细胞器相互作用在肝纤维化进展中的作用机制及中药防治策略
DOI: 10.12449/JCH260329
Mechanism of action of organelle interactions in the progression of liver fibrosis and traditional Chinese medicine prevention and treatment strategies
-
摘要: 肝纤维化是多种慢性肝病进展至肝硬化的核心病理阶段,其发生发展主要依赖于肝星状细胞的活化及胶原纤维的异常沉积。近年研究发现,肝星状细胞的活化受到多种细胞器(包括线粒体、内质网、高尔基体、溶酶体和过氧化物酶体等)彼此间复杂相互作用的调控,这些互相作用共同影响能量代谢、蛋白质合成与折叠、活性氧平衡及自噬等关键细胞过程,进而参与肝纤维化进展。同时,具有多靶点协同作用的中药及其活性成分受到研究者的广泛关注。本文从细胞器间相互作用的角度出发,系统阐述其在肝纤维化进展中的具体作用机制,并重点综述中药如何通过调控上述细胞器功能及其互作网络,抑制肝星状细胞活化及胶原生成,从而发挥抗肝纤维化效应,以期为深入解析肝纤维化病理机制及开发中药新型干预策略提供理论依据。Abstract: Liver fibrosis is the core pathological stage of the progression of various chronic liver diseases to liver cirrhosis, and hepatic stellate cell (HSC) activation and the abnormal accumulation of collagen fibers are important processes for the development and progression of liver fibrosis. In recent years, studies have shown that HSC activation is regulated by the complex interactions between various organelles (including mitochondria, endoplasmic reticulum, Golgi apparatus, lysosome, and peroxisomes), and such interactions affect the key cellular processes such as energy metabolism, protein synthesis and folding, reactive oxygen species balance, and autophagy, thereby participating in the progression of liver fibrosis. Meanwhile, traditional Chinese medicine and its active ingredients with multi-target synergistic effects have attracted wide attention. From the perspective of the interaction between organelles, this article systematically elaborates on the specific mechanism of such interactions in the progression of liver fibrosis and reviews how traditional Chinese medicine inhibits HSC activation and collagen production by regulating the function of these organelle and their interaction networks, thereby exerting an anti-liver fibrosis effect, in order to provide a theoretical basis for in-depth understanding of the pathological mechanism of liver fibrosis and the development of new traditional Chinese medicine intervention strategies.
-
Key words:
- Hepatic Fibrosis /
- Organelles /
- Therapeutics
-
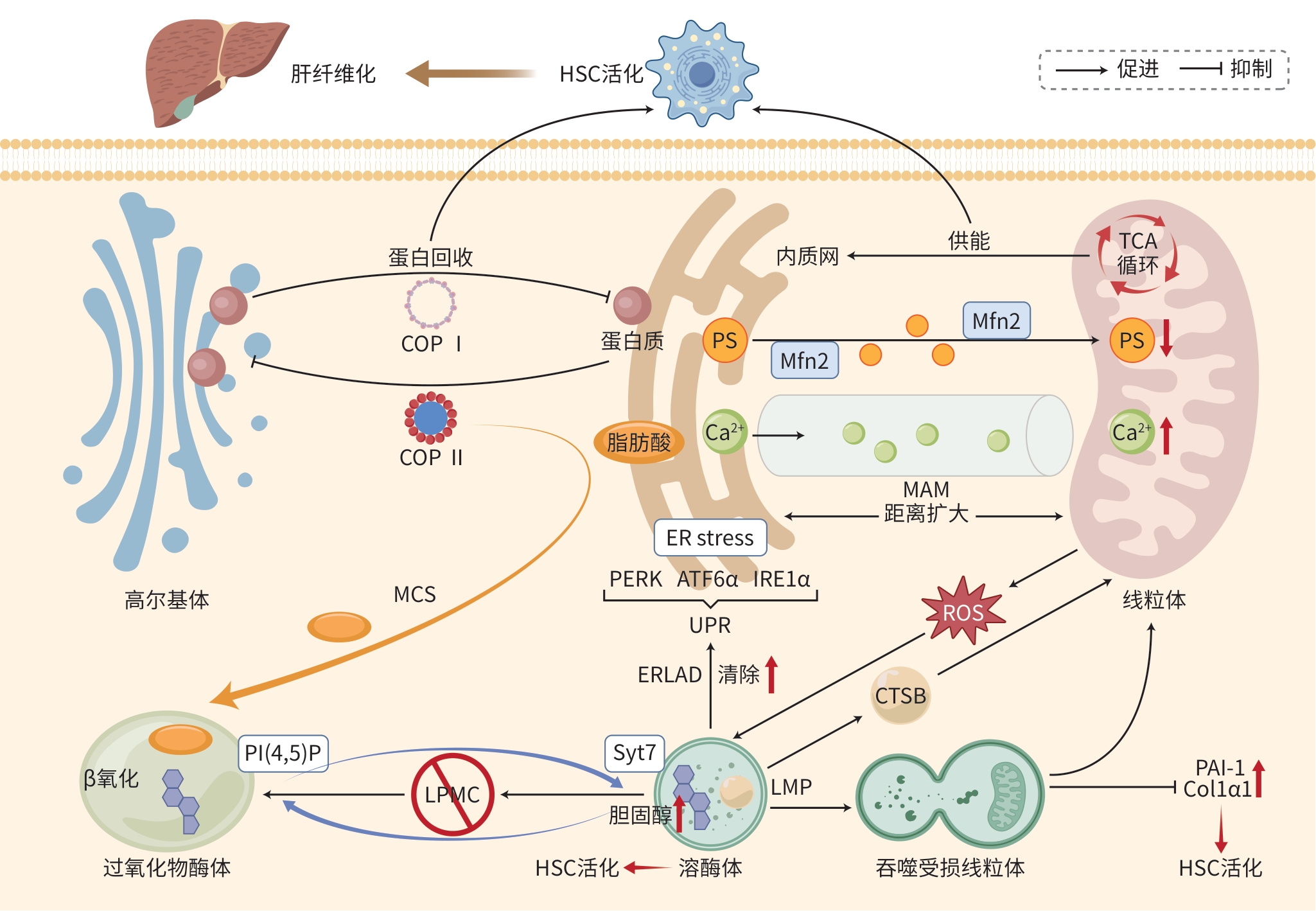
注: HSC,肝星状细胞;COPⅠ,包被蛋白Ⅰ;COPⅡ,包被蛋白Ⅱ;TCA循环,三羧酸循环;PS,磷脂酰丝氨酸;Mfn2,线粒体融合蛋白2;ER stress,内质网应激;PERK,蛋白激酶R样内质网激酶;ATF6α,激活转录因子6α;IRE1α,肌醇需求蛋白1α;UPR,未折叠蛋白质反应;ERLAD,ER-溶酶体相关降解途径;Syt7,溶酶体突触结合蛋白Ⅶ;MCS,膜接触位点;PI(4,5)P,磷脂酰肌醇-4,5-二磷酸;CTSB,组织蛋白酶B;LMP,溶酶体膜透化;PAI-1,纤溶酶原激活物抑制剂-1;Col1α1,Ⅰ型胶原α1。
图 1 细胞器间相互作用影响肝纤维化进展
Figure 1. Intercellular organelle interactions in liver fibrosis progression
-
[1] VALM AM, COHEN S, LEGANT WR, et al. Applying systems-level spectral imaging and analysis to reveal the organelle interactome[J]. Nature, 2017, 546( 7656): 162- 167. DOI: 10.1038/nature22369. [2] LIAO ZH, XIE ZY. Research progress in molecular mechanism of hepatic fibrosis and related therapeutic targets[J]. J Jilin Univ Med Ed, 2024, 50( 5): 1450- 1456. DOI: 10.13481/j.1671-587X.20240532.廖昭辉, 谢正元. 肝纤维化发病的分子机制及其相关治疗靶点的研究进展[J]. 吉林大学学报(医学版), 2024, 50( 5): 1450- 1456. DOI: 10.13481/j.1671-587X.20240532. [3] ILHA M, SEHGAL R, MATILAINEN J, et al. Indole-3-propionic acid promotes hepatic stellate cells inactivation[J]. J Transl Med, 2025, 23( 1): 253. DOI: 10.1186/s12967-025-06266-z. [4] KIM H, CHEN Q, JU DH, et al. ER-tethered stress sensor CREBH regulates mitochondrial unfolded protein response to maintain energy homeostasis[J]. Proc Natl Acad Sci USA, 2024, 121( 49): e2410486121. DOI: 10.1073/pnas.2410486121. [5] JIANG Y, LI LL, CHEN X, et al. Three-dimensional ATUM-SEM reconstruction and analysis of hepatic endoplasmic reticulum-organelle interactions[J]. J Mol Cell Biol, 2021, 13( 9): 636- 645. DOI: 10.1093/jmcb/mjab032. [6] GUPTA P, SATA TN, YADAV AK, et al. TGF-β induces liver fibrosis via miRNA-181a-mediated down regulation of augmenter of liver regeneration in hepatic stellate cells[J]. PLoS One, 2019, 14( 6): e0214534. DOI: 10.1371/journal.pone.0214534. [7] WANG XL, HE JH, XIE P, et al. Augmenter of liver regeneration crotonylation assists in mitochondria-ER contact to alleviate hepatic steatosis[J]. Cell Mol Gastroenterol Hepatol, 2025, 19( 3): 101436. DOI: 10.1016/j.jcmgh.2024.101436. [8] WU HX, CARVALHO P, VOELTZ GK. Here, there, and everywhere: The importance of ER membrane contact sites[J]. Science, 2018, 361( 6401): eaan5835. DOI: 10.1126/science.aan5835. [9] HERNÁNDEZ-ALVAREZ MI, SEBASTIÁN D, VIVES S, et al. Deficient endoplasmic reticulum-mitochondrial phosphatidylserine transfer causes liver disease[J]. Cell, 2019, 177( 4): 881- 895.e17. DOI: 10.1016/j.cell.2019.04.010. [10] ILHA M, MEIRA MARTINS LA, SILVEIRA MORAES K DA, et al. Caveolin-1 influences mitochondrial plasticity and function in hepatic stellate cell activation[J]. Cell Biol Int, 2022, 46( 11): 1787- 1800. DOI: 10.1002/cbin.11876. [11] ZIEGLER DV, VINDRIEUX D, GOEHRIG D, et al. Calcium channel ITPR2 and mitochondria-ER contacts promote cellular senescence and aging[J]. Nat Commun, 2021, 12( 1): 720. DOI: 10.1038/s41467-021-20993-z. [12] KIM SH, KIM JY, PARK SY, et al. Activation of the EGFR-PI3K-CaM pathway by PRL-1-overexpressing placenta-derived mesenchymal stem cells ameliorates liver cirrhosis via ER stress-dependent calcium[J]. Stem Cell Res Ther, 2021, 12( 1): 551. DOI: 10.1186/s13287-021-02616-y. [13] SUN T, LV HM, SHAO HR, et al. Chondroitin sulfate as a lysosomal enhancer attenuates lipid-driven inflammation via lipophagy and mitophagy[J]. Mar Drugs, 2025, 23( 6): 228. DOI: 10.3390/md23060228. [14] PAN M, SHI XY. Role of mitophagy in the development and progression of liver-related diseases[J]. J Clin Hepatol, 2024, 40( 2): 413- 418. DOI: 10.12449/JCH240232.潘萌, 史晓燕. 线粒体自噬在肝脏相关疾病发生发展中的作用[J]. 临床肝胆病杂志, 2024, 40( 2): 413- 418. DOI: 10.12449/JCH240232. [15] LEE JH, SEO KH, YANG JH, et al. CCCP induces hepatic stellate cell activation and liver fibrogenesis via mitochondrial and lysosomal dysfunction[J]. Free Radic Biol Med, 2024, 225: 181- 192. DOI: 10.1016/j.freeradbiomed.2024.10.259. [16] BAULIES A, RIBAS V, NÚÑEZ S, et al. Lysosomal cholesterol accumulation sensitizes to acetaminophen hepatotoxicity by impairing mitophagy[J]. Sci Rep, 2015, 5: 18017. DOI: 10.1038/srep18017. [17] GOU YS, WANG LF, ZHAO JH, et al. PNPLA3-I148M variant promotes the progression of liver fibrosis by inducing mitochondrial dysfunction[J]. Int J Mol Sci, 2023, 24( 11): 9681. DOI: 10.3390/ijms24119681. [18] TORRES S, SOLSONA-VILARRASA E, NUÑEZ S, et al. Acid ceramidase improves mitochondrial function and oxidative stress in Niemann-Pick type C disease by repressing STARD1 expression and mitochondrial cholesterol accumulation[J]. Redox Biol, 2021, 45: 102052. DOI: 10.1016/j.redox.2021.102052. [19] SANTRA A, BISHNU D, SANTRA S, et al. Arsenic-induced injury of mouse hepatocytes through lysosome and mitochondria: An in vitro study[J]. Int J Hepatol, 2022, 2022: 1546297. DOI: 10.1155/2022/1546297. [20] WEI HR, XIAO F, WEI HS. Research progress on the mechanism of endoplasmic reticulum stress in metabolic dysfunction-associated fatty liver disease[J/OL]. Chin J Liver Dis Electron Version, 2024, 16( 1): 13- 17. DOI: 10.3969/j.issn.1674-7380.2024.01.003.韦何锐, 肖凡, 魏红山. 内质网应激在代谢相关脂肪性肝病中的作用机制研究进展[J/OL]. 中国肝脏病杂志(电子版), 2024, 16( 1): 13- 17. DOI: 10.3969/j.issn.1674-7380.2024.01.003. [21] OKISHIO S, YAMAGUCHI K, ISHIBA H, et al. PPARα agonist and metformin co-treatment ameliorates NASH in mice induced by a choline-deficient, amino acid-defined diet with 45% fat[J]. Sci Rep, 2020, 10( 1): 19578. DOI: 10.1038/s41598-020-75805-z. [22] ANTELO-CEA DA, MARTÍNEZ-ROJAS L, CABRERIZO-IBÁÑEZ I, et al. Regulation of mitochondrial and peroxisomal metabolism in female obesity and type 2 diabetes[J]. Int J Mol Sci, 2024, 25( 20): 11237. DOI: 10.3390/ijms252011237. [23] HERNÁNDEZ-GEA V, HILSCHER M, ROZENFELD R, et al. Endoplasmic reticulum stress induces fibrogenic activity in hepatic stellate cells through autophagy[J]. J Hepatol, 2013, 59( 1): 98- 104. DOI: 10.1016/j.jhep.2013.02.016. [24] HETZ C, ZHANG KZ, KAUFMAN RJ. Mechanisms, regulation and functions of the unfolded protein response[J]. Nat Rev Mol Cell Biol, 2020, 21( 8): 421- 438. DOI: 10.1038/s41580-020-0250-z. [25] XUE F, LU JW, BUCHL SC, et al. Coordinated signaling of activating transcription factor 6α and inositol-requiring enzyme 1α regulates hepatic stellate cell-mediated fibrogenesis in mice[J]. Am J Physiol Gastrointest Liver Physiol, 2021, 320( 5): G864- G879. DOI: 10.1152/ajpgi.00453.2020. [26] YU HJ, WANG CQ, QIAN BL, et al. GRINA alleviates hepatic ischemia-eperfusion injury-induced apoptosis and ER-phagy by enhancing HRD1-mediated ATF6 ubiquitination[J]. J Hepatol, 2025, 83( 1): 131- 145. DOI: 10.1016/j.jhep.2025.01.012. [27] MOHAMMADPOUR-ASL S, ROSHAN-MILANI B, ROSHAN-MILANI S, et al. Endoplasmic reticulum stress PERK-ATF4-CHOP pathway is involved in non-alcoholic fatty liver disease in type 1 diabetic rats: The rescue effect of treatment exercise and insulin-like growth factor I[J]. Heliyon, 2024, 10( 5): e27225. DOI: 10.1016/j.heliyon.2024.e27225. [28] KIM HJ, JOE Y, KIM SK, et al. Carbon monoxide protects against hepatic steatosis in mice by inducing sestrin-2 via the PERK-eIF2α-ATF4 pathway[J]. Free Radic Biol Med, 2017, 110: 81- 91. DOI: 10.1016/j.freeradbiomed.2017.05.026. [29] UZHYTCHAK M, LUNOVA M, SMOLKOVÁ B, et al. Iron oxide nanoparticles trigger endoplasmic reticulum damage in steatotic hepatic cells[J]. Nanoscale Adv, 2023, 5( 16): 4250- 4268. DOI: 10.1039/d3na00071k. [30] MAEDA M, ARAKAWA M, SAITO K. Disease-associated factors at the endoplasmic reticulum-Golgi interface[J]. Traffic, 2025, 26( 1-3): e70001. DOI: 10.1111/tra.70001. [31] JI C. Dissecting the role of disturbed ER-Golgi trafficking in antivirals and alcohol abuse-induced pathogenesis of liver disorders[J]. J Drug Abuse, 2017, 3( 3): 14. DOI: 10.21767/2471-853X.100054. [32] KHALATBARI A, MISHRA P, HAN H, et al. Ritonavir and lopinavir suppress RCE1 and CAAX rab proteins sensitizing the liver to organelle stress and injury[J]. Hepatol Commun, 2020, 4( 6): 932- 944. DOI: 10.1002/hep4.1515. [33] HAN H, HE YX, HU J, et al. Disrupted ER-to-Golgi trafficking underlies anti-HIV drugs and alcohol-induced cellular stress and hepatic injury[J]. Hepatol Commun, 2017, 1( 2): 122- 139. DOI: 10.1002/hep4.1030. [34] KE CL, XIAO CC, LI JM, et al. FMO2 ameliorates nonalcoholic fatty liver disease by suppressing ER-to-Golgi transport of SREBP1[J]. Hepatology, 2025, 81( 1): 181- 197. DOI: 10.1097/HEP.0000000000000643. [35] MAST FD, RACHUBINSKI RA, AITCHISON JD. Peroxisome prognostications: Exploring the birth, life, and death of an organelle[J]. J Cell Biol, 2020, 219( 3): e201912100. DOI: 10.1083/jcb.201912100. [36] ILACQUA N, ANASTASIA I, RAIMONDI A, et al. A three-organelle complex made by wrappER contacts with peroxisomes and mitochondria responds to liver lipid flux changes[J]. J Cell Sci, 2022, 135( 5): jcs259091. DOI: 10.1242/jcs.259091. [37] CHU BB, LIAO YC, QI W, et al. Cholesterol transport through lysosome-peroxisome membrane contacts[J]. Cell, 2015, 161( 2): 291- 306. DOI: 10.1016/j.cell.2015.02.019. [38] WANG YH, TWU YC, WANG CK, et al. Niemann-pick type C2 protein regulates free cholesterol accumulation and influences hepatic stellate cell proliferation and mitochondrial respiration function[J]. Int J Mol Sci, 2018, 19( 6): 1678. DOI: 10.3390/ijms19061678. [39] HE AY, CHEN XW, TAN M, et al. Acetyl-CoA derived from hepatic peroxisomal β-oxidation inhibits autophagy and promotes steatosis via mTORC1 activation[J]. Mol Cell, 2020, 79( 1): 30- 42.e4. DOI: 10.1016/j.molcel.2020.05.007. [40] HOU XX, LI YW, SONG JL, et al. Cryptotanshinone induces apoptosis of activated hepatic stellate cells via modulating endoplasmic reticulum stress[J]. World J Gastroenterol, 2023, 29( 17): 2616- 2627. DOI: 10.3748/wjg.v29.i17.2616. [41] MEI GB. Quercetin ameliorates metabolic-associated fatty liver disease in type 2 diabetes mellitus and the mediated mechanisms of MAMs-NLRP3[D]. Wuhan: Huazhong University of Science and Technology, 2021. DOI: 10.27157/d.cnki. ghzku.2021.004842.梅桂斌. 槲皮素缓解T2DM代谢相关脂肪性肝损伤及MAMs-NLRP3的介导机制[D]. 武汉: 华中科技大学, 2021. DOI: 10.27157/d.cnki. ghzku.2021.004842. [42] GAO ZS. Study on the protective effects and mechanisms of salidroside in drug-induced liver injury[D]. Chongqing: Southwest University, 2024. DOI: 10.27684/d.cnki.gxndx.2024.000062.高正山. 红景天苷对药物性肝损伤的保护作用及机制研究[D]. 重庆: 西南大学, 2024. DOI: 10.27684/d.cnki. gxndx.2024.000062. [43] MEIRA MARTINS LA, VIEIRA MQ, ILHA M, et al. The interplay between apoptosis, mitophagy and mitochondrial biogenesis induced by resveratrol can determine activated hepatic stellate cells death or survival[J]. Cell Biochem Biophys, 2015, 71( 2): 657- 672. DOI: 10.1007/s12013-014-0245-5. [44] LEE AY, JANG Y, HONG SH, et al. Ephedrine-induced mitophagy via oxidative stress in human hepatic stellate cells[J]. J Toxicol Sci, 2017, 42( 4): 461- 473. DOI: 10.2131/jts.42.461. [45] MENG ZQ. Study on the mechanism of apigenin alleviates liver injury induced by high fat diet through activating[D]. Changchun: Jilin University, 2023. DOI: 10.27162/d.cnki.gjlin.2023.007517.孟卓群. 芹菜素激活自噬缓解高脂饮食所致肝损伤的作用机制研究[D]. 长春: 吉林大学, 2023. DOI: 10.27162/ d.cnki.gjlin.2023.007517. [46] DUSABIMANA T, KIM SR, KIM HJ, et al. Nobiletin ameliorates hepatic ischemia and reperfusion injury through the activation of SIRT-1/FOXO3a-mediated autophagy and mitochondrial biogenesis[J]. Exp Mol Med, 2019, 51( 4): 51. DOI: 10.1038/s12276-019-0245-z. [47] XIAO HY, WANG L, WANG JH, et al. Exploring the role of curcumol on mitochondrial autophagy in hepatic stellate cells based on the PINK1/Parkin signalling pathway[J]. Acta Univ Med Anhui, 2025, 60( 5): 919- 928. DOI: 10.19405/j.cnki.issn1000-1492.2025.05.020.肖华业, 汪磊, 王佳慧, 等. 基于PINK1/Parkin信号通路探讨莪术醇对肝星状细胞线粒体自噬的作用[J]. 安徽医科大学学报, 2025, 60( 5): 919- 928. DOI: 10.19405/j.cnki.issn1000-1492.2025.05.020. [48] DENG ZW, REN CX, TANG CL, et al. Syringin alleviates hepatic fibrosis by enhancing autophagic flux and attenuating ER stress-TRIB3/SMAD3 in diabetic mice[J]. Tissue Cell, 2023, 83: 102159. DOI: 10.1016/j.tice.2023.102159. [49] SUN YF, PAN HH, XIA ZN, et al. Alisma Shugan Decoction attenuates hepatic fibrosis and endoplasmic reticulum stress in mice with carbon tetrachloride-induced fibrosis[J]. Afr Health Sci, 2023, 23( 2): 422- 434. DOI: 10.4314/ahs.v23i2.49. [50] GAO XB, GUO S, ZHANG S, et al. Matrine attenuates endoplasmic reticulum stress and mitochondrion dysfunction in nonalcoholic fatty liver disease by regulating SERCA pathway[J]. J Transl Med, 2018, 16( 1): 319. DOI: 10.1186/s12967-018-1685-2. -



 PDF下载 ( 822 KB)
PDF下载 ( 822 KB)

 下载:
下载:


