J]. 临床肝胆病杂志, 2021, 37( 3): 556- 559. DOI: 10.3969/j.issn.1001-5256.2021.03.011. [22] ZHANG XJ, WU R, HUANG W, et al. Clinical study of hepatitis B virus RNA in different HBeAg states in chronic hepatitis B[J]. Chin J Health Lab Technol, 2023, 33( 3): 257- 260.张晓晶, 武瑞, 黄伟, 等. 乙型肝炎病毒RNA在慢性乙型病毒性肝炎不同HBeAg状态下的临床研究[J]. 中国卫生检验杂志, 2023, 33( 3): 257- 260. [23] HOOFNAGLE J, DI BISCEGLIE A. Serologic diagnosis of acute and chronic viral hepatitis[J]. Semin Liver Dis, 1991, 11( 2): 73- 83. DOI: 10.1055/s-2008-1040426. [24] FLORES JE, THOMPSON AJ, RYAN M, et al. The global impact of hepatitis B vaccination on hepatocellular carcinoma[J]. Vaccines, 2022, 10( 5): 793. DOI: 10.3390/vaccines10050793. [25] LI ZB, LI L, NIU XX, et al. Switching from entecavir to tenofovir alafenamide for chronic hepatitis B patients with low-level viraemia[J]. Liver Int, 2021, 41( 6): 1254- 1264. DOI: 10.1111/liv.14786. [26] HUANG DQ, TAMAKI N, LEE HW, et al. Outcome of untreated low-level viremia versus antiviral therapy-induced or spontaneous undetectable HBV-DNA in compensated cirrhosis[J]. Hepatology, 2023, 77( 5): 1746- 1756. DOI: 10.1097/HEP.0000000000000037. [27] ZHANG Q, PENG H, LIU X, et al. Chronic hepatitis B infection with low level viremia correlates with the progression of the liver disease[J]. J Clin Transl Hepatol, 2021, 9( 6): 850- 859. DOI: 10.14218/JCTH.2021.00046. [28] LIU S, DENG R, ZHOU B, et al. Association of serum hepatitis B virus RNA with hepatocellular carcinoma risk in chronic hepatitis B patients under nucleos(t)ide analogues therapy[J]. J Infect Dis, 2022, 226( 5): 881- 890. DOI: 10.1093/infdis/jiab597. [29] HE MW, YANG WC, WANG CY, et al. Influence of antiviral treatment adjustment on the prognosis of chronic hepatitis B patients with low-level viremia[J]. J Clin Hepatol, 2025, 41( 6): 1056- 1061. DOI: 10.12449/JCH250609.贺梦雯, 杨武才, 王春艳, 等. 调整抗病毒治疗方案对慢性乙型肝炎低病毒血症患者预后的影响[J]. 临床肝胆病杂志, 2025, 41( 6): 1056- 1061. DOI: 10.12449/JCH250609. [30] WANG XM, CHI XM, WU RH, et al. Serum HBV RNA correlated with intrahepatic cccDNA more strongly than other HBV markers during peg-interferon treatment[J]. Virol J, 2021, 18( 1): 4. DOI: 10.1186/s12985-020-01471-2. [31] FAN XL, ZHAN AQ, AN Y, et al. Correlation between serum HBV RNA and duration of treatment with nucleos(t)ide analogues in patients with chronic hepatitis B[J]. J Clin Hepatol, 2024, 40( 1): 58- 63. DOI: 10.12449/JCH240111.范雪莉, 詹爱琴, 安轶, 等. 慢性乙型肝炎患者血清HBV RNA水平与核苷(酸)类似物治疗时间的关系[J]. 临床肝胆病杂志, 2024, 40( 1): 58- 63. DOI: 10.12449/JCH240111. [32] CHEN Y, LUO XY, CHEN YH, et al. Expression of HBV RNA in hepatitis B virus-associated hepatocellular carcinoma[J]. J Pract Med, 2025, 41( 21): 3435- 3441. DOI: 10.3969/j.issn.1006-5725.2025.21.020.陈娅, 罗晓永, 陈应华, 等. HBV RNA预测乙型肝炎病毒相关肝细胞癌发病的价值[J]. 实用医学杂志, 2025, 41( 21): 3435- 3441. DOI: 10.3969/j.issn.1006-5725.2025.21.020. -



 PDF下载 ( 787 KB)
PDF下载 ( 787 KB)

 下载:
下载:


