下瘀血汤抑制胶质细胞源性神经营养因子抗肝纤维化的作用机制
DOI: 10.3969/j.issn.1001-5256.2021.03.015
Mechanism of action of Xiayuxue decoction in inhibiting liver fibrosis by regulating glial cell line-derived neurotrophic factor
-
摘要:
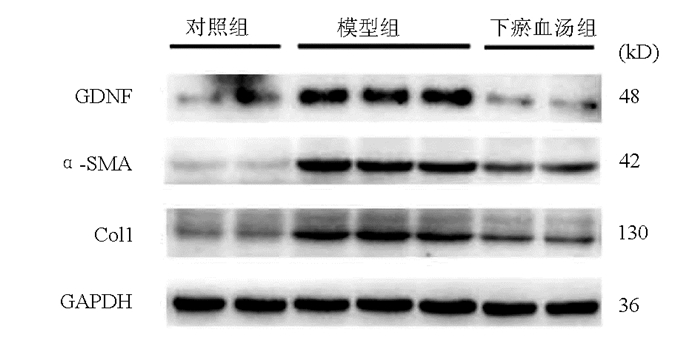
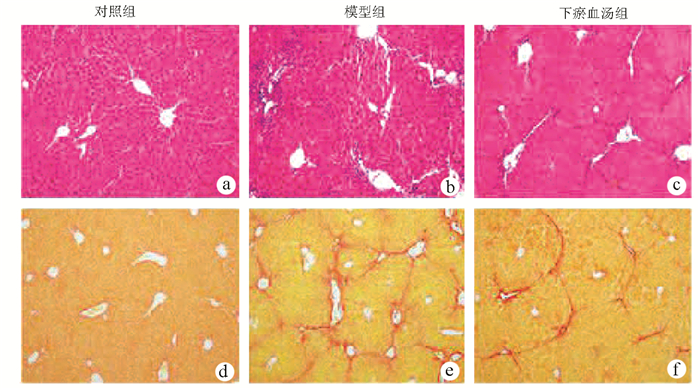
目的 探讨下瘀血汤是否通过抑制胶质细胞源性神经营养因子(GDNF)发挥抗肝纤维化的作用。 方法 24只C57BL/6小鼠随机分为对照组、模型组、下瘀血汤组,每组各8只。模型组和下瘀血汤小鼠腹腔注射10% CCl4,第4周开始下瘀血汤组小鼠给予0.467 8 g/kg下瘀血汤灌胃。检测肝功能指标ALT、AST水平,观测肝脏组织病理形态学。免疫组化检测平滑肌肌动蛋白(α-SMA)及GDNF蛋白表达。GDNF(10 ng/ml)处理GFP-Col-HSC和人原代肝星状细胞(HSC),检测HSC活化。计量资料多组间比较采用单因素方差分析,进一步两两比较采用LSD-t检验。 结果 模型组ALT和AST水平较对照组显著升高,下瘀血汤组ALT和AST水平较模型组显著降低(P值均<0.01)。肝组织病理学显示,模型组炎症细胞浸润明显,增生的胶原纤维形成纤维间隔,下瘀血汤组胶原纤维间隔较疏松及炎症细胞浸润减轻。免疫组化显示,与对照组相比,模型组α-SMA及GDNF阳性表达显著升高(P值均<0.01),均分布在纤维间隔,下瘀血汤组α-SMA与GDNF表达较模型组均显著降低(P值均<0.05)。免疫印迹结果显示,对照小鼠肝组织GDNF表达比较低,CCl4造模6周肝纤维化形成,GDNF表达上调10倍左右,下瘀血汤显著抑制模型小鼠GDNF蛋白表达(P值均<0.01);α-SMA和Ⅰ型胶原α1(Col1)表达在肝纤维化模型小鼠显著上调,下瘀血汤处理后α-SMA与Col1显著下降(P值均<0.01)。体外结果显示,GDNF可诱导HSC细胞α-SMA及Ⅰ型胶原α1蛋白表达显著上调,而下瘀血汤对此有显著抑制作用(P值均<0.01)。 结论 肝纤维化形成中GDNF表达显著上调,GDNF可诱导HSC活化,下瘀血汤可抑制GDNF从而抗肝纤维化。 -
关键词:
- 肝硬化 /
- 下瘀血汤 /
- 胶质细胞源性神经营养因子
Abstract:Objective To investigate whether Xiayuxue decoction exerts an anti-liver fibrosis effect by inhibiting glial cell line-derived neurotrophic factor (GDNF). Methods A total of 24 C57BL/6 mice were randomly divided into control group, model group, and Xiayuxue decoction group. The mice in the model group and the Xiayuxue decoction group were given intraperitoneal injection of 10% CCl4, and those in the Xiayuxue decoction group were given 0.4678 g/kg Xiayuxue decoction by gavage since week 4. The liver function parameters alanine aminotransferase (ALT) and aspartate aminotransferase (AST) were measured, and liver histopathology was observed. Immunohistochemistry was used to measure the protein expression of alpha-smooth muscle actin (α-SMA) and GDNF. GFP-Col-HSC and human primary hepatic stellate cells (HSCs) were treated with GDNF (10 ng/ml), and HSC activation was measured. A one-way analysis of variance was used for comparison of continuous data between multiple groups, and the least significant difference t-test was used for further comparison between two groups. Results Compared with the control group, the model group had significant increases in the levels of ALT and AST, and compared with the model group, the Xiayuxue decoction group had significant reductions in the levels of ALT and AST (all P < 0.01). Liver histopathology showed that the model group had marked inflammatory cell infiltration and formation of fibrous septa by proliferated collagen fibers, and the Xiayuxue decoction group had loose fibrous septa and alleviated inflammatory cell infiltration. Immunohistochemistry showed that compared with the control group, the model group had significant increases in the expression of α-SMA and GDNF (both P < 0.01), which were observed in fibrous septa, and compared with the model group, the Xiayuxue decoction group had significant reductions in the expression of α-SMA and GDNF (both P < 0.05). Western blotting showed that the control group had relatively low expression of GDNF in liver tissue, the formation of liver fibrosis at week 6 of CCl4 modeling, and an around 10-fold increase in the expression of GDNF, and the Xiayuxue decoction group had significantly inhibited protein expression of GDNF (P < 0.01); there were significant increases in the expression of α-SMA and collagen type I α1 (Col1) in mice with liver fibrosis, with significant reductions in α-SMA and Col1 after treatment with Xiayuxue decoction (all P < 0.01). The in vitro experiment showed that GDNF induced the significant increases in the protein expression of α-SMA and Col1 in HSCs, which was significantly inhibited by Xiayuxue decoction (all P < 0.01). Conclusion The expression of GDNF is significantly upregulated in the formation of liver fibrosis. GDNF can induce HSC activation, and Xiayuxue decoction can exert an anti-liver fibrosis effect by inhibiting GDNF. -
表 1 引物序列表
基因 上游 下游 18S rRNA 5′-AGTCCCTGCCCTTTGTACACA-3′ 5′-CGATCCGAGGGCCTCACTA-3′ Col4 A2 5′-TTATGCACTGCCTAAAGAGGAGC-3′ 5′-CCCTTAACTCCGTAGAAACCAAG-3′ Col5 A1 5′-GCCCGGATGTCGCTTACAG-3′ 5′-AAATGCAGACGCAGGGTACAG-3′ Col5 A2 5′-GACTGTGCCGACCCTGTAAC-3′ 5′-CCTGGACGACCACGTATGC-3′ 注: Col,Ⅰ型胶原α。 表 2 各组小鼠血清转氨酶水平
组别 动物数(只) ALT(U/L) AST(U/L) 对照组 8 26.51±5.70 19.07±6.22 模型组 8 141.79±39.861) 62.74±14.881) 下瘀血汤组 8 70.84±15.382) 29.48±7.252) F值 43.69 31.46 P值 <0.001 <0.001 注:与对照组比较, 1)P<0.001;与模型组比较, 2)P<0.01。 表 3 各组小鼠炎性浸润半定量及天狼星红阳性半定量结果
组别 动物数(只) 炎症细胞阳性面积(%) 天狼星红阳性面积(%) 对照组 8 0.94±0.09 0.73±0.07 模型组 8 5.55±0.521) 3.98±0.371) 下瘀血汤组 8 1.74±0.162) 1.82±0.172) F值 120.40 95.30 P值 0.001 4 0.001 9 注:与对照组比较, 1)P<0.01;与模型组比较, 2)P<0.01。 表 4 各组小鼠肝脏组织中α-SMA及GDNF含量半定量分析
组别 动物数(只) α-SMA阳性面积(%) GDNF阳性面积(%) 对照组 8 0.22±0.02 0.06±0.01 模型组 8 1.96±0.181) 1.66±0.151) 下瘀血汤组 8 0.90±0.082) 0.66±0.062) F值 111.50 141.60 P值 0.001 5 0.001 1 注:与对照组比较, 1)P<0.01;与模型组比较, 2)P<0.05。 表 5 各组小鼠肝脏组织中α-SMA及GDNF含量灰度值半定量分析
组别 动物数(只) GDNF灰度值 α-SMA灰度值 Col1灰度值 对照组 8 1.00±0.71 1.00±0.17 1.00±0.11 模型组 8 9.71±0.121) 9.45±0.121) 3.71±0.311) 下瘀血汤组 8 1.60±0.112) 3.90±0.052) 1.46±0.082) F值 269.50 2452.00 108.90 P值 0.000 4 <0.000 1 0.001 6 注:与对照组比较, 1)P<0.01;与模型组比较, 2)P<0.01。 表 6 体外实验中炎症因子及纤维化指标灰度值半定量分析
组别 pNF-κB TNFα α-SMA Col1 对照组 1.00±0.08 1.00±0.13 1.00±0.27 1.00±0.28 GDNF组 3.57±0.091) 2.94±0.071) 2.71±0.16 1) 4.89±0.141) GDNF+下淤血汤组 1.40±0.032) 1.57±0.042) 1.63±0.192) 2.95±0.212) F值 733.20 257.00 33.77 157.10 P值 <0.000 1 0.000 4 0.008 8 0.000 9 注:与对照组比较, 1)P<0.01;与GDNF组比较, 2)P<0.01。 表 7 体外实验中纤维化相关因子mRNA相对表达量比较
组别 Col4 A2 Col5 A1 Col5 A2 对照组 1.00±0.01 1.00±0.07 1.00±0.01 GDNF组 5.02±1.391) 4.10±0.491) 3.19±0.191) GDNF+下瘀血汤组 2.71±0.242) 1.46±0.352) 1.79±0.352) F值 12.24 45.63 46.57 P值 0.036 1 0.005 7 0.005 5 注:与对照组比较, 1)P<0.05;与GDNF组比较, 2)P<0.05。 -
[1] SCHUMACHER JD, KONG B, WU J, et al. Direct and indirect effects of Fibroblast Growth Factor (FGF)15 and FGF19 on liver fibrosis development[J]. Hepatology, 2020, 71(2): 670-685. DOI: 10.1002/hep.30810 [2] SHI N, CHEN XL, WU H, et al. Role of hepatic lymphangiogenesis in the progression of liver fibrosis[J]. J Clin Hepatol, 2020, 36(9): 2079-2082. (in Chinese. DOI: 10.3969/j.issn.1001-5256.2020.09.038石纳, 陈修利, 吴昊, 等. 肝脏淋巴管新生在肝纤维化进展中的作用[J]. 临床肝胆病杂志, 2020, 36(9): 2079-2082. DOI: 10.3969/j.issn.1001-5256.2020.09.038 [3] XIANG J, ZHANG N, SUN H, et al. Disruption of SIRT7 increases the efficacy of checkpoint inhibitor via MEF2D regulation of programmed cell death 1 ligand 1 in hepatocellular carcinoma cells[J]. Gastroenterology, 2020, 158(3): 664-678. e24. DOI: 10.1053/j.gastro.2019.10.025 [4] DONNELLY CR, SHAH AA, MISTRETTA CM, et al. Biphasic functions for the GDNF-Ret signaling pathway in chemosensory neuron development and diversification[J]. Proc Natl Acad Sci U S A, 2018, 115(3): e516-e525. DOI: 10.1073/pnas.1708838115 [5] TAO L, MA W, WU L, et al. Glial cell line-derived neurotrophic factor (GDNF) mediates hepatic stellate cell activation via ALK5/Smad signalling[J]. Gut, 2019, 68(12): 2214-2227. DOI: 10.1136/gutjnl-2018-317872 [6] TAO L, ZHANG J, YAN P, et al. Clinical observation of the effects of supplemented discharging blood stasis decoction on chronic viral hepatitis B[J]. Henan Tradit Chin Med, 2017, 37(11): 1946-1949. (in Chinese. https://www.cnki.com.cn/Article/CJFDTOTAL-HNZY201711026.htm陶乐, 张洁, 严萍, 等. 加味下瘀血汤治疗慢性乙型病毒性肝炎临床观察[J]. 河南中医, 2017, 37(11): 1946-1949. https://www.cnki.com.cn/Article/CJFDTOTAL-HNZY201711026.htm [7] LIU C, YUAN X, TAO L, et al. Xia-yu-xue decoction (XYXD) reduces carbon tetrachloride (CCl4)-induced liver fibrosis through inhibition hepatic stellate cell activation by targeting NF-κB and TGF-β1 signaling pathways[J]. BMC Complement Altern Med, 2015, 15: 201. DOI: 10.1186/s12906-015-0733-1 [8] MA W, TAO L, ZHANG W, et al. Xia-Yu-Xue decoction inhibits intestinal epithelial cell apoptosis in CCl4-induced liver fibrosis[J]. Cell Physiol Biochem, 2017, 44(1): 333-344. DOI: 10.1159/000484904 [9] LI Y, TANG L, GUO L, et al. CXCL13-mediated recruitment of intrahepatic CXCR5+CD8+ T cells favors viral control in chronic HBV infection[J]. J Hepatol, 2020, 72(3): 420-430. DOI: 10.1016/j.jhep.2019.09.031 [10] GONG H, LI LP. Research advances of portal vein thrombosis in nonalcoholic fatty liver disease[J]. J Clin Hepatol, 2020, 36(9): 2107-2110. (in Chinese. DOI: 10.3969/j.issn.1001-5256.2020.09.045龚航, 李良平. 非酒精性脂肪性肝病并发门静脉血栓的研究进展[J]. 临床肝胆病杂志, 2020, 36(9): 2107-2110. DOI: 10.3969/j.issn.1001-5256.2020.09.045 [11] CHU H, DUAN Y, LANG S, et al. The Candida albicans exotoxin candidalysin promotes alcohol-associated liver disease[J]. J Hepatol, 2020, 72(3): 391-400. DOI: 10.1016/j.jhep.2019.09.029 [12] SHE S, WU X, ZHENG D, et al. PSMP/MSMP promotes hepatic fibrosis through CCR2 and represents a novel therapeutic target[J]. J Hepatol, 2020, 72(3): 506-518. DOI: 10.1016/j.jhep.2019.09.033 [13] DAI KM. Jiang Chunhua's experience in using Xiayuxue decoction[J]. Shanxi J Tradit Chin Med, 2012, 28(1): 4-6. (in Chinese. https://www.cnki.com.cn/Article/CJFDTOTAL-SHIX201201003.htm戴克敏. 姜春华运用下瘀血汤的经验[J]. 山西中医, 2012, 28(1): 4-6. https://www.cnki.com.cn/Article/CJFDTOTAL-SHIX201201003.htm [14] LIU C, CAI J, CHENG Z, et al. Xiayuxue decoction reduces renal injury by promoting macrophage apoptosis in hepatic cirrhotic rats[J]. Genet Mol Res, 2015, 14(3): 10760-10773. DOI: 10.4238/2015.September.9.15 [15] WU L, ZHANG J, MA WT, et al. Xiayuxue decoction inhibits methionine-choline-deficient-induced nonalcoholic steatohepatitis in mice[J/CD]. Chin J Liver Dis (Electronic Version), 2018, 10(3): 48-55. (in Chinese.吴柳, 张洁, 马文婷, 等. 下瘀血汤对MCD诱导小鼠非酒精性脂肪性肝炎干预作用研究[J/CD]. 中国肝脏病杂志(电子版), 2018, 10(3): 48-55. [16] SHEN DX, MA WT, WU L, et al. Mechanism of Xiayuxue decoction on improving liver fibrosis by inhibiting pancreatic macrophage infiltration[J]. Acta Univ Tradit Med Sin Pharmacol Shanghai, 2019, 33(2): 73-80. (in Chinese. https://www.cnki.com.cn/Article/CJFDTOTAL-SHZD201902015.htm沈东晓, 马文婷, 吴柳, 等. 下瘀血汤抑制胰腺巨噬细胞浸润改善肝纤维化的机制研究[J]. 上海中医药大学学报, 2019, 33(2): 73-80. https://www.cnki.com.cn/Article/CJFDTOTAL-SHZD201902015.htm [17] NAHARI E, RAZI M. Silymarin amplifies apoptosis in ectopic endometrial tissue in rats with endometriosis; implication on growth factor GDNF, ERK1/2 and Bcl-6b expression[J]. Acta Histochem, 2018, 120(8): 757-767. DOI: 10.1016/j.acthis.2018.08.003 [18] KAWAMOTO K, YAGI M, STÖVER T, et al. Hearing and hair cells are protected by adenoviral gene therapy with TGF-beta1 and GDNF[J]. Mol Ther, 2003, 7(4): 484-492. DOI: 10.1016/S1525-0016(03)00058-3 [19] NENCINI S, RINGUET M, KIM DH, et al. GDNF, Neurturin, and Artemin activate and sensitize bone afferent neurons and contribute to inflammatory bone pain[J]. J Neurosci, 2018, 38(21): 4899-4911. DOI: 10.1523/JNEUROSCI.0421-18.2018 -



 PDF下载 ( 2815 KB)
PDF下载 ( 2815 KB)

 下载:
下载: