凝血异常在HBV相关慢加急性肝衰竭患者血栓形成中的作用
DOI: 10.3969/j.issn.1001-5256.2021.03.012
Role of coagulation abnormalities in thrombosis in patients with hepatitis B virus-associated acute-on-chronic liver failure
-
摘要:
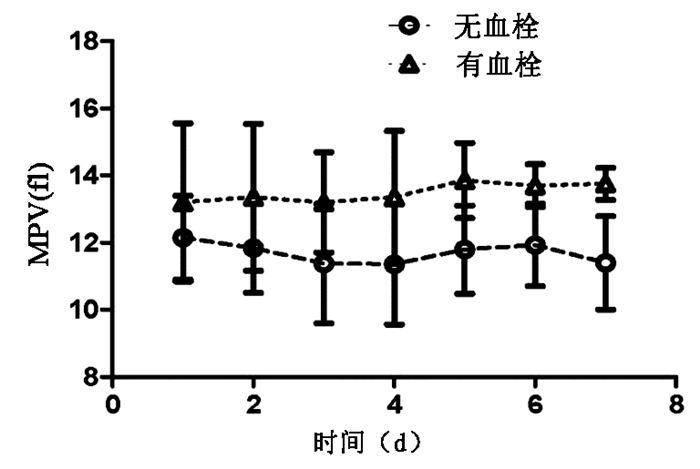
目的 观察传统凝血功能和血小板相关指标对HBV相关慢加急性肝衰竭(HBV-ACLF)患者血栓形成事件的作用。 方法 选取2015年1月—2019年12月在苏州大学附属第一医院住院的HBV-ACLF患者56例,分为发生血栓组(n=24)与未发生血栓组(n=32)。回顾性分析两组患者入院时的一般临床资料,观察入院后第1~7天的凝血功能,血小板计数和血小板功能相关指标平均血小板体积(MPV)的变化。符合正态分布的计量资料两组间比较采用t检验,不符合正态分布的计量资料两组间比较采用Mann-Whitney U秩和检验;计数资料两组间比较采用χ2检验。采用重复测量资料方差分析比较组内及组间不同时间凝血指标的差异。 结果 HBV-ACLF患者在入院时,发生血栓组年龄31.5(29.0~34.0)岁,较未发生血栓组年龄48.5(36.0~50.7)岁小,差异有统计学意义(Z=-2.637,P=0.008);在入院当天,MPV在发生血栓组与未发生血栓组间差异无统计学意义(P>0.05)。在入院后第2~7天,MPV值发生血栓组与未发生血栓组间差异均有统计学意义(t值分别为-2.696、-2.742、-2.894、-4.174、-3.945、-4.716,P值均<0.01)。发生血栓组MPV的峰值为入院第5天,均值为(13.90±1.12)fl,高于正常值范围。所有纳入患者在入院时,PT均值为(28.8±7.2)s、APTT均值(50.5±8.7)s、INR均值(2.6±0.7),均高于正常值;Fb均值为(1.16±0.3)g/L、血小板计数均值为(107.7±26.5)×109/L,均低于正常值。而PT、APTT、Fbg、INR及血小板计数在发生血栓组与未发生血栓组间差异均无统计学意义(P值均>0.05)。 结论 肝衰竭患者凝血功能障碍,更多是一种低平衡状态,是复杂与异质性的,需要个体化处理。HBV-ACLF患者中,易发生血栓事件者可能与血小板的功能有关,而与血小板计数及凝血常规指标关系不大。 Abstract:Objective To investigate the role of coagulation function parameters and platelet indices in thrombotic events in patients with hepatitis B virus-associated acute-on-chronic liver failure (HBV-ACLF). Methods A total of 56 patients with HBV-ACLF who were hospitalized in The First Affiliated Hospital of Soochow University from January 2015 to December 2019 were enrolled and divided into thrombotic complication (TC) group with 24 patients and non-thrombotic complication (NTC) group with 32 patients. A retrospective analysis was performed for their general clinical data on admission, and the patients were observed in terms of the changes in coagulation function, platelet count (PLT), and the platelet function-related index mean platelet volume (MPV) on days 1-7 after admission. The t-test was used for comparison of normally distributed continuous data between two groups, and the Mann-Whitney U rank sum test was used for comparison of non-normally distributed continuous data between two groups; the chi-square test was used for comparison of categorical data between two groups. A repeated measures analysis of variance was used to compare coagulation markers within and between groups at different time points. Results On admission, the TC group had a significantly younger age than the NTC group [31.5 (29.0-34.0) years vs 48.5 (36.0-50.7) years, Z=-2.637, P=0.008]. On the day of admission, there was no significant difference in MPV between the TC group and the NTC group (P > 0.05), while on days 2-7 after admission, there was a significant difference in MPV between the two groups (t=-2.696、-2.742、-2.894、-4.174、-3.945、-4.716, all P < 0.01). In the TC group, MPV reached the peak value on day 5 of admission, with a mean value of 13.90±1.12 fl, which was higher than the range of normal values. On admission, all patients had a mean prothrombin time (PT) of 28.8±7.2 s, a mean activated partial thromboplastin time (APTT) of 50.5±8.7 s, and a mean international normalized ratio (INR) of 2.6±0.7, which were higher than normal values; all patients had a mean fibrinogen (Fb) level of 1.16±0.3 g/L and a mean PLT of (107.7±26.5)×109/L, which were lower than normal values. There were no significant differences in PT, APTT, Fb, INR, and PLT between the TC group and the NTC group (all P > 0.05). Conclusion Coagulation disorder in patients with liver failure is more of a low-equilibrium state, which is complex and heterogeneous and requires individualized treatment. For patients with HBV-ACLF, the development of thrombotic events may be more associated with platelet function than PLT or conventional coagulation markers. -
Key words:
- Acute-On-Chronic Liver Failure /
- Hepatitis B Virus /
- Mean Platelet Volume /
- Thrombosis
-
表 1 患者一般临床资料与血制品使用情况
指标 NTC组(n=32) TC组(n=24) 统计值 P值 年龄(岁) 48.5(36.0~50.7) 31.5(29.0~34.0) Z=-2.637 0.008 性别(男/女) 20/12 15/9 χ2=-1.236 0.216 血小板计数(×109/L) 102.5(87.2~128.7) 91.0(58.0~124.0) Z=-1.127 0.260 MPV(fl) 12.1(11.6~12.9) 13.2(12.6~14.8) Z=-1.128 0.259 NH3(μmol/L) 67.1(47.7~81.5) 122.8(97.1~148.5) Z=-3.755 <0.001 Alb(g/L) 31.4(27.4~35.9) 30.8(29.3~32.3) Z=0 1.000 Cr(μmol/L) 55.3(48.3~72.6) 54.1(42.4~65.8) Z=-1.127 0.260 BUN(mmol/L) 3.8(3.0~5.1) 3.9(3.8~4.0) Z=-0.567 0.571 TBil(μmol/L) 298.5(209.6~353.7) 219.4(158.8~280) Z=-2.253 0.024 MELD评分 28.5(23.3~30.0) 25.5(23.0~28.0) Z=-1.514 0.130 入院时凝血指标 PT(s) 30.2(22.5~33.0) 26.8(24.3~29.3) Z=-1.502 0.133 APTT(s) 52.6(43.1~58.9) 44.4(39.3~49.5) Z=-2.623 0.008 Fb(g/L) 1.10(0.90~1.40) 0.90(0.98~1.01) Z=-1.878 0.620 INR 2.7(2.0~3.1) 2.4(2.1~2.6) Z=-1.502 0.133 住院期间输注血液制品情况 新鲜冰冻血浆 ≥1000 ml[例(%)] 29(90.6) 18(75.0) χ2=1.459 0.227 中位数 6400(3000~12 500) 5000(5000~13 500) Z=-0.141 0.888 冷沉淀(u) 例数(%) 3(9.3) 3(12.5) χ2=0.000 1.000 中位数 7.5(3.8~9.0) 6.0(3.0~8.0) Z=-0.373 0.709 血小板(u) 例数(%) 7(21.8) 6(25.0) χ2=0.075 0.784 中位数 30.0(12.5~55.0) 20.0(12.5~40.0) Z=-0.781 0.435 红细胞(u) 例数(%) 10(34.3) 9(37.5) χ2=0.239 0.625 中位数 6.0(4.0~8.5) 6.0(4.0~8.0) Z=-0.501 0.959 血浆置换[例(%)] 16(50.0) 15(62.5) χ2=0.867 0.352 注:Fb,纤维蛋白原。 表 2 TC组与NTC组ACLF患者凝血功能与血小板指数变化比较
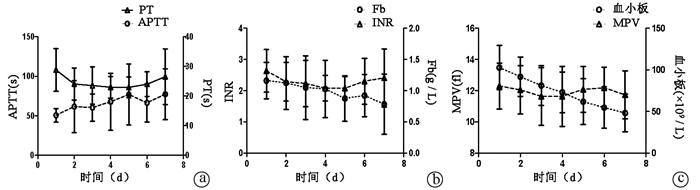
指标 第1天 第2天 第3天 第4天 第5天 第6天 第7天 F值 P值 PT(s) NTC组 29.2±7.5 24.0±5.7 23.6±6.8 22.7±3.9 22.6±3.6 24.0±4.4 26.6±10.1 10.9621) <0.0011) TC组 26.0±3.5 24.0±2.7 22.9±0.6 24.2±0.4 24.7±2.2 23.4±2.6 26.1±2.1 1.9951) 0.2011) t值 1.181 0.044 0.294 -1.048 -1.543 0.416 0.125 0.0222) 0.8822) P值 0.243 0.965 0.770 0.299 0.129 0.679 0.901 0.8883) 0.4393) APTT(s) NTC组 51.5±8.8 61.6±35.0 59.3±16.0 67.3±37.5 78.3±40.6 63.8±22.7 78.4±34.6 6.1841) <0.0011) TC组 44.7±5.1 60.5±16.0 65.6±24.5 69.7±27.2 67.8±22.0 81.5±28.3 70.0±5.7 6.4911) 0.0381) t值 2.115 0.090 -0.946 -0.175 0.714 -1.968 0.681 0.0002) 0.9902) P值 0.039 0.929 0.348 0.861 0.478 0.054 0.499 0.9873) 0.4093) Fb(g/L) NTC组 1.20±0.30 1.10±0.45 1.10±0.52 1.10±0.48 0.90±0.38 0.90±0.37 0.80±0.52 16.1311) <0.0011) TC组 1.00±0.01 1.10±0.14 0.70±0.27 0.80±0.1 0.80±0.22 0.90±0.05 0.70±0.01 13.4731) 0.0081) t值 1.783 0.098 2.122 1.821 0.880 -0.160 0.524 1.4362) 0.2362) P值 0.080 0.992 0.038 0.074 0.383 0.874 0.603 2.8653) 0.0443) INR NTC组 2.70±0.72 2.30±0.65 2.30±0.78 2.10±0.44 2.10±0.4 2.40±0.78 2.50±0.97 8.6551) <0.0011) TC组 2.40±0.28 2.10±0.27 1.80±0.18 2.00±0.11 2.10±0.11 2.00±0.12 1.90±0.32 8.3451) 0.0231) t值 1.162 0.788 1.724 0.448 -0.439 1.298 1.821 2.2532) 0.1392) P值 0.250 0.434 0.090 0.656 0.663 0.200 0.074 1.7243) 0.1653) 血小板计数(×109/L) NTC组 104±26 95±24 85±24 76±25 61±28 56±26 47±24 145.7051) <0.0011) TC组 97±29 73±5 60±3 54±4 66±28 50±19 56±16 5.6721) <0.0011) t值 0.655 2.543 2.966 2.483 -0.400 0.574 -1.045 1.1492) 0.2342) P值 0.515 0.014 0.004 0.016 0.691 0.569 0.301 7.3213) 0.0013) MPV(fl) NTC组 12.1±1.3 11.8±1.3 11.4±1.8 11.4±1.8 11.8±1.3 11.9±1.2 11.4±1.4 6.7181) <0.0011) TC组 13.2±2.4 13.4±2.2 13.2±1.5 13.4±2.0 13.9±1.1 13.7±0.6 13.8±0.5 1.0741) 0.3351) t值 -1.916 -2.696 -2.742 -2.894 -4.174 -3.945 -4.716 13.9142) <0.0012) P值 0.061 0.009 0.008 0.005 <0.001 <0.001 <0.001 1.7363) 0.1623) 注:1)不同时间比较;2)不同组间比较;3)时间与血栓形成的交互作用。 -
[1] Liver Failure and Artificial Liver Group, Chinese Society of Infectious Diseases, Chinese Medical Association; Severe Liver Disease and Artificial Liver Group, Chinese Society of Hepatology, Chinese Medical Association. Guideline for diagnosis and treatment of liver failure(2018)[J]. J Clin Hepatol, 2019, 35(1): 38-44. (in Chinese) DOI: 10.3969/j.issn.1001-5256.2019.01.007中华医学会感染病学分会肝衰竭与人工肝学组, 中华医学会肝病学分会重型肝病与人工肝学组. 肝衰竭诊治指南(2018年版)[J]. 临床肝胆病杂志, 2019, 35(1): 38-44. DOI: 10.3969/j.issn.1001-5256.2019.01.007 [2] VIOLI F, BASILI S, RAPARELLI V, et al. Patients with liver cirrhosis suffer from primary haemostatic defects? Fact or fiction?[J]. J Hepatol, 2011, 55(6): 1415-1427. DOI: 10.1016/j.jhep.2011.06.008 [3] DABBAGH O, OZA A, PRAKASH S, et al. Coagulopathy does not protect against venous thromboembolism in hospitalized patients with chronic liver disease[J]. Chest, 2010, 137(5): 1145-1149. DOI: 10.1378/chest.09-2177 [4] AGARWAL B, WRIGHT G, GATT A, et al. Evaluation of coagulation abnormalities in acute liver failure[J]. J Hepatol, 2012, 57(4): 780-786. DOI: 10.1016/j.jhep.2012.06.020 [5] WARRILLOW S, FISHER C, TIBBALLS H, et al. Coagulation abnormalities, bleeding, thrombosis, and management of patients with acute liver failure in Australia and New Zealand[J]. J Gastroenterol Hepatol, 2020, 35(5): 846-854. DOI: 10.1111/jgh.14876 [6] HABIB M, ROBERTS LN, PATEL RK, et al. Evidence of rebalanced coagulation in acute liver injury and acute liver failure as measured by thrombin generation[J]. Liver Int, 2014, 34(5): 672-678. DOI: 10.1111/liv.12369 [7] ALLISON MG, SHANHOLTZ CB, SACHDEVA A. Hematological issues in liver disease[J]. Crit Care Clin, 2016, 32(3): 385-396. DOI: 10.1016/j.ccc.2016.03.004 [8] BLASI A. Coagulopathy in liver disease: Lack of an assessment tool[J]. World J Gastroenterol, 2015, 21(35): 10062-10071. DOI: 10.3748/wjg.v21.i35.10062 [9] LYU BY, LI Q. Strategies in the evaluation of coagulation status in patients with liver failure[J]. J Clin Hepatol, 2020, 36(4): 928-931. (in Chinese) DOI: 10.3969/j.issn.1001-5256.2020.04.049吕滨月, 李谦. 肝衰竭患者的出凝血状态评估策略[J]. 临床肝胆病杂志, 2020, 36(4): 928-931. DOI: 10.3969/j.issn.1001-5256.2020.04.049 [10] STRAVITZ RT, ELLERBE C, DURKALSKI V, et al. Bleeding complications in acute liver failure[J]. Hepatology, 2018, 67(5): 1931-1942. DOI: 10.1002/hep.29694 [11] YIN JB, NIU Y, QIAN LY, et al. Mean platelet volume predicts survival in patients with hepatocellular carcinoma and type 2 diabetes[J]. Diabetes Res Clin Pract, 2019, 151: 120-127. DOI: 10.1016/j.diabres.2019.04.012 [12] MADAN SA, JOHN F, Pitchumoni CS. Nonalcoholic fatty liver disease and mean platelet volume: A systemic review and Meta-analysis[J]. J Clin Gastroenterol, 2016, 50(1): 69-74. DOI: 10.1097/MCG.0000000000000340 [13] POKORA RODAK A, KICIAK S, TOMASIEWICZ K. Neutrophil-lymphocyte ratio and mean platelet volume as predictive factors for liver fibrosis and steatosis in patients with chronic hepatitis B[J]. Ann Agric Environ Med, 2018, 25(4): 690-692. DOI: 10.26444/aaem/99583 [14] KISACIK B, TUFAN A, KALYONCU U, et al. Mean platelet volume (MPV) as an inflammatory marker in ankylosing spondylitis and rheumatoid arthritis[J]. Joint Bone Spine, 2008, 75(3): 291-294. DOI: 10.1016/j.jbspin.2007.06.016 [15] YATES KR, WELSH J, ECHRISH HH, et al. Pancreatic cancer cell and microparticle procoagulant surface characterization: Involvement of membrane-expressed tissue factor, phosphatidylserine and phosphatidylethanolamine[J]. Blood Coagul Fibrinolysis, 2011, 22(8): 680-687. DOI: 10.1097/MBC.0b013e32834ad7bc [16] EXNER T, JOSEPH JE, CONNOR D, et al. Increased procoagulant phospholipid activity in blood from patients with suspected acute coronary syndromes: A pilot study[J]. Blood Coagul Fibrinolysis, 2005, 16(5): 375-379. DOI: 10.1097/01.mbc.0000173465.45613.a3 [17] BRAEKKAN SK, MATHIESEN EB, NJ lSTAD I, et al. Mean platelet volume is a risk factor for venous thromboembolism: The tromsø study, tromsø, norway[J]. J Thromb Haemost, 2010, 8(1): 157-62. DOI: 10.1111/j.1538-7836.2009.03498.x [18] LISMAN T, BONGERS TN, ADELMEIJER J, et al. Elevated levels of von Willebrand Factor in cirrhosis support platelet adhesion despite reduced functional capacity[J]. Hepatology, 2006, 44(1): 53-61. DOI: 10.1002/hep.21231 [19] O'SHAUGHNESSY DF, ATTERBURY C, BOLTON MAGGS P, et al. Guidelines for the use of fresh-frozen plasma, cryoprecipitate and cryosupernatant[J]. Br J Haematol, 2004, 126(1): 11-28. DOI: 10.1111/j.1365-2141.2004.04972.x [20] SEGAL JB, DZIK WH, Transfusion Medicine/Hemostasis Clinical Trials Network. Paucity of studies to support that abnormal coagulation test results predict bleeding in the setting of invasive procedures: An evidence-based review[J]. Transfusion, 2005, 45(9): 1413-1425. DOI: 10.1111/j.1537-2995.2005.00546.x -



 PDF下载 ( 1915 KB)
PDF下载 ( 1915 KB)

 下载:
下载: