铁死亡抑制蛋白1(FSP1)-辅酶Q10(CoQ10)和二氢乳清酸脱氢酶(DHODH)双通路在肝细胞癌铁死亡中的作用及其临床意义
DOI: 10.12449/JCH260331
Dual ferroptosis suppressor protein 1-coenzyme Q10 and dihydroorotate dehydrogenase pathways in ferroptosis of hepatocellular carcinoma: Mechanisms and its clinical significance
-
摘要: 肝细胞癌(HCC)是全球常见且致死率高的恶性肿瘤,患者总体生存获益有限、耐药问题突出,亟需新的增敏策略和分层体系。铁死亡是一种铁依赖的脂质过氧化细胞死亡形式,与肿瘤治疗反应密切相关。除经典的谷胱甘肽过氧化物酶4(GPX4)/谷胱甘肽(GSH)通路外,铁死亡抑制蛋白1(FSP1)-辅酶Q10(CoQ10)和二氢乳清酸脱氢酶(DHODH)是两条新的抗铁死亡通路,分别在质膜和线粒体发挥作用,与GPX4协同决定细胞对铁死亡的敏感性。本文系统综述了FSP1-CoQ10和DHODH通路在HCC中的作用机制与研究进展,并提出相关治疗策略以及展望其临床转化应用前景。Abstract: Hepatocellular carcinoma (HCC) is a common malignant tumor with a high fatality rate worldwide, with limited overall survival benefits and pronounced drug resistance issues, highlighting the urgent need for novel sensitization strategies and patient stratification systems. Ferroptosis, as an iron-dependent form of lipid peroxidation-driven cell death, is closely associated with tumor treatment responses. In addition to the classic glutathione peroxidase 4 (GPX4)/glutathione (GSH) pathway, the ferroptosis suppressor protein 1 (FSP1)-coenzyme Q10 (CoQ10) pathways and the dihydroorotate dehydrogenase (DHODH) pathway are two newly identified anti-ferroptosis pathways that function at the plasma membrane and mitochondria, respectively, and determine cellular sensitivity to ferroptosis in synergy with GPX4. This article systematically reviews the mechanism of action of the FSP1-CoQ10 and DHODH pathways in HCC and related research advances, proposes related therapeutic strategies, and look forward to its clinical translation and application prospects.
-
Key words:
- Carcinoma, Hepatocellular /
- Ferroptosis /
- Signal Transduction /
- Pathologic Processes /
- Therapeutics
-
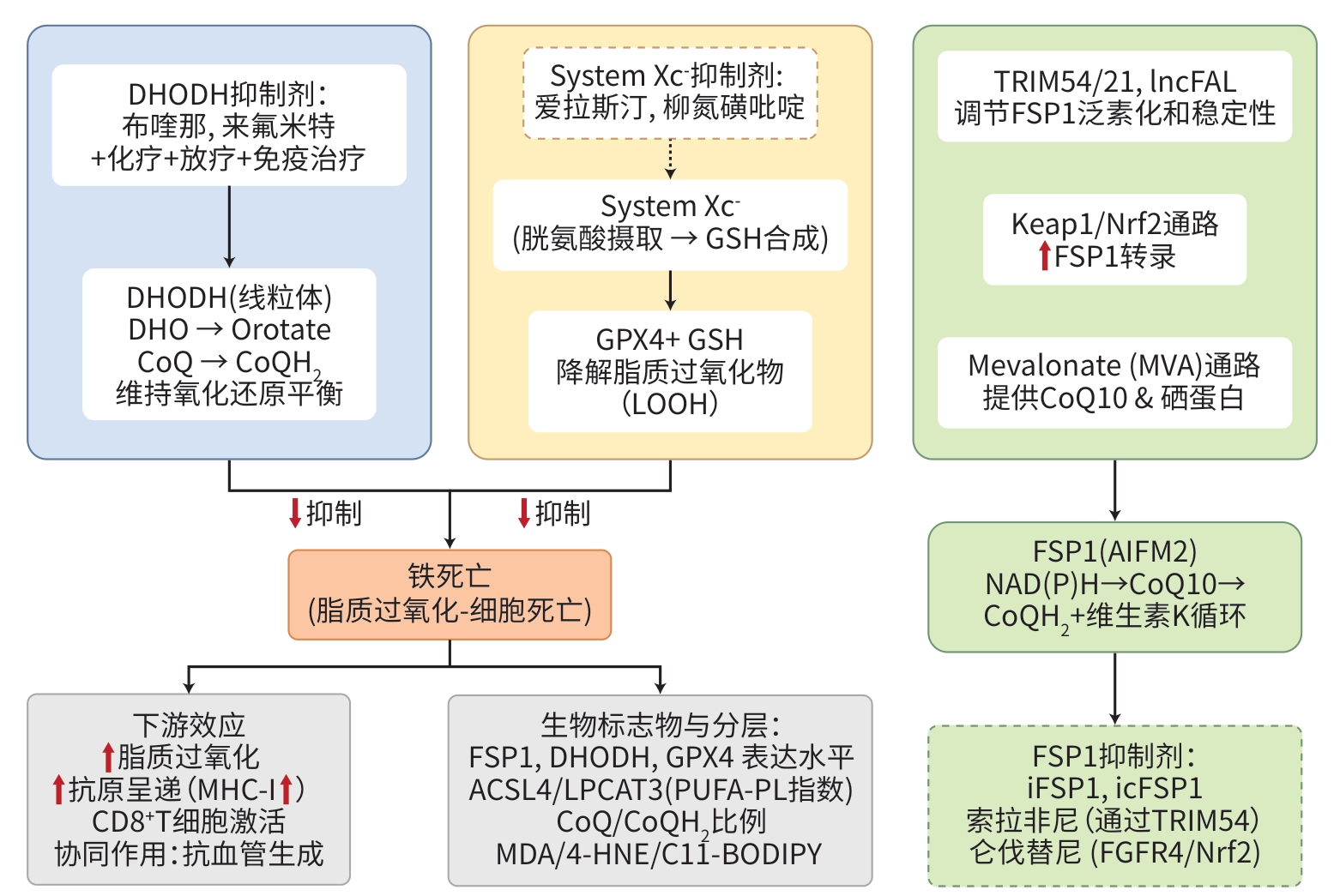
注: DHODH,二氢乳清酸脱氢酶;DHO,二氢乳清酸;Orotate,乳清酸根;CoQ/CoQH2,辅酶Q/泛醌醇;System Xc⁻,胱氨酸/谷氨酸反向转运系统;GSH,谷胱甘肽;LOOH,脂质过氧化物;GPX4,谷胱甘肽过氧化物酶4;TRIM54/21,含三联结构域蛋白54/21;lncFAL,铁死亡相关长链非编码RNA;Keap1/Nrf2,Kelch样ECH相关蛋白1/核因子E2相关因子2;MVA,甲羟戊酸;FSP1,铁死亡抑制蛋白1;AIFM2,凋亡诱导因子线粒体相关蛋白2;NAD(P)H,还原型烟酰胺腺嘌呤二核苷酸;FGFR4,成纤维生长因子受体4;MHC-Ⅰ,主要组织相容性复合体Ⅰ类分子;ACSL4/LPCAT3,长链脂酰辅酶A合成酶4/溶血磷脂酰胆碱酰基转移酶3;PUFA-PL,多不饱和脂肪酸磷脂;MDA/4-HNE/C11-BODIPY,丙二醛/4-羟基壬烯醛/C11-BODIPY探针。
图 1 FSP1-CoQ10与DHODH双通路在肝细胞癌铁死亡中的作用机制与转化策略
Figure 1. Dual-pathway mechanism and translational strategy of FSP1-CoQ10 and DHODH in ferroptosis of HCC
-
[1] SUNG H, FERLAY J, SIEGEL RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2021, 71( 3): 209- 249. DOI: 10.3322/caac.21660. [2] WANG Y, DENG BC. Hepatocellular carcinoma: Molecular mechanism, targeted therapy, and biomarkers[J]. Cancer Metastasis Rev, 2023, 42( 3): 629- 652. DOI: 10.1007/s10555-023-10084-4. [3] ZHANG C, LIU XY, JIN SD, et al. Ferroptosis in cancer therapy: A novel approach to reversing drug resistance[J]. Mol Cancer, 2022, 21( 1): 47. DOI: 10.1186/s12943-022-01530-y. [4] ZHU HY, YANG YJ, DUAN YN, et al. Nrf2/FSP1/CoQ10 axis-mediated ferroptosis is involved in sodium aescinate-induced nephrotoxicity[J]. Arch Biochem Biophys, 2024, 759: 110100. DOI: 10.1016/j.abb.2024.110100. [5] CAO JH, CHEN X, CHEN LL, et al. DHODH-mediated mitochondrial redox homeostasis: A novel ferroptosis regulator and promising therapeutic target[J]. Redox Biol, 2025, 85: 103788. DOI: 10.1016/j.redox.2025.103788. [6] JIANG XJ, STOCKWELL BR, CONRAD M. Ferroptosis: Mechanisms, biology and role in disease[J]. Nat Rev Mol Cell Biol, 2021, 22( 4): 266- 282. DOI: 10.1038/s41580-020-00324-8. [7] DIXON SJ, LEMBERG KM, LAMPRECHT MR, et al. Ferroptosis: An iron-dependent form of nonapoptotic cell death[J]. Cell, 2012, 149( 5): 1060- 1072. DOI: 10.1016/j.cell.2012.03.042. [8] STOCKWELL BR. Ferroptosis turns 10: Emerging mechanisms, physiological functions, and therapeutic applications[J]. Cell, 2022, 185( 14): 2401- 2421. DOI: 10.1016/j.cell.2022.06.003. [9] NGHIEM TH THI, KUSUMA F, PARK J, et al. Brief guide to detecting ferroptosis[J]. Mol Cells, 2025, 48( 11): 100276. DOI: 10.1016/j.mocell.2025.100276. [10] MARTINEZ AM, KIM A, YANG WS. Detection of ferroptosis by BODIPYTM 581/591 C11[J]. Methods Mol Biol, 2020, 2108: 125- 130. DOI: 10.1007/978-1-0716-0247-8_11. [11] YAN HF, ZOU T, TUO QZ, et al. Ferroptosis: Mechanisms and links with diseases[J]. Signal Transduct Target Ther, 2021, 6( 1): 49. DOI: 10.1038/s41392-020-00428-9. [12] ZILKA O, SHAH R, LI B, et al. On the mechanism of cytoprotection by ferrostatin-1 and liproxstatin-1 and the role of lipid peroxidation in ferroptotic cell death[J]. ACS Cent Sci, 2017, 3( 3): 232- 243. DOI: 10.1021/acscentsci.7b00028. [13] LI WT, LIANG L, LIU SY, et al. FSP1: A key regulator of ferroptosis[J]. Trends Mol Med, 2023, 29( 9): 753- 764. DOI: 10.1016/j.molmed.2023.05.013. [14] MISHIMA E, ITO J, WU ZJ, et al. A non-canonical vitamin K cycle is a potent ferroptosis suppressor[J]. Nature, 2022, 608( 7924): 778- 783. DOI: 10.1038/s41586-022-05022-3. [15] FANG WX, XIE SY, DENG W. Ferroptosis mechanisms and regulations in cardiovascular diseases in the past, present, and future[J]. Cell Biol Toxicol, 2024, 40( 1): 17. DOI: 10.1007/s10565-024-09853-w. [16] WEAVER K, SKOUTA R. The selenoprotein glutathione peroxidase 4: From molecular mechanisms to novel therapeutic opportunities[J]. Biomedicines, 2022, 10( 4): 891. DOI: 10.3390/biomedicines10040891. [17] CONRAD M, KAGAN VE, BAYIR H, et al. Regulation of lipid peroxidation and ferroptosis in diverse species[J]. Genes Dev, 2018, 32( 9-10): 602- 619. DOI: 10.1101/gad.314674.118. [18] BERSUKER K, HENDRICKS JM, LI ZP, et al. The CoQ oxidoreductase FSP1 acts parallel to GPX4 to inhibit ferroptosis[J]. Nature, 2019, 575( 7784): 688- 692. DOI: 10.1038/s41586-019-1705-2. [19] DOLL S, FREITAS FP, SHAH R, et al. FSP1 is a glutathione-independent ferroptosis suppressor[J]. Nature, 2019, 575( 7784): 693- 698. DOI: 10.1038/s41586-019-1707-0. [20] WANG C, CHEN CC, ZHOU JB, et al. DHODH alleviates heart failure via the modulation of CoQ-related ferroptotic inhibition[J]. Front Biosci, 2024, 29( 7): 267. DOI: 10.31083/j.fbl2907267. [21] PENG W, LIANG J, QIAN XL, et al. IGF2BP1/AIFM2 axis regulates ferroptosis and glycolysis to drive hepatocellular carcinoma progression[J]. Cell Signal, 2025, 130: 111660. DOI: 10.1016/j.cellsig.2025.111660. [22] PEI WC, JIANG MR, LIU HY, et al. The prognostic and antitumor roles of key genes of ferroptosis in liver hepatocellular cancer and stomach adenocarcinoma[J]. Cancer Biomark, 2024, 39( 4): 335- 347. DOI: 10.3233/CBM-230114. [23] CHEU JW, LEE D, LI QD, et al. Ferroptosis suppressor protein 1 inhibition promotes tumor ferroptosis and anti-tumor immune responses in liver cancer[J]. Cell Mol Gastroenterol Hepatol, 2023, 16( 1): 133- 159. DOI: 10.1016/j.jcmgh.2023.03.001. [24] KOPPULA P, LEI G, ZHANG YL, et al. A targetable CoQ-FSP1 axis drives ferroptosis- and radiation-resistance in KEAP1 inactive lung cancers[J]. Nat Commun, 2022, 13( 1): 2206. DOI: 10.1038/s41467-022-29905-1. [25] EMMANUEL N, LI HE, CHEN J, et al. FSP1 a novel KEAP1/NRF2 target gene regulating ferroptosis and radioresistance in lung cancers[J]. Oncotarget, 2022, 13: 1136- 1139. DOI: 10.18632/oncotarget.28301. [26] LEE J, SEO Y, ROH JL. Emerging therapeutic strategies targeting GPX4-mediated ferroptosis in head and neck cancer[J]. Int J Mol Sci, 2025, 26( 13): 6452. DOI: 10.3390/ijms26136452. [27] CHEN YL, LEE D, KWAN KK, et al. Mevalonate pathway promotes liver cancer by suppressing ferroptosis through CoQ10 production and selenocysteine-tRNA modification[J]. J Hepatol, 2025, 83( 6): 1338- 1352. DOI: 10.1016/j.jhep.2025.06.034. [28] GONG J, LIU YH, WANG WJ, et al. TRIM21-promoted FSP1 plasma membrane translocation confers ferroptosis resistance in human cancers[J]. Adv Sci, 2023, 10( 29): 2302318. DOI: 10.1002/advs.202302318. [29] LIU MR, SHI C, SONG QY, et al. Sorafenib induces ferroptosis by promoting TRIM54-mediated FSP1 ubiquitination and degradation in hepatocellular carcinoma[J]. Hepatol Commun, 2023, 7( 10): e0246. DOI: 10.1097/HC9.0000000000000246. [30] YUAN JS, LV T, YANG J, et al. HDLBP-stabilized lncFAL inhibits ferroptosis vulnerability by diminishing Trim69-dependent FSP1 degradation in hepatocellular carcinoma[J]. Redox Biol, 2022, 58: 102546. DOI: 10.1016/j.redox.2022.102546. [31] ZHU XY, SHA XD, ZANG Y, et al. Current progress of ferroptosis study in hepatocellular carcinoma[J]. Int J Biol Sci, 2024, 20( 9): 3621- 3637. DOI: 10.7150/ijbs.96014. [32] ISEDA N, ITOH S, TOSHIDA K, et al. Ferroptosis is induced by lenvatinib through fibroblast growth factor receptor-4 inhibition in hepatocellular carcinoma[J]. Cancer Sci, 2022, 113( 7): 2272- 2287. DOI: 10.1111/cas.15378. [33] ZENG FR, CHEN X, DENG GT. The anti-ferroptotic role of FSP1: Current molecular mechanism and therapeutic approach[J]. Mol Biomed, 2022, 3( 1): 37. DOI: 10.1186/s43556-022-00105-z. [34] XAVIER DA SILVA TN, SCHULTE C, ALVES AN, et al. Molecular characterization of AIFM2/FSP1 inhibition by iFSP1-like molecules[J]. Cell Death Dis, 2023, 14( 4): 281. DOI: 10.1038/s41419-023-05787-z. [35] NAKAMURA T, MISHIMA E, YAMADA N, et al. Integrated chemical and genetic screens unveil FSP1 mechanisms of ferroptosis regulation[J]. Nat Struct Mol Biol, 2023, 30( 11): 1806- 1815. DOI: 10.1038/s41594-023-01136-y. [36] HENDRICKS JM, DOUBRAVSKY CE, WEHRI E, et al. Identification of structurally diverse FSP1 inhibitors that sensitize cancer cells to ferroptosis[J]. Cell Chem Biol, 2023, 30( 9): 1090- 1103. DOI: 10.1016/j.chembiol.2023.04.007. [37] NAKAMURA T, HIPP C, SANTOS DIAS MOURÃO A, et al. Phase separation of FSP1 promotes ferroptosis[J]. Nature, 2023, 619( 7969): 371- 377. DOI: 10.1038/s41586-023-06255-6. [38] MAO C, LIU XG, ZHANG YL, et al. DHODH-mediated ferroptosis defence is a targetable vulnerability in cancer[J]. Nature, 2021, 593( 7860): 586- 590. DOI: 10.1038/s41586-021-03539-7. [39] MA TY, DU JT, ZHANG YF, et al. GPX4-independent ferroptosis-a new strategy in disease’s therapy[J]. Cell Death Discov, 2022, 8( 1): 434. DOI: 10.1038/s41420-022-01212-0. [40] WANG FD, MIN JX. DHODH tangoing with GPX4 on the ferroptotic stage[J]. Signal Transduct Target Ther, 2021, 6( 1): 244. DOI: 10.1038/s41392-021-00656-7. [41] MULLEN NJ, SHUKLA SK, THAKUR R, et al. DHODH inhibition enhances the efficacy of immune checkpoint blockade by increasing cancer cell antigen presentation[J]. eLife, 2024, 12: RP87292. DOI: 10.7554/eLife.87292. [42] YUAN YH, SUN WJ, XIE JQ, et al. RNA nanotherapeutics for hepatocellular carcinoma treatment[J]. Theranostics, 2025, 15( 3): 965- 992. DOI: 10.7150/thno.102964. [43] ZHENG XG, JIN XD, YE F, et al. Ferroptosis: A novel regulated cell death participating in cellular stress response, radiotherapy, and immunotherapy[J]. Exp Hematol Oncol, 2023, 12( 1): 65. DOI: 10.1186/s40164-023-00427-w. [44] SCHWARTSMANN G, DODION P, VERMORKEN JB, et al. Phase I study of Brequinar sodium(NSC 368390) in patients with solid malignancies[J]. Cancer Chemother Pharmacol, 1990, 25( 5): 345- 351. DOI: 10.1007/BF00686235. [45] BANKA VK, SAINAS S, MARTINO E, et al. Radiosynthesis of[18F] brequinar for in vivo PET imaging of hDHODH for potential studies of acute myeloid leukemia and cancers[J]. RSC Med Chem, 2024, 15( 9): 3147- 3161. DOI: 10.1039/d4md00433g. [46] KEPP O, KROEMER G. Is ferroptosis immunogenic? The devil is in the details![J]. Oncoimmunology, 2022, 11( 1): 2127273. DOI: 10.1080/2162402X.2022.2127273. [47] GAO CL, ZHANG HR, WANG XW. Current advances on the role of ferroptosis in tumor immune evasion[J]. Discov Oncol, 2024, 15( 1): 736. DOI: 10.1007/s12672-024-01573-1. [48] LOPES-COELHO F, MARTINS F, HIPÓLITO A, et al. The activation of endothelial cells relies on a ferroptosis-like mechanism: Novel perspectives in management of angiogenesis and cancer therapy[J]. Front Oncol, 2021, 11: 656229. DOI: 10.3389/fonc.2021.656229. [49] ZHOU Y, TAO L, ZHOU X, et al. DHODH and cancer: Promising prospects to be explored[J]. Cancer Metab, 2021, 9( 1): 22. DOI: 10.1186/s40170-021-00250-z. [50] CHEN HS, WANG CY, LIU ZM, et al. Ferroptosis and its multifaceted role in cancer: Mechanisms and therapeutic approach[J]. Antioxidants, 2022, 11( 8): 1504. DOI: 10.3390/antiox11081504. [51] CHEN FQ, KANG R, LIU J, et al. The ACSL4 network regulates cell death and autophagy in diseases[J]. Biology, 2023, 12( 6): 864. DOI: 10.3390/biology12060864. [52] DOLL S, PRONETH B, TYURINA YY, et al. ACSL4 dictates ferroptosis sensitivity by shaping cellular lipid composition[J]. Nat Chem Biol, 2017, 13( 1): 91- 98. DOI: 10.1038/nchembio.2239. [53] EBELING MC, POLANCO JR, QU J, et al. Improving retinal mitochondrial function as a treatment for age-related macular degeneration[J]. Redox Biol, 2020, 34: 101552. DOI: 10.1016/j.redox.2020.101552. -



 PDF下载 ( 733 KB)
PDF下载 ( 733 KB)

 下载:
下载:


