胸腺素β4对四氯化碳诱导的肝纤维化小鼠模型的影响及其作用机制
DOI: 10.12449/JCH260314
利益冲突声明:本文不存在任何利益冲突。
作者贡献声明:朱耘函负责设计论文框架,撰写文章;王思琪负责实验操作,研究过程的实施;景登娅负责数据收集,统计学分析,绘制图表;冯勤颖负责论文修改,拟定写作思路,指导撰写文章并最后定稿。
Effect of thymosin β4 on a mouse model of carbon tetrachloride-induced hepatic fibrosis and its mechanism
-
摘要:
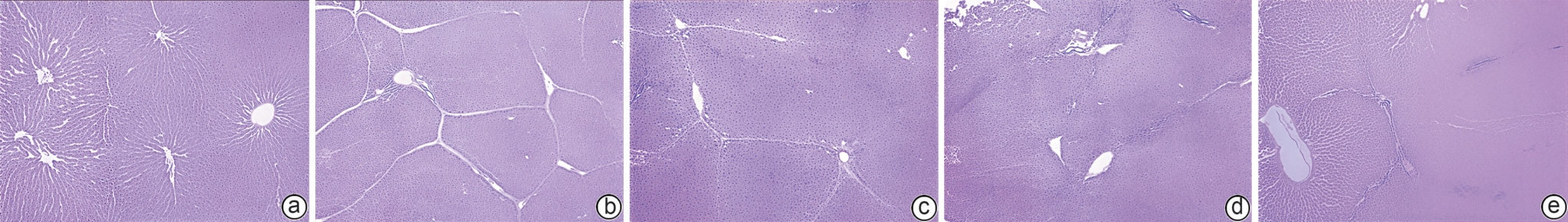
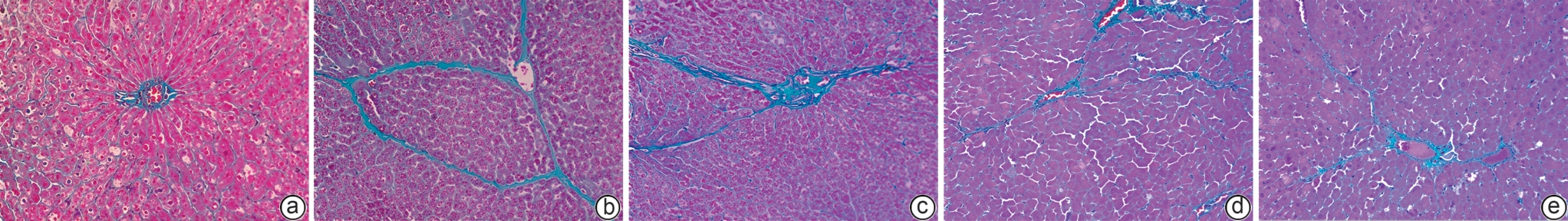
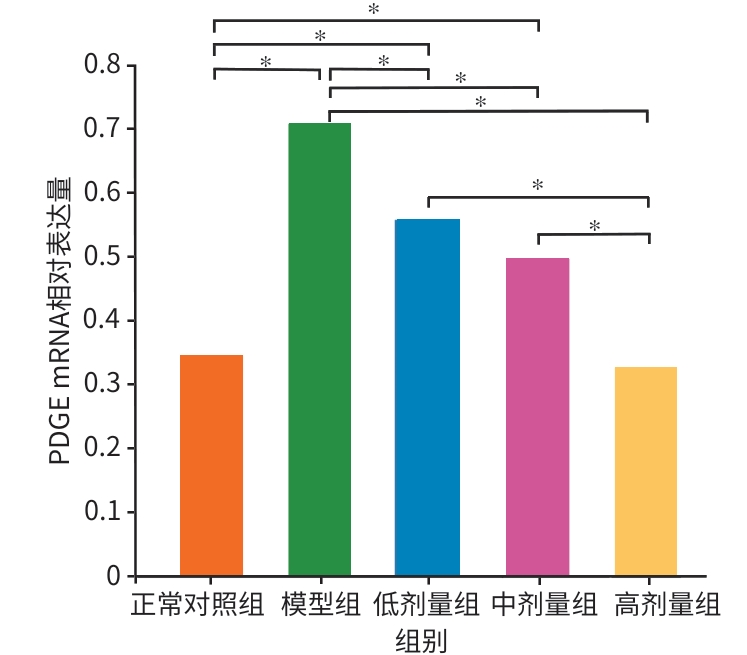
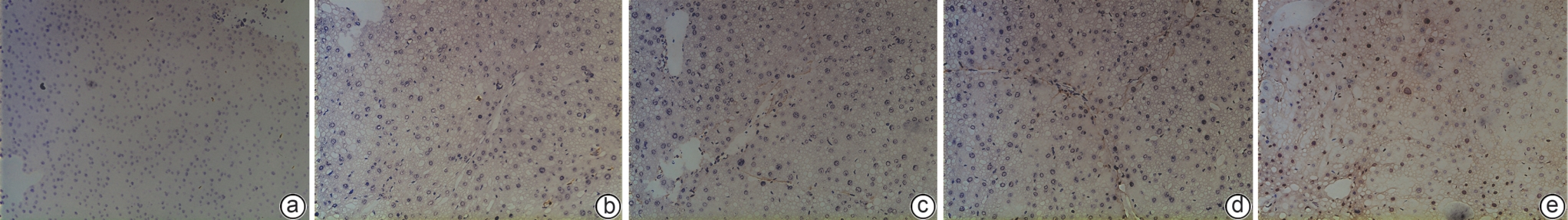
目的 本研究拟探讨胸腺素β4(Tβ4)通过调控血小板衍生生长因子(PDGF)表达及诱导肝星状细胞(HSC)凋亡,对四氯化碳(CCl4)诱导的肝纤维化小鼠模型的干预作用与潜在机制,为临床抗肝纤维化治疗提供新的实验依据。 方法 选取30只雄性C57小鼠,随机分为正常对照组、模型组,以及Tβ4低(3 mg/kg)、中(6 mg/kg)和高(12 mg/kg)剂量治疗组,每组6只。正常对照组自由摄食普通饲料,其余各组腹腔注射50% CCl4与橄榄油混合液建立肝纤维化模型,经超声及病理学验证造模成功后,对各治疗组小鼠连续皮下注射Tβ4干预4周。实验结束后取肝组织行苏木精-伊红及Masson染色观察组织病理变化,应用实时荧光定量聚合酶链反应检测血PDGF mRNA表达,原位末端转移酶标记(TUNEL)法检测HSC凋亡情况。计量资料多组间比较采用单因素方差分析,进一步两两比较采用LSD-t检验。 结果 与模型组比较,Tβ4中、高剂量治疗组小鼠肝纤维化程度均呈现不同程度的减轻。实时荧光定量聚合酶链反应结果显示,Tβ4可显著下调肝组织中PDGF mRNA表达,各剂量治疗组间差异有统计学意义(P<0.05);Tβ4高剂量组与正常对照组PDGF mRNA表达差异无统计学意义(P<0.05)。TUNEL检测结果显示,Tβ4中、高剂量组HSC凋亡数量显著多于模型组。 结论 Tβ4可能通过抑制PDGF表达和促进HSC凋亡,从而改善CCl4诱导的小鼠肝纤维化,提示其在肝纤维化治疗中具有潜在应用价值。 -
关键词:
- 肝纤维化 /
- 胸腺素β4 /
- 小鼠, 近交C57BL
Abstract:Objective To investigate the therapeutic effect and potential mechanism of thymosin β4 (Tβ4) on carbon tetrachloride (CCl4)-induced hepatic fibrosis by regulating the expression of platelet-derived growth factor (PDGF) and inducing the apoptosis of hepatic stellate cell (HSC), and to provide new experimental evidence for anti-hepatic fibrosis treatment in clinical practice. Methods A total of 30 male C57 mice were randomly divided into normal control group, model group, low-dose Tβ4 treatment group (3 mg/kg), middle-dose Tβ4 treatment group (6 mg/kg), and high-dose Tβ4 treatment group (12 mg/kg), with 6 mice in each group. The mice in the normal control group were fed with a normal diet ad libitum, and those in the other groups were given intraperitoneal injection of 50% CCl4 mixed with olive oil to establish a model of hepatic fibrosis. After successful modeling confirmed by ultrasound and histopathology, the mice in each treatment group were given subcutaneous injection of Tβ4 for 4 consecutive weeks. Liver tissue was collected at the end of the experiment, and HE staining and Masson staining were used to observe histopathological changes; quantitative real-time PCR was used to measure the mRNA expression level of PDGF; TUNEL assay was used to assess the apoptosis of HSC. A one-way analysis of variance was used for comparison of continuous data between multiple groups, and the least significant difference t-test was used for further comparison between two groups. Results Compared with the model group, the middle- and high-dose Tβ4 treatment groups had varying degrees of alleviation of hepatic fibrosis. Quantitative real-time PCR showed that Tβ4 could significantly downregulate the mRNA expression level of PDGF in liver tissue, with a significant difference between the treatment groups (P>0.05), and there was no significant difference in the mRNA expression level of PDGF between the high-dose Tβ4 treatment group and the normal control group (P>0.05). TUNEL assay showed that the middle- and high-dose Tβ4 treatment groups had a significantly higher number of apoptotic HSCs than the model group. Conclusion Tβ4 may improve CCl4-induced hepatic fibrosis in mice by downregulating the expression of PDGF and promoting the apoptosis of HSC, suggesting that it has a potential application value in the treatment of hepatic fibrosis. -
Key words:
- Hepatic Fibrosis /
- Thymosin Beta 4 /
- Mice, Inbred C57BL
-
-
[1] FAN XM, YU FJ. Frontier of liver fibrosis research[M]. Shanghai: Fudan University Press, 2015.樊晓明, 俞富军. 肝纤维化研究前沿[M]. 上海: 复旦大学出版社, 2015. [2] SI J, TANG L, LI CC, et al. Pathogenesis of liver fibrosis and progress of research on its related therapeutic drugs[J]. Prog Pharm Sci, 2021, 45( 3): 205- 211.司晶, 唐露, 李臣诚, 等. 肝纤维化的发病机制及其治疗药物研究进展[J]. 药学进展, 2021, 45( 3): 205- 211. [3] LIAO ZH, XIE ZY. Research progress in molecular mechanism of hepatic fibrosis and related therapeutic targets[J]. J Jilin Univ(Med Edit), 2024, 50( 5): 1450- 1456. DOI: 10.13481/j.1671-587X.20240532.廖昭辉, 谢正元. 肝纤维化发病的分子机制及其相关治疗靶点的研究进展[J]. 吉林大学学报(医学版), 2024, 50( 5): 1450- 1456. DOI: 10.13481/j.1671-587X.20240532. [4] FAN ML, YING MF, ZHAO R, et al. Research progress on the role of TGF-β signaling pathway in fibrotic diseases[J]. Med J Chin PLA, 2020, 45( 11): 1171- 1177. DOI: 10.11855/j.issn.0577-7402.2020.11.11.范美玲, 应苗法, 赵蕊, 等. TGF-β信号通路在纤维化疾病中的作用研究进展[J]. 解放军医学杂志, 2020, 45( 11): 1171- 1177. DOI: 10.11855/j.issn.0577-7402.2020.11.11. [5] SUN SH. Study on the mechanism of modified Chaihu Danggui powder in treating hepatic fibrosis based on the PI3K/AKT/Bcl-2 signaling pathway[D]. Nanning: Guangxi University of Chinese Medicine, 2024. DOI: 10.27879/d.cnki.ggxzy.2024.000003.孙素红. 基于PI3K/AKT/Bcl-2信号通路探讨加味柴胡当归汤治疗肝纤维化的机制研究[D]. 南宁: 广西中医药大学, 2024. DOI: 10.27879/d.cnki.ggxzy.2024.000003. [6] ZHU ZX, LIAO YF, MOU QJ, et al. Thymosin β4 regulates tissue inflammatory response in mouse nonalcoholic fatty liver disease by promoting macrophage M2-type polarization[J]. J Inflamm Res, 2025, 18: 5791- 5809. DOI: 10.2147/JIR.S492814. [7] XING Y, YE YM, ZUO HY, et al. Progress on the function and application of thymosin β4[J]. Front Endocrinol(Lausanne), 2021, 12: 767785. DOI: 10.3389/fendo.2021.767785. [8] MASON WJ, VASILOPOULOU E. The pathophysiological role of thymosin β4 in the kidney glomerulus[J]. Int J Mol Sci, 2023, 24( 9): 7684. DOI: 10.3390/ijms24097684. [9] BJØRKLUND G, DADAR M, AASETH J, et al. Thymosin β4: A multi-faceted tissue repair stimulating protein in heart injury[J]. Curr Med Chem, 2020, 27( 37): 6294- 6305. DOI: 10.2174/0929867326666190716125456. [10] YANG L, FU WL, ZHU Y, et al. Tβ4 suppresses lincRNA-p21-mediated hepatic apoptosis and fibrosis by inhibiting PI3K-AKT-NF-κB pathway[J]. Gene, 2020, 758: 144946. DOI: 10.1016/j.gene.2020.144946. [11] WANG W, JIA WN, ZHANG CP. The role of Tβ4-POP-Ac-SDKP axis in organ fibrosis[J]. Int J Mol Sci, 2022, 23( 21): 13282. DOI: 10.3390/ijms232113282. [12] KLEINMAN HK, KULIK V, GOLDSTEIN AL. Thymosin β4 and the anti-fibrotic switch[J]. Int Immunopharmacol, 2023, 115: 109628. DOI: 10.1016/j.intimp.2022.109628. [13] YU R, LI SM, CHEN L, et al. Inhaled exogenous thymosin beta 4 suppresses bleomycin-induced pulmonary fibrosis in mice via TGF-β1 signalling pathway[J]. J Pharm Pharmacol, 2025, 77( 4): 582- 592. DOI: 10.1093/jpp/rgae143. [14] HOU YC, ZHAO HQ, DONG CE, et al. Research advances in immune checkpoint inhibitor-related cholangitis[J]. J Clin Hepatol, 2023, 39( 2): 463- 468. DOI: 10.3969/j.issn.1001-5256.2023.02.034.侯昱丞, 赵洪强, 董草儿, 等. 免疫检查点抑制剂相关胆管炎的研究进展[J]. 临床肝胆病杂志, 2023, 39( 2): 463- 468. DOI: 10.3969/j.issn.1001-5256.2023.02.034. [15] ZHU LL, CHENG ML, LIU YM, et al. Thymosin-β4 inhibits proliferation and induces apoptosis of hepatic stellate cells through PI3K/AKT pathway[J]. Oncotarget, 2017, 8( 40): 68847- 68853. DOI: 10.18632/oncotarget.18748. [16] YANG JJ. Molecular mechanism of DNMT3A mediated LncRNA ANRIL methylation promoted liver fibrosis[D]. Hefei: Anhui Medical University, 2021. DOI: 10.26921/d.cnki.ganyu.2021.000035.杨晶晶. DNMT3A介导LncRNA ANRIL甲基化促进肝纤维化的分子机制[D]. 合肥: 安徽医科大学, 2021. DOI: 10.26921/d.cnki.ganyu.2021.000035. [17] ZHANG WF. Exploring the role and mechanism of mitochondrial autophagy in the inhibition of hepatic fibrosis by soft liver fibrotic particles based on miR-135a/FOXO1/PINK1 pathway[D]. Nanning: Guangxi University of Chinese Medicine, 2024. DOI: 10.27879/d.cnki.ggxzy.2024.000009.张文富. 基于miR-135a/FOXO1/PINK1通路探讨线粒体自噬在柔肝化纤颗粒抑制肝纤维化中的作用及机制[D]. 南宁: 广西中医药大学, 2024. DOI: 10.27879/d.cnki.ggxzy.2024.000009. [18] HU PY. Experimental study of curcumin combined withdihydromyricetin nanoparticle co-delivery system forsynergistic treatment of liver fibrosis[D]. Hefei: Anhui Medical University, 2025. DOI: 10.26921/d.cnki.ganyu.2025.000015.胡潘宜. 姜黄素联合二氢杨梅素纳米颗粒共递送系统协同治疗肝纤维化的实验研究[D]. 合肥: 安徽医科大学, 2025. DOI: 10.26921/d.cnki.ganyu.2025.000015. [19] ZHAO TT, LI JF, ZHANG LT. Progress in the potential therapeutic mechanism of mesenchymal stem cell-derived exosomes for liver fibrosis[J]. Chin J Clin Pharmacol Ther, 2024, 29( 4): 475- 480. DOI: 10.12092/j.issn.1009-2501.2024.04.017.赵婷婷, 李俊峰, 张立婷. 间充质干细胞源性外泌体对肝纤维化潜在治疗机制的研究进展[J]. 中国临床药理学与治疗学, 2024, 29( 4): 475- 480. DOI: 10.12092/j.issn.1009-2501.2024.04.017. [20] ZOU JJ. The study on mechanism of Shenkang injection in treating renal fibrosis from the metabolomics and regulation of E3 ubiquitin ligase Smurfs on TGF-β/Smads signaling transduction[D]. Chengdu: Chengdu University of Traditional Chinese Medicine, 2021. DOI: 10.26988/d.cnki.gcdzu.2021.000374.邹俊驹. 从代谢组学及E3泛素连接酶Smurfs对TGF-β/Smads信号传导的调节探讨肾康注射液治疗肾纤维化的机制[D]. 成都: 成都中医药大学, 2021. DOI: 10.26988/d.cnki.gcdzu.2021.000374. [21] WANG F, HE YJ, YAO NJ, et al. Thymosin β4 protects against cardiac damage and subsequent cardiac fibrosis in mice with myocardial infarction[J]. Cardiovasc Ther, 2022, 2022: 1308651. DOI: 10.1155/2022/1308651. [22] YU H, WANG B, LI ZH, et al. Tβ4-exosome-loaded hemostatic and antibacterial hydrogel to improve vascular regeneration and modulate macrophage polarization for diabetic wound treatment[J]. Mater Today Bio, 2025, 31: 101585. DOI: 10.1016/j.mtbio.2025.101585. [23] WANG YL, YU SN, SHEN HR, et al. Thymosin β4 released from functionalized self-assembling peptide activates epicardium and enhances repair of infarcted myocardium[J]. Theranostics, 2021, 11( 9): 4262- 4280. DOI: 10.7150/thno.52309. [24] LI QX, ZHANG QJ, KIM YR, et al. Deficiency of endothelial sirtuin1 in mice stimulates skeletal muscle insulin sensitivity by modifying the secretome[J]. Nat Commun, 2023, 14( 1): 5595. DOI: 10.1038/s41467-023-41351-1. -



 PDF下载 ( 32076 KB)
PDF下载 ( 32076 KB)

 下载:
下载: