氩氦刀冷冻消融联合靶向药物和程序性死亡受体1单抗治疗年龄>60岁不可切除肝细胞癌患者的效果及安全性分析
DOI: 10.12449/JCH260318
Safety and efficacy of argon-helium cryoablation combined with targeted therapy and anti-programmed death-1 monoclonal antibody in treatment of patients with unresectable hepatocellular carcinoma aged 60 years or older
-
摘要:
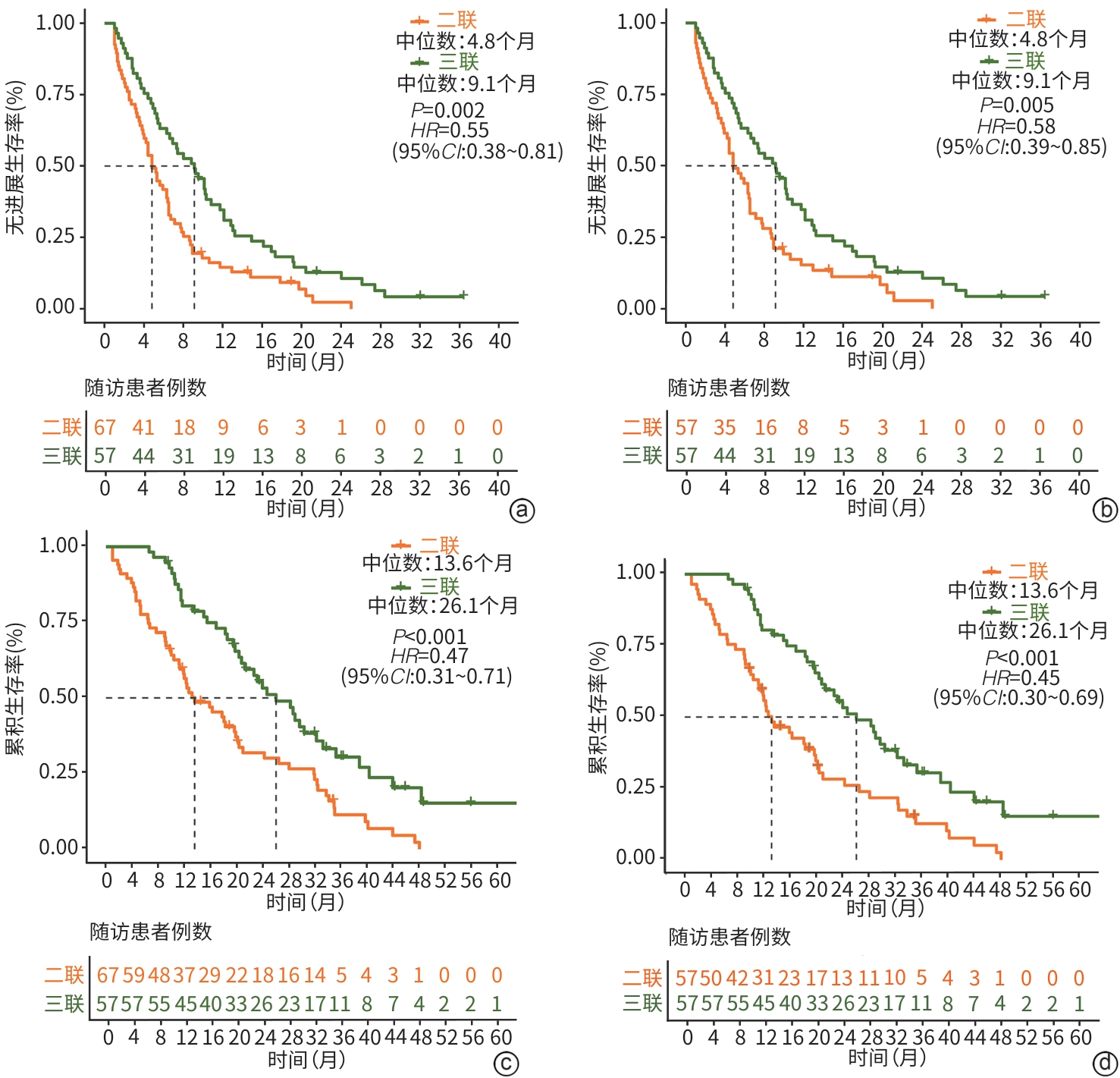
目的 评估程序性死亡受体1(PD-1)单抗能否提高氩氦刀冷冻消融联合靶向药物治疗年龄>60岁不可切除肝细胞癌(uHCC)患者的疗效及安全性。 方法 回顾性分析2013年1月—2024年9月中国人民解放军总医院第五医学中心收治的124例年龄>60岁中晚期uHCC患者临床资料。采用倾向性评分匹配后,氩氦刀冷冻消融联合靶向药物治疗(二联组)和氩氦刀冷冻消融联合靶向药物及PD-1单抗治疗(三联组)各57例。疗效评价指标包括客观缓解率(ORR)、疾病控制率(DCR)、无进展生存期(PFS)、总生存期(OS)及不良事件发生率。计量资料两组间比较采用Mann-Whitney U检验;计数资料两组间比较采用χ2检验或Fisher精确检验。采用Kaplan-Meier法绘制生存曲线,并运用Log-rank检验比较组间差异。Cox比例风险回归模型分析生存预后的影响因素。 结果 三联组患者的ORR显著高于二联组(59.6% vs 29.8%,χ2=9.083,P=0.003),但DCR未见显著差异(87.7% vs 77.2%,χ2=1.516,P=0.218);三联组患者中位PFS(9.1个月vs 4.8个月,χ2=7.813,P=0.005)及中位OS(26.1个月vs 13.6个月,χ2=14.199,P<0.001)均显著长于二联组。多因素Cox回归分析显示,三联治疗方案是PFS(风险比=0.52,95%置信区间:0.35~0.78,P=0.001)和OS(风险比=0.32,95%置信区间:0.20~0.51,P<0.001)的独立影响因素。两组治疗相关不良事件发生率差异均无统计学意义(P值均>0.05)。 结论 氩氦刀冷冻消融联合靶向药物及PD-1单抗三联治疗可显著改善年龄>60岁uHCC患者的生存获益,且安全性可控。 Abstract:Objective To investigate whether anti-programmed death-1 (PD-1) monoclonal antibody can enhance the efficacy and safety of argon-helium cryoablation combined with targeted therapy in patients with unresectable hepatocellular carcinoma (uHCC) aged 60 years or older. Methods A retrospective analysis was performed for the clinical data of 124 patients with advanced uHCC aged 60 years or older who were treated at The Fifth Medical Center of Chinese PLA General Hospital from January 2013 to September 2024. After propensity score matching, 57 patients received cryoablation combined with targeted therapy (double combination group), while 57 received cryoablation combined with targeted therapy and anti-PD-1 monoclonal antibody (triple combination group). The indicators for efficacy assessment included objective response rate (ORR), disease control rate (DCR), progression-free survival (PFS), overall survival (OS), and the incidence rate of adverse events. The Mann-Whitney U test was used for comparison of continuous data between two groups, and the chi-square test or the Fisher’s exact test was used for comparison of categorical data between two groups. The Kaplan-Meier method was used to plot survival curves, and the Log-rank test was used for comparison between groups. A Cox proportional-hazards regression model analysis was used to investigate the influencing factors for survival prognosis. Results The triple combination group had a significantly higher ORR than the double combination group (59.6% vs 29.8%, χ2=9.083, P=0.003), while there was no significant difference in DCR between the two groups (87.7% vs 77.2%, χ2=1.516, P=0.218), and compared with the double combination group, the triple combination group had significantly longer median PFS (9.1 months vs 4.8 months, χ2=7.813, P=0.005) and median OS (26.1 months vs 13.6 months, χ2=14.199, P<0.001). The multivariate Cox proportional-hazards regression model analysis showed that triple combination treatment was an independent influencing factor for PFS (hazard ratio [HR]=0.52, 95% confidence interval [CI]: 0.35 — 0.78, P=0.001) and OS (HR=0.32, 95%CI: 0.20 — 0.51, P<0.001). There was no significant difference in the incidence rate of adverse events between the two groups (P>0.05). Conclusion Triple combination treatment with argon-helium cryoablation, targeted therapy, and anti-PD-1 monoclonal antibody can significantly improve survival benefits in uHCC patients aged 60 years or older, with a controllable safety profile. -
表 1 PSM前后基线特征
Table 1. Clinical features before and after PSM
指标 PSM前 PSM后 二联组
(n=67)三联组
(n=57)P值 SMD 二联组
(n=57)三联组
(n=57)P值 SMD 年龄[例(%)] 0.525 0.152 0.832 0.080 60~70岁 46(68.7) 43(75.4) 41(71.9) 43(75.4) ≥70岁 21(31.3) 14(24.6) 16(28.1) 14(24.6) 男[例(%)] 52(77.6) 44(77.2) >0.05 0.010 43(75.4) 44(77.2) >0.05 0.041 BMI(kg/m2) 24.0(22.2~26.4) 23.4(21.7~25.6) 0.342 0.156 23.6(21.5~26.6) 23.4(21.7~25.6) 0.575 0.098 糖尿病[例(%)] 8(11.9) 7(12.3) >0.05 0.010 7(12.3) 7(12.3) >0.05 <0.001 病因[例(%)] 0.025 0.479 >0.05 0.086 病毒性肝炎 55(82.1) 55(96.5) 54(94.7) 55(96.5) 其他 12(17.9) 2(3.5) 3(5.3) 2(3.5) TBil(μmol/L) 17.4(11.9~24.5) 15.0(11.9~20.9) 0.492 0.187 17.4(11.5~21.7) 15.0(11.9~20.9) 0.624 0.161 ALT(U/L) 35.0(19.5~131.5) 39.0(22.0~111.0) 0.731 0.049 36.0(20.0~118.0) 39.0(22.0~111.0) 0.894 0.075 AST(U/L) 45.0(29.5~112.0) 37.0(28.0~67.0) 0.217 0.104 46.0(29.0~116.0) 37.0(28.0~67.0) 0.232 0.137 PLT(×109/L) 120.0(86.5~144.0) 114.0(79.0~140.0) 0.521 0.107 119.0(83.0~140.0) 114.0(79.0~140.0) 0.861 0.014 AFP≥400 ng/mL[例(%)] 23(34.3) 16(28.1) 0.580 0.135 21(36.8) 16(28.1) 0.424 0.188 Child-Pugh分级[例(%)] >0.05 0.053 >0.05 0.059 A级 61(91.0) 51(89.5) 52(91.2) 51(89.5) B级 6(9.0) 6(10.5) 5(8.8) 6(10.5) ECOG PS评分[例(%)] 0.599 0.128 0.453 0.176 0分 36(53.7) 27(47.4) 32(56.1) 27(47.4) 1分 31(46.3) 30(52.6) 25(43.9) 30(52.6) BCLC C期[例(%)] 46(68.7) 39(68.4) >0.05 0.005 39(68.4) 39(68.4) >0.05 <0.001 肿瘤多发[例(%)] 49(73.1) 43(75.4) 0.931 0.053 45(78.9) 43(75.4) 0.823 0.084 肿瘤最大直径(cm) 4.4(2.8~7.6) 4.7(3.0~7.6) 0.888 0.029 4.2(2.8~7.4) 4.7(3.0~7.6) 0.764 0.080 肝外转移[例(%)] 17(25.4) 15(26.3) >0.05 0.022 13(22.8) 15(26.3) 0.828 0.082 血管侵犯[例(%)] 37(55.2) 32(56.1) >0.05 0.018 31(54.4) 32(56.1) >0.05 0.035 既往局部治疗[例(%)] 18(26.9) 19(33.3) 0.557 0.141 17(29.8) 19(33.3) 0.840 0.076 注:PSM,倾向性评分匹配;BMI,体重指数;TBil,总胆红素;ALT,丙氨酸氨基转移酶;AST,天冬氨酸氨基转移酶;PLT,血小板计数;AFP,甲胎蛋白;Child-Pugh分级,蔡尔德-皮尤分级;ECOG PS,美国东部肿瘤协作组体力状况;BCLC,巴塞罗那临床肝癌。
表 2 mRECIST评估肿瘤消融效果
Table 2. Tumor ablation response evaluated with mRECIST
指标 PSM前 PSM后 二联组(n=67) 三联组(n=57) χ2值 P值 二联组(n=57) 三联组(n=57) χ2值 P值 CR 0(0.00) 0(0.00) 0(0.00) 0(0.00) PR 20(29.9) 34(59.6) 9.945 0.002 17(29.8) 34(59.6) 9.083 0.003 SD 34(50.7) 16(28.1) 5.673 0.017 27(47.4) 16(28.1) 3.734 0.053 PD 13(19.4) 7(12.3) 0.688 0.407 13(22.8) 7(12.3) 1.516 0.218 ORR 20(29.9) 34(59.6) 9.945 0.002 17(29.8) 34(59.6) 9.083 0.003 DCR 54(80.6) 50(87.7) 0.688 0.407 44(77.2) 50(87.7) 1.516 0.218 注:mRECIST,改良的实体瘤疗效评估标准;PSM,倾向性评分匹配;CR,完全反应;PR,部分反应;SD,疾病稳定;PD,疾病进展;ORR,客观缓解率;DCR,疾病控制率。
表 3 RECIST 1.1评估肿瘤消融效果
Table 3. Tumor ablation response evaluated with RECIST 1.1
指标 PSM前 PSM后 二联组(n=67) 三联组(n=57) χ2值 P值 二联组(n=57) 三联组(n=57) χ2值 P值 CR 0(0.00) 0(0.00) 0(0.00) 0(0.00) PR 19(28.4) 29(50.9) 5.668 0.017 17(29.8) 29(50.9) 4.410 0.036 SD 35(52.2) 21(36.8) 2.360 0.125 27(47.4) 21(36.8) 0.900 0.343 PD 13(19.4) 7(12.3) 0.688 0.407 13(22.8) 7(12.3) 1.516 0.218 ORR 19(28.4) 29(50.9) 5.668 0.017 17(29.8) 29(50.9) 4.410 0.036 DCR 54(80.6) 50(87.7) 0.688 0.407 44(77.2) 50(87.7) 1.516 0.218 注:RECIST,实体瘤疗效评估标准;PSM,倾向性评分匹配;CR,完全反应;PR,部分反应;SD,疾病稳定;PD,疾病进展;ORR,客观缓解率;DCR,疾病控制率。
表 4 后续治疗方案
Table 4. Subsequent treatment
指标 二联组
(n=57)三联组
(n=57)χ2值 P值 局部治疗[例(%)] 2.366 0.498 介入治疗 19(33.3) 18(31.6) 微波消融 3(5.3) 8(14.0) 射频消融 1(1.8) 1(1.8) 局部放射治疗 4(7.0) 6(10.5) 系统药物治疗[例(%)] 9.650 0.140 靶向药物 仑伐替尼 10(17.5) 3(5.3) 多纳非尼 2(3.5) 4(7.0) 瑞戈非尼 14(24.6) 12(21.1) 贝伐珠单抗 0(0.0) 3(5.3) 阿帕替尼 0(0.0) 1(1.8) 安罗替尼 0(0.0) 1(1.8) 免疫药物 3.080 0.214 信迪利单抗 0(0.0) 1(1.8) 卡瑞利珠单抗 0(0.0) 2(3.5) 表 5 影响患者OS的单因素及多因素Cox回归分析
Table 5. Univariate and multivariate Cox proportional hazards regression model analysis of influencing OS
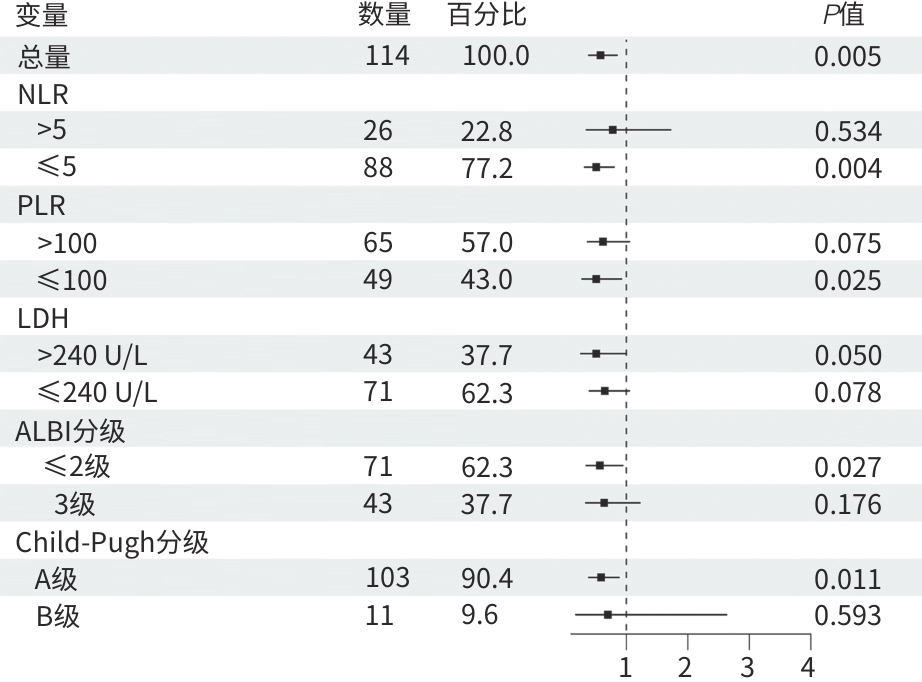
变量 单因素 多因素 HR(95%CI) P值 HR(95%CI) P值 治疗方式(三联vs二联) 0.45(0.30~0.69) <0.001 0.32(0.20~0.51) <0.001 年龄(≥70岁 vs <70岁) 1.17(0.74~1.86) 0.506 性别(男vs女) 0.71(0.44~1.15) 0.164 BMI(kg/m2) 1.02(0.95~1.09) 0.603 病因学(病毒性肝炎vs其他) 0.76(0.31~1.88) 0.552 TBil(μmol/L) 1.01(0.99~1.03) 0.178 ALT(U/L) 1.00(1.00~1.00) 0.116 AST(U/L) 1.00(1.00~1.00) 0.176 AFP(≥400 ng/mL vs <400 ng/mL) 2.09(1.35~3.25) <0.001 2.35(1.44~3.84) <0.001 PLT(×109/L) 1.00(1.00~1.01) 0.358 Child-Pugh分级(B级 vs A级) 2.13(1.05~4.33) 0.036 2.12(1.01~4.45) 0.048 ECOG PS评分(1分 vs 0分) 1.08(0.71~1.64) 0.717 肿瘤个数(多发vs单发) 1.13(0.70~1.84) 0.602 肿瘤最大直径(cm) 1.09(1.02~1.16) 0.007 1.02(0.95~1.09) 0.648 肝外转移(有vs无) 1.65(1.04~2.62) 0.034 1.97(1.18~3.28) 0.009 血管侵犯(有vs无) 1.29(0.84~1.96) 0.242 BCLC分期(C期 vs B期) 1.45(0.91~2.33) 0.120 既往局部治疗(有vs无) 0.77(0.49~1.23) 0.272 冷冻消融次数(≥2次 vs 1次) 1.04(0.68~1.58) 0.857 靶向药物治疗时长(月) 0.92(0.88~0.96) <0.001 0.92(0.89~0.96) <0.001 后续局部治疗(有vs无) 0.50(0.32~0.77) 0.002 0.59(0.37~0.95) 0.029 后续系统药物治疗(有vs无) 0.64(0.42~0.97) 0.037 0.46(0.29~0.75) 0.002 注:OS,总生存期;BMI,体重指数;TBil,总胆红素;ALT,丙氨酸氨基转移酶;AST,天冬氨酸氨基转移酶;AFP,甲胎蛋白;PLT,血小板计数;Child-Pugh分级,蔡尔德-皮尤分级;ECOG PS,美国东部肿瘤协作组体力状况;BCLC,巴塞罗那临床肝癌;HR,风险比;95%CI,95%置信区间。
表 6 影响患者PFS的单因素及多因素Cox回归分析
Table 6. Univariate and multivariate Cox proportional hazards regression model analysis of influencing PFS
变量 单因素 多因素 HR(95%CI) P值 HR(95%CI) P值 治疗方式(三联vs二联) 0.58(0.39~0.85) 0.005 0.52(0.35~0.78) 0.001 年龄(≥70岁 vs <70岁) 0.70(0.45~1.08) 0.108 性别(男vs女) 0.83(0.53~1.29) 0.410 BMI(kg/m2) 1.01(0.96~1.08) 0.567 病因学(病毒性肝炎vs其他) 0.81(0.33~1.99) 0.640 TBil(μmol/L) 1.00(0.98~1.01) 0.690 ALT(U/L) 1.00(1.00~1.00) 0.122 AST(U/L) 1.00(1.00~1.00) 0.168 AFP(≥400 ng/mL vs <400 ng/mL) 1.40(0.93~2.10) 0.103 PLT(×109/L) 1.00(1.00~1.00) 0.822 Child-Pugh分级(B级 vs A级) 1.03(0.53~1.98) 0.937 ECOG PS评分(1分 vs 0分) 1.05(0.72~1.53) 0.812 肿瘤个数(多发vs单发) 1.22(0.78~1.90) 0.384 肿瘤最大直径(cm) 1.03(0.98~1.09) 0.278 肝外转移(有vs无) 1.35(0.88~2.08) 0.173 血管侵犯(有vs无) 1.41(0.95~2.09) 0.089 0.76(0.42~1.37) 0.361 BCLC分期(C期 vs B期) 1.82(1.19~2.79) 0.006 1.88(0.91~3.87) 0.088 既往局部治疗(有vs无) 0.56(0.37~0.84) 0.006 0.66(0.39~1.10) 0.110 冷冻消融次数(≥2次 vs 1次) 1.28(0.86~1.90) 0.216 靶向药物治疗时长(月) 0.95(0.91~0.99) 0.009 0.94(0.90~0.99) 0.014 后续局部治疗(有vs无) 0.90(0.61~1.34) 0.606 后续系统药物治疗(有vs无) 0.67(0.45~0.98) 0.038 0.77(0.50~1.17) 0.213 注:PFS,无进展生存期;BMI,体重指数;TBil,总胆红素;ALT,丙氨酸氨基转移酶;AST,天冬氨酸氨基转移酶;AFP,甲胎蛋白;PLT,血小板计数;Child-Pugh分级,蔡尔德-皮尤分级;ECOG PS,美国东部肿瘤协作组体力状况;BCLC,巴塞罗那临床肝癌;HR,风险比;95%CI,95%置信区间。
表 7 治疗相关不良事件
Table 7. Treatment-related adverse reactions
不良事件 1~2级 3~4级 二联组(n=57) 三联组(n=57) P值 二联组(n=57) 三联组(n=57) P值 氩氦刀冷冻消融相关 转氨酶水平升高 57(100.0) 55(96.5) 0.476 0(0.0) 0(0.0) 发热 39(68.4) 39(68.4) >0.05 0(0.0) 0(0.0) 胆红素升高 33(57.9) 25(43.9) 0.190 0(0.0) 0(0.0) 胸腔积液 29(50.9) 28(49.1) >0.05 10(17.5) 6(10.5) 0.419 疼痛 25(43.9) 32(56.1) 0.261 0(0.0) 0(0.0) 血小板减少 28(49.1) 23(40.4) 0.451 0(0.0) 0(0.0) 低蛋白血症 29(50.9) 20(35.1) 0.130 0(0.0) 0(0.0) 感染 23(40.4) 20(35.1) 0.699 0(0.0) 0(0.0) 恶心呕吐 14(24.6) 25(43.9) 0.051 0(0.0) 0(0.0) 低钾血症 12(21.1) 19(33.3) 0.207 0(0.0) 0(0.0) 出血 2(3.5) 0(0.0) 0.476 2(3.5) 0(0.0) 0.476 靶向或免疫系统药物相关 高氨血症 18(31.6) 26(45.6) 0.178 2(3.5) 0(0.0) 0.476 低蛋白血症 18(31.6) 25(43.9) 0.246 0(0.0) 0(0.0) 贫血 17(29.8) 25(43.9) 0.174 3(5.3) 3(5.3) >0.05 蛋白尿 12(21.1) 15(26.3) 0.660 2(3.5) 2 (3.5) >0.05 血小板减少 9(15.8) 18(31.6) 0.078 4(7.0) 8(14.0) 0.205 白细胞减少 9(15.8) 13(22.8) 0.476 3(5.3) 10(17.5) 0.154 胆红素升高 12(21.1) 9(15.8) 0.629 1(1.8) 2(3.5) >0.05 高血压 9(15.8) 11(19.3) 0.805 4(7.0) 4(7.0) >0.05 乏力 10(17.5) 3(5.3) 0.077 3(5.3) 2(3.5) >0.05 腹泻 9(15.8) 4(7.0) 0.239 2(3.5) 2 (3.5) >0.05 手足综合征 3(5.3) 8(14.0) 0.205 2(3.5) 1(1.8) >0.05 食欲下降 5(8.8) 2(3.5) 0.435 3(5.3) 1(1.8) 0.611 -
[1] SUNG H, FERLAY J, SIEGEL RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA A Cancer J Clin, 2021, 71( 3): 209- 249. DOI: 10.3322/caac.21660. [2] CAO W, CHEN HD, YU YW, et al. Changing profiles of cancer burden worldwide and in China: A secondary analysis of the global cancer statistics 2020[J]. Chin Med J, 2021, 134( 7): 783- 791. DOI: 10.1097/CM9.0000000000001474. [3] JU W, ZHENG RS, ZHANG SW, et al. Cancer statistics in Chinese older people, 2022: Current burden, time trends, and comparisons with the US, Japan, and the Republic of Korea[J]. Sci China Life Sci, 2023, 66( 5): 1079- 1091. DOI: 10.1007/s11427-022-2218-x. [4] SEKIGUCHI S, TSUCHIYA K, YASUI Y, et al. Clinical usefulness of geriatric assessment in elderly patients with unresectable hepatocellular carcinoma receiving sorafenib or lenvatinib therapy[J]. Cancer Rep, 2022, 5( 11): e1613. DOI: 10.1002/cnr2.1613. [5] EOCHAGAIN C MAC, POWER R, SAM C, et al. Inclusion, characteristics, and reporting of older adults in FDA registration studies of immunotherapy, 2018-2022[J]. J Immunother Cancer, 2024, 12( 8): e009258. DOI: 10.1136/jitc-2024-009258. [6] National Health Commission of the People’s Republic of China. Standard for diagnosis and treatment of primary liver cancer(2024 edition)[J]. J Clin Hepatol, 2024, 40( 5): 893- 918. DOI: 10.12449/JCH240508.中华人民共和国国家卫生健康委员会. 原发性肝癌诊疗指南(2024年版)[J]. 临床肝胆病杂志, 2024, 40( 5): 893- 918. DOI: 10.12449/JCH240508. [7] REIG M, FORNER A, RIMOLA J, et al. BCLC strategy for prognosis prediction and treatment recommendation: The 2022 update[J]. J Hepatol, 2022, 76( 3): 681- 693. DOI: 10.1016/j.jhep.2021.11.018. [8] FINN RS, QIN SK, IKEDA M, et al. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma[J]. N Engl J Med, 2020, 382( 20): 1894- 1905. DOI: 10.1056/NEJMoa1915745. [9] TAMAI Y, FUJIWARA N, TANAKA T, et al. Combination therapy of immune checkpoint inhibitors with locoregional therapy for hepatocellular carcinoma[J]. Cancers(Basel), 2023, 15( 20): 5072. DOI: 10.3390/cancers15205072. [10] CAI JQ, ZHAO H, CHEN X. A new pattern of comprehensive treatment for hepatocellular carcinoma[J]. Chin J Dig Surg, 2024, 23( 2): 204- 208. DOI: 10.3760/cma.j.cn115610-20231208-00243.蔡建强, 赵宏, 陈晓. 肝癌综合治疗新格局[J]. 中华消化外科杂志, 2024, 23( 2): 204- 208. DOI: 10.3760/cma.j.cn115610-20231208-00243. [11] Liver Oncology Branch, China Association for the Promotion of International Exchange in Healthcare; Branch Immunology, China Association for the Promotion of International Exchange in Healthcare; Cooperative Group for Chinese Expert Consensus on Targeted Immunotherapy Combined with Local Therapy for Advanced Hepatocellular Cancer. Chinese expert consensus on targeted immunotherapy combined with local therapy for advanced hepatocellular cancer[J]. J Clin Hepatol, 2023, 39( 12): 2782- 2792. DOI: 10.3969/j.issn.1001-5256.2023.12.006.中国医疗保健国际交流促进会肝脏肿瘤学分会, 中国医疗保健国际交流促进会免疫学分会,《靶免联合局部治疗中晚期肝细胞癌中国专家共识》协作组. 靶向免疫联合局部治疗中晚期肝细胞癌中国专家共识[J]. 临床肝胆病杂志, 2023, 39( 12): 2782- 2792. DOI: 10.3969/j.issn.1001-5256.2023.12.006. [12] DEN BROK MHMGM, SUTMULLER RPM, NIERKENS S, et al. Efficient loading of dendritic cells following cryo and radiofrequency ablation in combination with immune modulation induces anti-tumour immunity[J]. Br J Cancer, 2006, 95( 7): 896- 905. DOI: 10.1038/sj.bjc.6603341. [13] LIU T, CHANG XJ, HE QW, et al. Efficacy and safety of cryoablation combined with lenvatinib and anti-PD-1 monoclonal antibody in treatment of unresectable hepatocellular carcinoma[J]. J Clin Hepatol, 2024, 40( 3): 539- 549. DOI: 10.12449/JCH240317.刘腾, 常秀娟, 何权威, 等. 冷冻消融协同仑伐替尼和程序性死亡受体1单抗治疗不可切除肝细胞癌的效果及安全性分析[J]. 临床肝胆病杂志, 2024, 40( 3): 539- 549. DOI: 10.12449/JCH240317. [14] WANG H, LITTRUP PJ, DUAN Y, et al. Thoracic masses treated with percutaneous cryotherapy: Initial experience with more than 200 procedures[J]. Radiology, 2005, 235( 1): 289- 298. DOI: 10.1148/radiol.2351030747. [15] FREITES-MARTINEZ A, SANTANA N, ARIAS-SANTIAGO S, et al. Using the common terminology criteria for adverse events(CTCAE- version 5.0) to evaluate the severity of adverse events of anticancer therapies[J]. Actas Dermosifiliogr(Engl Ed), 2021, 112( 1): 90- 92. DOI: 10.1016/j.ad.2019.05.009. [16] CLAVIEN PA, BARKUN J, DE OLIVEIRA ML, et al. The Clavien-Dindo classification of surgical complications: Five-year experience[J]. Ann Surg, 2009, 250( 2): 187- 196. DOI: 10.1097/SLA.0b013e3181b13ca2. [17] HARDER VS, STUART EA, ANTHONY JC. Propensity score techniques and the assessment of measured covariate balance to test causal associations in psychological research[J]. Psychol Methods, 2010, 15( 3): 234- 249. DOI: 10.1037/a0019623. [18] ZHANG JX, CHEN P, LIU S, et al. Safety and efficacy of transarterial chemoembolization and immune checkpoint inhibition with camrelizumab for treatment of unresectable hepatocellular carcinoma[J]. J Hepatocell Carcinoma, 2022, 9: 265- 272. DOI: 10.2147/JHC.S358658. [19] LIU WB, XIE ZG, SHEN KF, et al. Analysis of the safety and effectiveness of TACE combined with targeted immunotherapy in the treatment of intermediate and advanced hepatocellular carcinoma[J]. Med Oncol, 2023, 40( 9): 251. DOI: 10.1007/s12032-023-02082-x. [20] LIM M, ESPINOZA M, HUANG YH, et al. Complete response to immunotherapy in patients with hepatocellular carcinoma[J]. JAMA Netw Open, 2025, 8( 2): e2461735. DOI: 10.1001/jamanetworkopen.2024.61735. [21] CHEN KN, SHUEN TWH, CHOW PKH. The association between tumour heterogeneity and immune evasion mechanisms in hepatocellular carcinoma and its clinical implications[J]. Br J Cancer, 2024, 131( 3): 420- 429. DOI: 10.1038/s41416-024-02684-w. [22] TONG J, TAN YC, OUYANG WW, et al. Targeting immune checkpoints in hepatocellular carcinoma therapy: Toward combination strategies with curative potential[J]. Exp Hematol Oncol, 2025, 14( 1): 65. DOI: 10.1186/s40164-025-00636-5. [23] EHLING J, TACKE F. Role of chemokine pathways in hepatobiliary cancer[J]. Cancer Lett, 2016, 379( 2): 173- 183. DOI: 10.1016/j.canlet.2015.06.017. [24] WEI JL, CUI W, FAN WZ, et al. Unresectable hepatocellular carcinoma: Transcatheter arterial chemoembolization combined with microwave ablation vs. combined with cryoablation[J]. Front Oncol, 2020, 10: 1285. DOI: 10.3389/fonc.2020.01285. [25] CHEN ZN, MENG LL, ZHANG J, et al. Progress in the cryoablation and cryoimmunotherapy for tumor[J]. Front Immunol, 2023, 14: 1094009. DOI: 10.3389/fimmu.2023.1094009. [26] TAN HT, JIANG YQ, SHEN LJ, et al. Cryoablation-induced neutrophil Ca2+ elevation and NET formation exacerbate immune escape in colorectal cancer liver metastasis[J]. J Exp Clin Cancer Res, 2024, 43( 1): 319. DOI: 10.1186/s13046-024-03244-z. [27] QIAN L, XIE L, ZHU Y, et al. Potent induction of antitumor immunity by combining cryo-thermal ablation with immune checkpoint inhibitors in hepatocellular carcinoma[J]. Liver Int, 2024, 44( 3): 723- 737. DOI: 10.1111/liv.15817. [28] RAHMA OE, HODI FS. The intersection between tumor angiogenesis and immune suppression[J]. Clin Cancer Res, 2019, 25( 18): 5449- 5457. DOI: 10.1158/1078-0432.CCR-18-1543. [29] SHIGETA K, DATTA M, HATO T, et al. Dual programmed death receptor-1 and vascular endothelial growth factor receptor-2 blockade promotes vascular normalization and enhances antitumor immune responses in hepatocellular carcinoma[J]. Hepatology, 2020, 71( 4): 1247- 1261. DOI: 10.1002/hep.30889. [30] FELISMINO T, MARTINS L, BARROSO M, et al. Neutrophil-to-lymphocyte ratio(NLR) as a predictive biomarker in advanced hepatocellular carcinoma treated with first-line immunotherapy[J]. J Gastrointest Cancer, 2025, 56( 1): 175. DOI: 10.1007/s12029-025-01299-5. [31] ROCCO A, SGAMATO C, PELIZZARO F, et al. Systemic inflammatory response markers improve the discrimination for prognostic model in hepatocellular carcinoma[J]. Hepatol Int, 2025, 19( 4): 915- 928. DOI: 10.1007/s12072-025-10806-6. [32] IMAI K, TAKAI K, AIBA M, et al. Adverse events in targeted therapy for unresectable hepatocellular carcinoma predict clinical outcomes[J]. Cancers(Basel), 2024, 16( 18): 3150. DOI: 10.3390/cancers16183150. [33] ZHANG Y, WANG M, CHEN Q, et al. Adverse events of immune checkpoint inhibitor-based therapies for unresectable hepatocellular carcinoma in prospective clinical trials: A systematic review and meta-analysis[J]. Liver Cancer, 2022, 12( 6): 521- 538. DOI: 10.1159/000528698. -



 PDF下载 ( 1772 KB)
PDF下载 ( 1772 KB)

 下载:
下载: