Wnt5a基因敲低的M1型骨髓源性巨噬细胞对肝硬化大鼠模型的影响及其作用机制
DOI: 10.12449/JCH260317
Effect and mechanism of Wnt5a knockdown on the efficacy of M1 bone marrow-derived macrophage in treatment of liver cirrhosis
-
摘要:
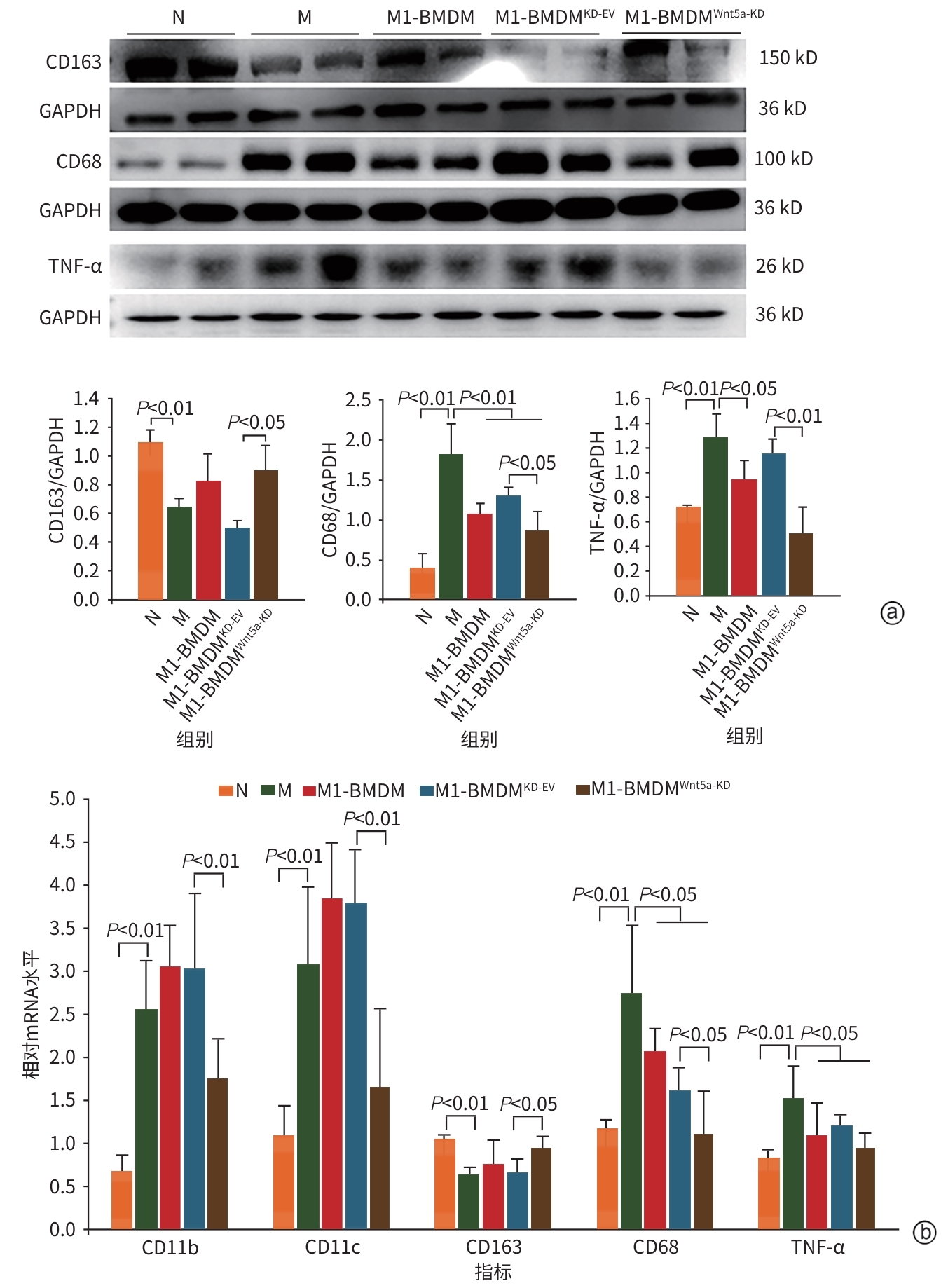
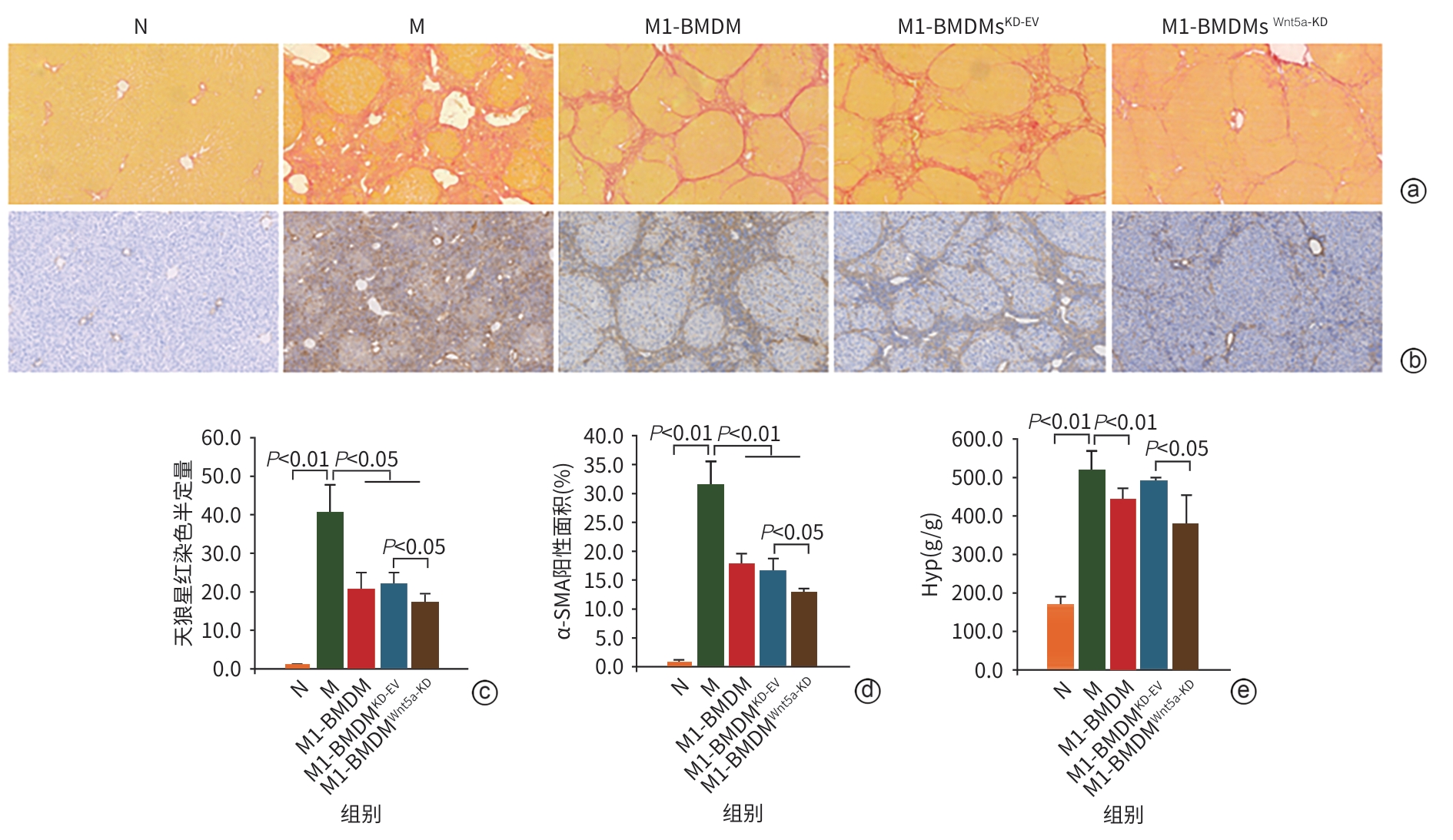
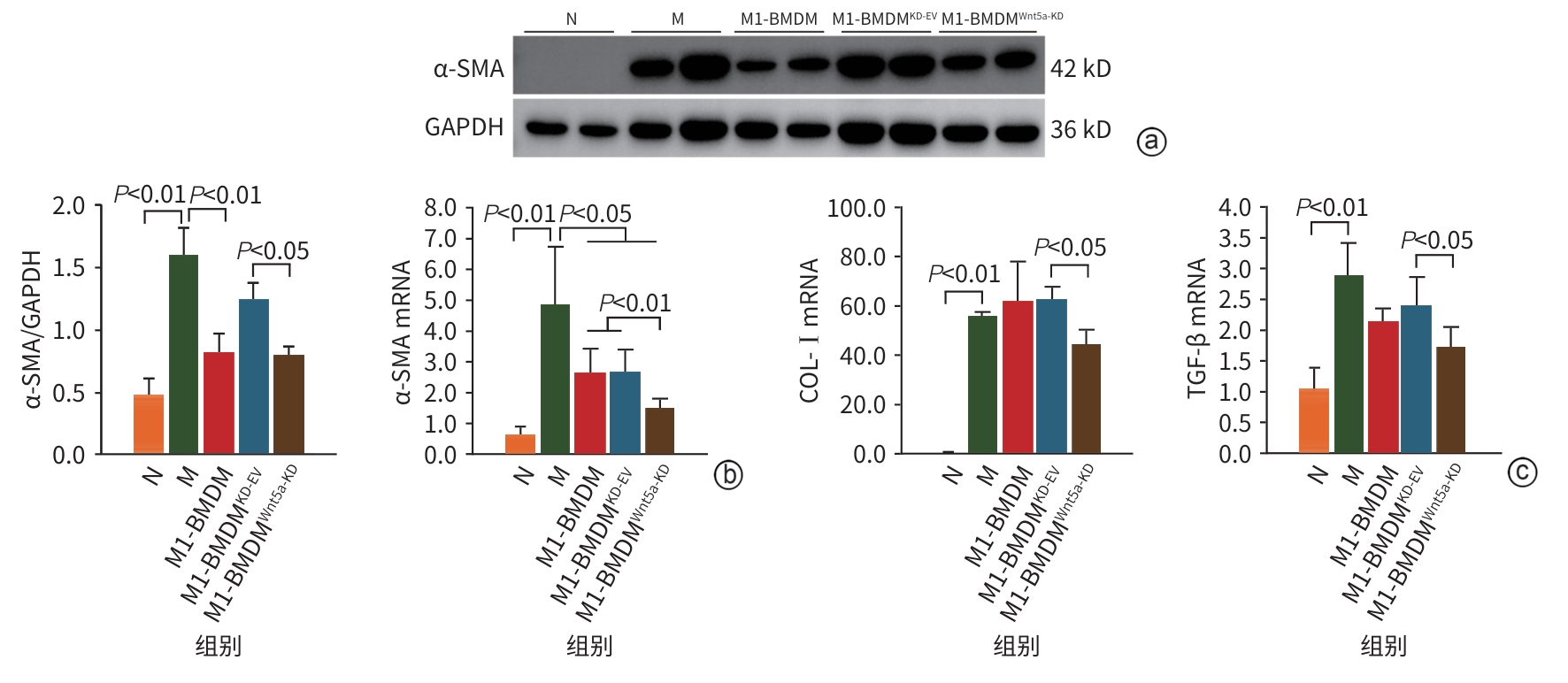
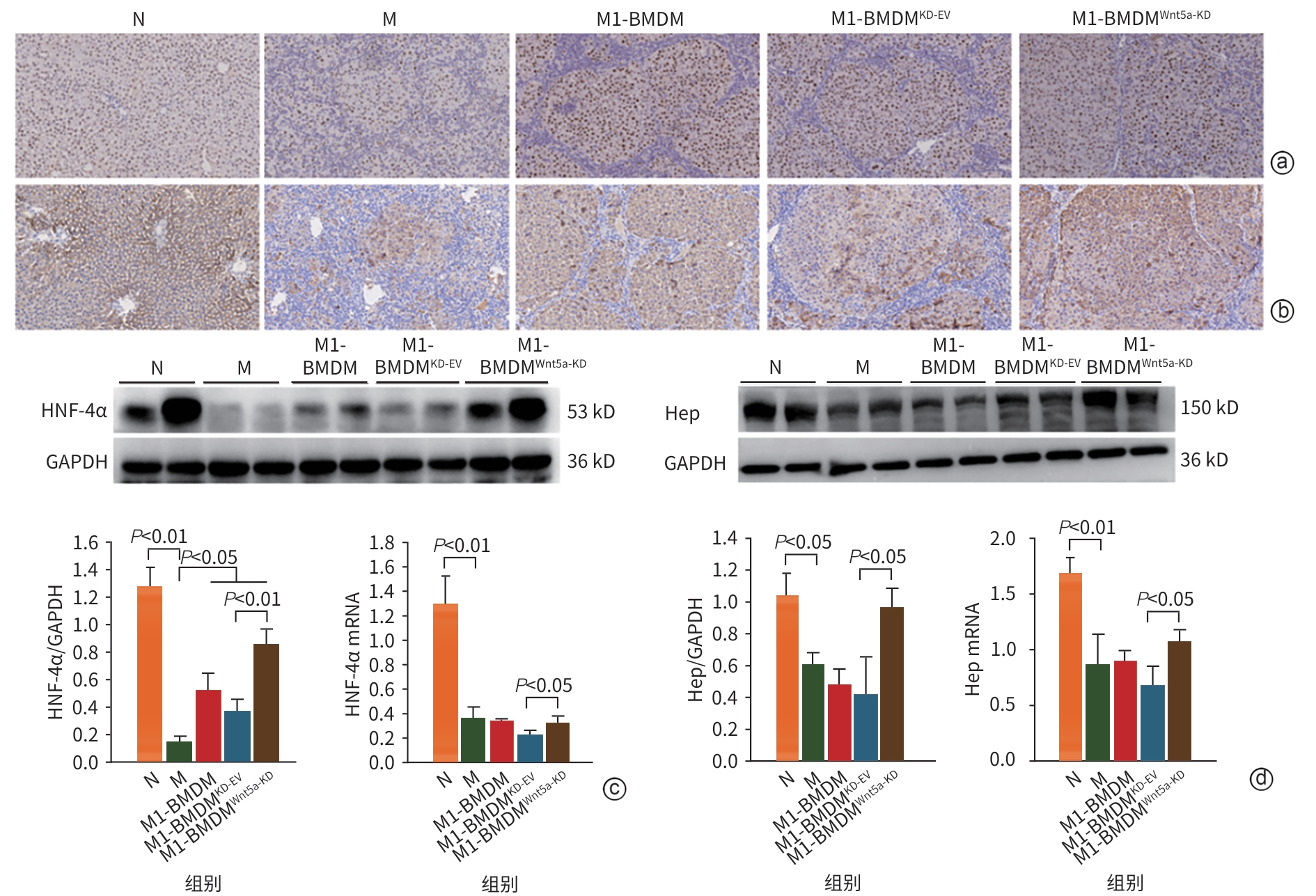
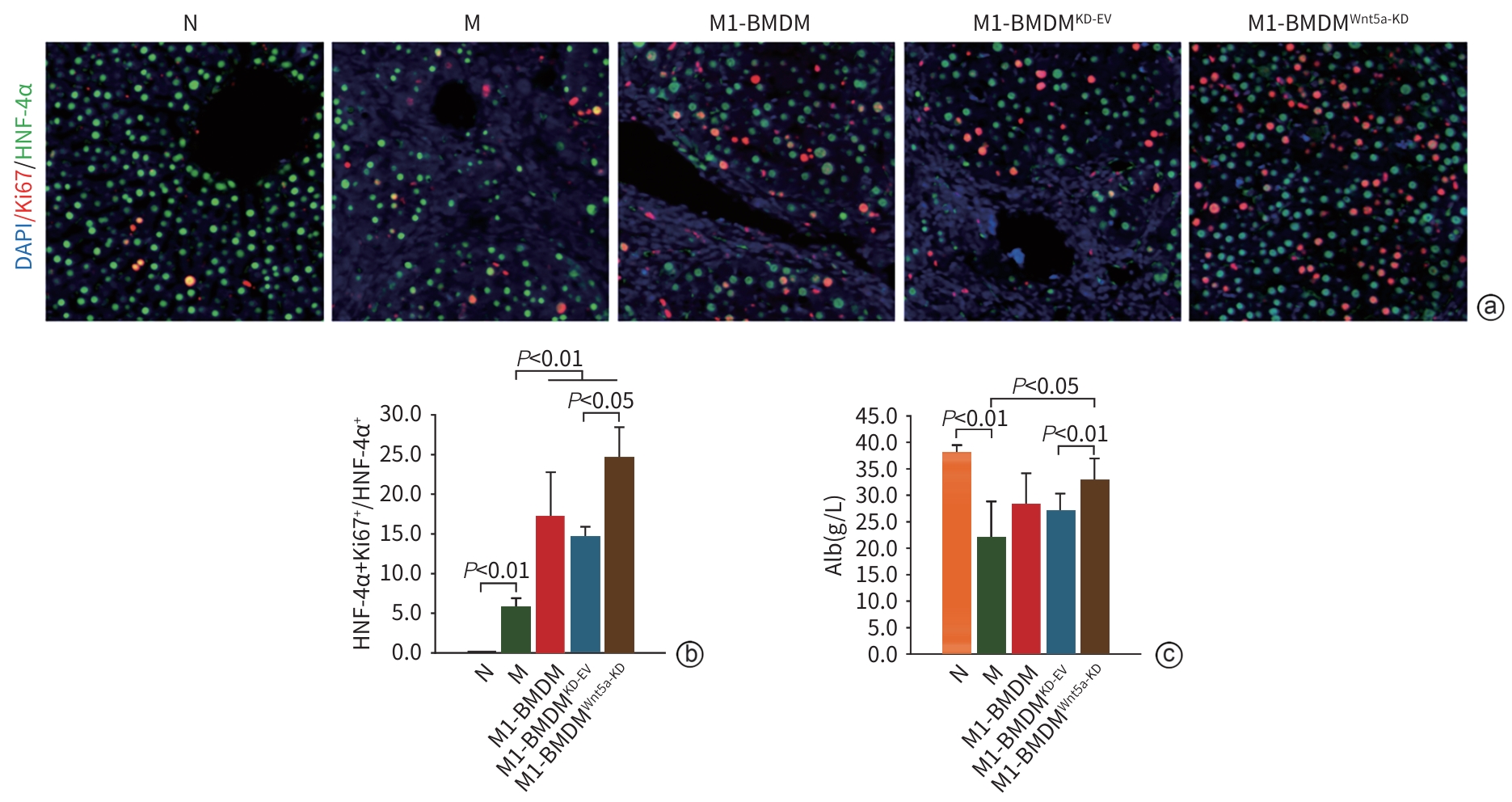
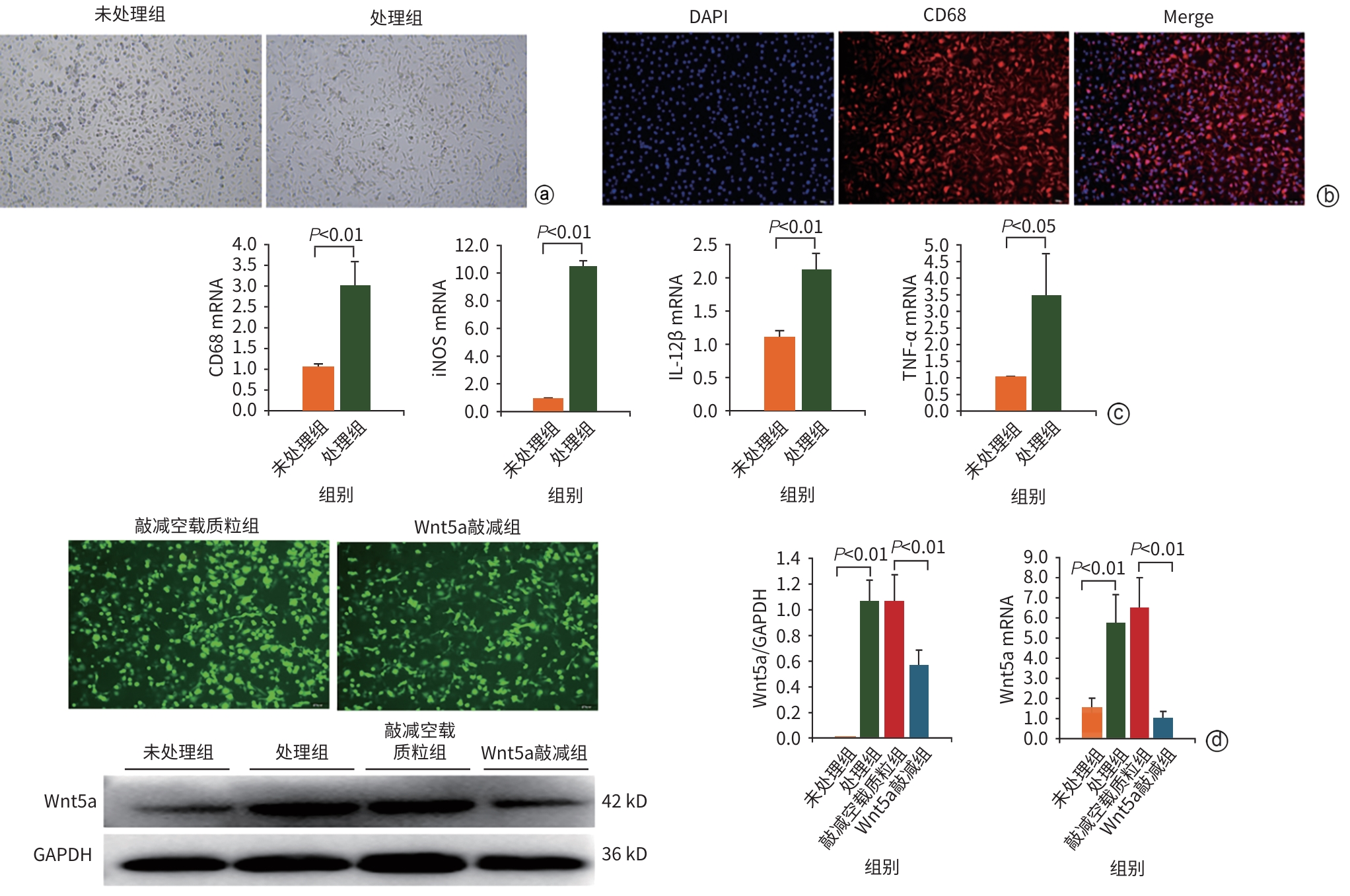
目的 本研究通过观察Wnt5a基因敲低的M1型骨髓源性巨噬细胞(BMDM)对肝硬化大鼠模型肝纤维化与再生的影响,旨在揭示其相对于未修饰M1-BMDM的功能增益效应。 方法 分离大鼠原代BMDM并极化为M1表型,构建Wnt5a敲低的M1-BMDM。建立四氯化碳/二乙酰氨基芴(CCl4/2-AAF)诱导的肝硬化大鼠模型,于第8周末随机分为模型组(n=6)、M1-BMDM组(n=6)、M1-BMDM Wnt5a基因敲减空载质粒组(M1-BMDMKD-EV组)(n=6)、M1-BMDM Wnt5a基因敲减组(M1-BMDMWnt5a-KD组)(n=6)。第9周首日,各组大鼠予以相应的细胞经尾静脉一次性注射,同时腹腔注射C-C趋化因子受体2型抑制剂。此外,设立未经任何处理的正常对照组(n=6)。于12周末取材,检测肝组织病理、血清肝功能、活化肝星状细胞以及成熟肝细胞标志物的表达水平。计量资料多组间比较采用单因素方差分析,进一步两两比较采用LSD-t检验。 结果 与模型组大鼠比较,各细胞治疗组大鼠肝脏炎症反应明显减轻,血清丙氨酸氨基转移酶(ALT)、天冬氨酸氨基转移酶(AST)活性均显著降低(P值均<0.01),且M1-BMDMWnt5a-KD组血清AST水平显著低于M1-BMDM组(P<0.05)。免疫组织化学染色半定量分析显示,与模型组比较,各细胞治疗组CD68阳性面积百分比均明显减少(P值均<0.05);与M1-BMDMKD-EV相比,M1-BMDMWnt5a-KD组CD68阳性面积百分比显著下降、CD163的阳性面积百分比显著升高(P值均<0.05)。与模型组比较,各细胞治疗组的CD68及肿瘤坏死因子α的mRNA表达显著降低(P值均<0.05),CD68的蛋白表达显著降低(P值均<0.01);与M1-BMDMKD-EV组比较,M1-BMDMWnt5a-KD组CD163蛋白及mRNA表达均显著升高(P值均<0.05),CD68蛋白及mRNA表达显著降低(P值均<0.05),肿瘤坏死因子α的蛋白表达显著降低(P<0.01)。天狼星红胶原染色和α-平滑肌肌动蛋白(α-SMA)的免疫组织化学染色结果显示,与模型组比较,各细胞治疗组肝脏胶原沉积和α-SMA阳性面积明显减轻,尤以M1-BMDMWnt5a-KD组最为显著;且M1-BMDMWnt5a-KD组天狼星红阳性面积、α-SMA阳性面积和肝组织羟脯氨酸(Hyp)含量亦显著低于M1-BMDMKD-EV组(P值均<0.05)。与M1-BMDMKD-EV组相比,M1-BMDMWnt5a-KD组α-SMA蛋白和mRNA以及Ⅰ型胶原蛋白和转化生长因子β的mRNA表达水平显著降低(P值均<0.05)。与模型组比较,各细胞治疗组肝组织肝细胞核因子4α(HNF-4α)蛋白表达显著升高(P值均<0.05),且M1-BMDMWnt5a-KD组HNF-4α和肝细胞特异性抗原的蛋白与mRNA的表达均显著高于M1-BMDMKD-EV组(P值均<0.05)。M1-BMDMWnt5a-KD组血清白蛋白含量亦显著高于M1-BMDMKD-EV组(P<0.01)。免疫荧光共染检测技术显示,与模型组相比,各细胞治疗组HNF-4α/Ki67共染细胞数量显著增多(P值均<0.01),且M1-BMDMWnt5a-KD组HNF-4α/Ki67共染细胞数量显著高于M1-BMDMKD-EV组(P<0.05)。 结论 抑制Wnt5a表达可提高M1-BMDM治疗CCl4/2-AAF诱导肝硬化大鼠模型的效果,为基因修饰提高M1-BMDM抗肝硬化作用提供了新思路。 Abstract:Objective To observe the effect of M1 bone marrow-derived macrophages (M1-BMDM) with Wnt5a knockdown on liver fibrosis and regeneration in a rat model of liver cirrhosis, and to investigate its gain-of-function effect compared with unmodified M1-BMDM. Methods Primary bone marrow-derived macrophages were isolated from rats and were polarized to M1 phenotype to construct M1-BMDMWnt5a-KD cells. A rat model of liver cirrhosis induced by CCl4/2-AAF was established, and at the end of week 8, rats were randomly divided into model group, M1-BMDM group, M1-BMDM Wnt5a-knockdown empty vector group (M1-BMDMKD-EV group), and M1-BMDM Wnt5a-knockdown group (M1-BMDMWnt5a-KD group), with 6 rats in each group. On the first day of week 9, the rats in each group were given a single injection of the corresponding cells via the caudal vein, along with an intraperitoneal injection of a CCR2 inhibitor. Six rats without any treatment were used as normal control group. Samples were collected at the end of week 12 to assess liver histopathology, serum liver function parameters, hepatic stellate cell activation, and the expression levels of mature hepatocyte markers. A one-way analysis of variance was used for comparison of continuous data between multiple groups, and the least significant difference t-test was used for further comparison between two groups. Results Compared with the model group, all cell treatment groups had significant alleviation of liver inflammatory response and significant reductions in the activities of alanine aminotransferase and aspartate aminotransferase (AST) in serum (all P<0.01), and the M1-BMDMWnt5a-KD group had a significantly lower serum level of AST than the M1-BMDM group (P<0.05). The semi-quantitative analysis based on immunohistochemical staining showed that compared with the model group, all cell treatment groups had a significant reduction in the percentage of CD68-positive area (all P<0.05), and compared with the M1-BMDMKD-EV group, the M1-BMDMWnt5a-KD group had a significant reduction in the percentage of CD68-positive area and a significant increase in the percentage of CD163-positive area (both P<0.05). Compared with the model group, all cell treatment groups had significant reductions in the mRNA expression levels of CD68 and tumor necrosis factor-α (all P<0.05) and the protein expression level of CD68 (all P<0.01); compared with the M1-BMDMKD-EV group, the M1-BMDMWnt5a-KD group had significant increases in the protein and mRNA expression levels of CD163 (both P<0.05), significant reductions in the protein and mRNA expression levels of CD68 (both P<0.05), and a significant reduction in the protein expression level of tumor necrosis factor-α (P<0.01). Sirius Red collagen staining and alpha-smooth muscle actin (α-SMA) immunohistochemical staining showed that compared with the model group, all cell treatment groups had significant alleviation of liver collagen deposition and α-SMA-positive area, with the most significant changes in the M1-BMDMWnt5a-KD group, and compared with the M1-BMDMKD-EV group, the M1-BMDMWnt5a-KD group had significantly smaller Sirius Red-positive area and α-SMA-positive area and a significantly lower content of hydroxyproline in liver tissue (all P<0.05). Compared with the M1-BMDMKD-EV group, the M1-BMDMWnt5a-KD group had significant reductions in the protein and mRNA expression levels of α-SMA and the mRNA expression level of COL-I and TGF-β (all P<0.05). Compared with the model group, all cell treatment groups had a significant increase in the protein expression level of HNF-4α in liver tissue (all P<0.05), and the M1-BMDMWnt5a-KD group had significantly higher protein and mRNA expression levels of HNF-4α and hepatocyte specific antigen than the M1-BMDMKD-EV group (both P<0.05). The M1-BMDMWnt5a-KD group had a significantly higher serum level of albumin than the M1-BMDMKD-EV group (P<0.01). Immunofluorescence co-staining showed that compared with the model group, all cell treatment groups had a significant increase in the number of cells stained positive for HNF and HNF-4α and Ki67 (all P<0.01), and the M1-BMDMWnt5a-KD group had a significantly higher number of such cells than the M1-BMDMKD-EV group (P<0.05). Conclusion Inhibition of Wnt5a expression enhances the therapeutic effect of M1-BMDM on rats with liver cirrhosis induced by CCl4/2-AAF, which provides new ideas for enhancing the anti-cirrhotic effect of M1-BMDM through genetic modification. -
-
[1] OAKLEY F. Interrogating mechanisms of liver fibrosis with omics[J]. Nat Rev Gastroenterol Hepatol, 2022, 19( 2): 89- 90. DOI: 10.1038/s41575-021-00567-6. [2] LIAO ZH, XIE ZY. Research progress in molecular mechanism of hepatic fibrosis and related therapeutic targets[J]. J Jilin Univ: Med Edit, 2024, 50( 5): 1450- 1456. DOI: 10.13481/j.1671-587X.20240532.廖昭辉, 谢正元. 肝纤维化发病的分子机制及其相关治疗靶点的研究进展[J]. 吉林大学学报(医学版), 2024, 50( 5): 1450- 1456. DOI: 10.13481/j.1671-587X.20240532. [3] LI L, CUI L, LIN P, et al. Kupffer-cell-derived IL-6 is repurposed for hepatocyte dedifferentiation via activating progenitor genes from injury-specific enhancers[J]. Cell Stem Cell, 2023, 30( 3): 283- 299.e9. DOI: 10.1016/j.stem.2023.01.009. [4] THOMAS JA, POPE C, WOJTACHA D, et al. Macrophage therapy for murine liver fibrosis recruits host effector cells improving fibrosis, regeneration, and function[J]. Hepatology, 2011, 53( 6): 2003- 2015. DOI: 10.1002/hep.24315. [5] MA PF, GAO CC, YI J, et al. Cytotherapy with M1-polarized macrophages ameliorates liver fibrosis by modulating immune microenvironment in mice[J]. J Hepatol, 2017, 67( 4): 770- 779. DOI: 10.1016/j.jhep.2017.05.022. [6] ZONG MY, JIAN X, WANG DY, et al. Effect of Yiguan Decoction on the efficacy of M1 bone marrow-derived macrophages in treatment of liver cirrhosis rats and its mechanism[J]. J Clin Hepatol, 2024, 40( 8): 1612- 1619. DOI: 10.12449/JCH240817.宗梦瑶, 简迅, 王丹阳, 等. 一贯煎对M1型骨髓巨噬细胞治疗肝硬化大鼠模型效果的影响及其机制分析[J]. 临床肝胆病杂志, 2024, 40( 8): 1612- 1619. DOI: 10.12449/JCH240817. [7] CAO MZ, CHAN RWS, CHENG FHC, et al. Myometrial cells stimulate self-renewal of endometrial mesenchymal stem-like cells through WNT5A/β-catenin signaling[J]. Stem Cells, 2019, 37( 11): 1455- 1466. DOI: 10.1002/stem.3070. [8] LIN QQ, WU LM, CHATLA S, et al. Hematopoietic stem cell regeneration through paracrine regulation of the Wnt5a/Prox1 signaling axis[J]. J Clin Invest, 2022, 132( 12): e155914. DOI: 10.1172/JCI155914. [9] ZHAO CG, QIN J, LI J, et al. LINGO-1 regulates Wnt5a signaling during neural stem and progenitor cell differentiation by modulating miR-15b-3p levels[J]. Stem Cell Res Ther, 2021, 12( 1): 372. DOI: 10.1186/s13287-021-02452-0. [10] TIAN Y, CHEN JC, HUANG WS, et al. Myeloid-derived Wnts play an indispensible role in macrophage and fibroblast activation and kidney fibrosis[J]. Int J Biol Sci, 2024, 20( 6): 2310- 2322. DOI: 10.7150/ijbs.94166. [11] JIAN X, WANG DY, XU YN, et al. Effect of polarized bone marrow-derived macrophage transplantation on the progression of CCl4-induced liver fibrosis in rats[J]. J Clin Hepatol, 2021, 37( 12): 2830- 2837. DOI: 10.3969/j.issn.1001-5256.2021.12.020.简迅, 王丹阳, 许燕楠, 等. 极化骨髓巨噬细胞移植对CCl4诱导的肝纤维化大鼠模型的影响[J]. 临床肝胆病杂志, 2021, 37( 12): 2830- 2837. DOI: 10.3969/j.issn.1001-5256.2021.12.020. [12] HAAG SM, MURTHY A. Murine monocyte and macrophage culture[J]. Bio Protoc, 2021, 11( 6): e3928. DOI: 10.21769/BioProtoc.3928. [13] JAMALL IS, FINELLI VN, QUE HEE SS. A simple method to determine nanogram levels of 4-hydroxyproline in biological tissues[J]. Anal Biochem, 1981, 112( 1): 70- 75. DOI: 10.1016/0003-2697(81)90261-x. [14] GUILLOT A, TACKE F. Spatial dimension of macrophage heterogeneity in liver diseases[J]. eGastroenterology, 2023, 1( 1): e000003. DOI: 10.1136/egastro-2023-000003. [15] FLORES MOLINA M, ABDELNABI MN, MAZOUZ S, et al. Distinct spatial distribution and roles of Kupffer cells and monocyte-derived macrophages in mouse acute liver injury[J]. Front Immunol, 2022, 13: 994480. DOI: 10.3389/fimmu.2022.994480. [16] NA YR, KIM SW, SEOK SH. A new era of macrophage-based cell therapy[J]. Exp Mol Med, 2023, 55( 9): 1945- 1954. DOI: 10.1038/s12276-023-01068-z. [17] CHEN T, CHEN Y. Research progress in mechanism of fibrosis regulated by macrophage polarization[J]. J Jilin Univ: Med Edit, 2024, 50( 5): 1465- 1473. DOI: 10.13481/j.1671-587X.20240534.陈潭, 陈艳. 巨噬细胞极化调控纤维化机制的研究进展[J]. 吉林大学学报(医学版), 2024, 50( 5): 1465- 1473. DOI: 10.13481/j.1671-587X.20240534. [18] SHAO Y, ZHENG QQ, WANG W, et al. Biological functions of macrophage-derived Wnt5a, and its roles in human diseases[J]. Oncotarget, 2016, 7( 41): 67674- 67684. DOI: 10.18632/oncotarget.11874. [19] MENCK K, KLEMM F, GROSS JC, et al. Induction and transport of Wnt 5a during macrophage-induced malignant invasion is mediated by two types of extracellular vesicles[J]. Oncotarget, 2013, 4( 11): 2057- 2066. DOI: 10.18632/oncotarget.1336. [20] LIU Q, SONG JL, PAN Y, et al. Wnt5a/CaMKII/ERK/CCL2 axis is required for tumor-associated macrophages to promote colorectal cancer progression[J]. Int J Biol Sci, 2020, 16( 6): 1023- 1034. DOI: 10.7150/ijbs.40535. [21] MA X, HUANG TD, CHEN XZ, et al. Molecular mechanisms in liver repair and regeneration: From physiology to therapeutics[J]. Signal Transduct Target Ther, 2025, 10( 1): 63. DOI: 10.1038/s41392-024-02104-8. [22] HUCK I, GUNEWARDENA S, ESPANOL-SUNER R, et al. Hepatocyte nuclear factor 4 alpha activation is essential for termination of liver regeneration in mice[J]. Hepatology, 2019, 70( 2): 666- 681. DOI: 10.1002/hep.30405. [23] SONG ZM, LIN SR, WU XW, et al. Hepatitis B virus-related intrahepatic cholangiocarcinoma originates from hepatocytes[J]. Hepatol Int, 2023, 17( 5): 1300- 1317. DOI: 10.1007/s12072-023-10556-3. [24] SCHOLZEN T, GERDES J. The Ki-67 protein: From the known and the unknown[J]. J Cell Physiol, 2000, 182( 3): 311- 322. DOI: 3.0.CO;2-9">10.1002/(SICI)1097-4652(200003)182: 3<311:: AID-JCP1>3.0.CO;2-9. [25] SUN DQ, ZHOU XL, WU T, et al. Quercetin promotes the M1-to-M2 macrophage phenotypic switch during liver fibrosis treatment by modulating the JAK2/STAT3 signaling pathway[J]. Recent Pat Anticancer Drug Discov, 2026, 21( 1): 31- 46. DOI: 10.2174/0115748928318948240920044716. [26] MORONI F, DWYER BJ, GRAHAM C, et al. Safety profile of autologous macrophage therapy for liver cirrhosis[J]. Nat Med, 2019, 25( 10): 1560- 1565. DOI: 10.1038/s41591-019-0599-8. [27] BRENNAN PN, MACMILLAN M, MANSHIP T, et al. Autologous macrophage therapy for liver cirrhosis: A phase 2 open-label randomized controlled trial[J]. Nat Med, 2025, 31( 3): 979- 987. DOI: 10.1038/s41591-024-03406-8. -



 PDF下载 ( 50530 KB)
PDF下载 ( 50530 KB)

 下载:
下载: