程序性细胞死亡受体1抑制剂联合仑伐替尼治疗晚期原发性肝癌的效果及不良反应
DOI: 10.3969/j.issn.1001-5256.2021.03.020
利益冲突声明:本研究不存在研究者、伦理委员会成员、受试者监护人以及与公开研究成果有关的利益冲突,特此声明。
作者贡献声明:滕颖、陈京龙负责课题设计,资料分析,撰写论文;滕颖、丁晓燕、李文东参与收集数据,修改论文;陈京龙负责拟定写作思路,指导撰写文章并最后定稿。
Clinical effect of programmed cell death-1 inhibitor combined with lenvatinib in treatment of advanced primary liver cancer and related adverse events
-
摘要:
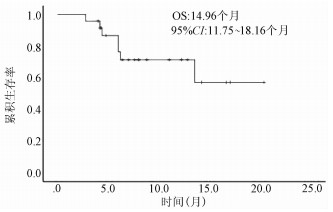
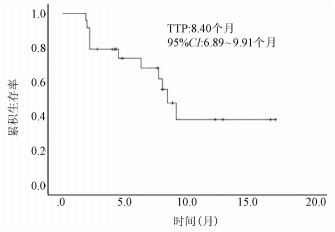
目的 初步探索国产程序性细胞死亡受体1(PD-1)抑制剂联合仑伐替尼在晚期原发性肝癌治疗中的临床疗效及不良反应。 方法 回顾性分析2019年1月1日—2020年4月2日于首都医科大学附属北京地坛医院使用国产PD-1抑制剂联合仑伐替尼治疗的24例晚期肝癌患者的临床资料,其中卡瑞利珠单抗联合仑伐替尼治疗组15例,信迪利单抗联合仑伐替尼治疗组7例,特瑞普利单抗联合仑伐替尼组2例。随访患者,肝内病灶使用mRECIST标准、肝外转移灶采用RECIST1.1标准评价疗效。Kaplan-Meier法绘制生存曲线。 结果 24例肝癌患者中,11例疗效评价为部分缓解,7例疗效评价为疾病稳定,6例疗效评价为疾病进展,客观缓解率为45.8%,疾病控制率为75.0%。中位疾病进展时间为8.40个月(95%CI:6.89~9.91个月)。不良反应发生率为54.17%,最常见的不良反应为疲乏(29.17%)、高血压(25.00%)。 结论 PD-1抑制剂联合仑伐替尼治疗晚期肝癌临床效果显著,严重不良反应发生率低,是一种安全、有效的治疗方案。 -
关键词:
- 肝肿瘤 /
- 程序性细胞死亡受体1 /
- 仑伐替尼 /
- 药物相关性副作用和不良反应
Abstract:Objective To investigate the clinical effect of domestic programmed cell death-1 (PD-1) inhibitor combined with lenvatinib in the treatment of advanced primary liver cancer and related adverse events. Methods A retrospective analysis was performed for the clinical data of 24 patients with advanced primary liver cancer who were treated with domestic PD-1 inhibitor combined with lenvatinib in Beijing Ditan Hospital, Capital Medical University, from January 1, 2019 to April 2, 2020, with 15 patients in the Camrelizumab+lenvatinib group, 7 patients in the Sintilimab+lenvatinib group, and 2 patients in the Toripalimab+lenvatinib group. During follow-up, Modified Response Evaluation Criteria in Solid Tumors was used to evaluate the treatment outcome of intrahepatic lesions, and RECIST1.1 was used to evaluate extrahepatic metastatic lesions. The Kaplan-Meier method was used to evaluate survival time. Results Among the 24 treatment-experienced patients, 11 achieved partial response, 7 achieved a stable disease, and 6 had disease progression, resulting in an objective response rate of 45.8% and a disease control rate of 75.0%. The median time to disease progression was 8.4 (95% confidence interval: 6.89-9.91) months. The incidence rate of adverse events was 54.17%, and the most common adverse events were fatigue (29.17%) and hypertension (25.00%). Conclusion PD-1 inhibitor combined with lenvatinib has a marked clinical effect in the treatment of advanced primary liver cancer, with a low incidence rate of serious adverse events, and thus it is a safe and effective treatment regimen. -
表 1 24例入组患者临床资料
项目 数值 年龄[例(%)] ≤50岁 8(33.3) >50岁 16(66.7) 性别[例(%)] 男 19(79.2) 女 5(20.8) 病毒感染[例(%)] HBV 19(79.2) HCV 2(8.3) 无 3(12.5) 肝硬化[例(%)] 有 17(70.8) 无 7(29.2) Child-Pugh分级[例(%)] A级 18(75.0) B级 6(25.0) AFP水平[例(%)] <200 ng/ml 14(58.3) ≥200 ng/ml 10(41.7) 门静脉癌栓[例(%)] Ⅰ 0 Ⅱ 8(33.3) Ⅲ 4(16.7) Ⅳ 3(12.5) 转移部位[例(%)] 淋巴结Ⅳ 12(50.0)Ⅳ 腹腔Ⅳ 5(20.8)Ⅳ 肺 3(12.5) 治疗情况[例(%)] 一线Ⅳ 11(45.8)Ⅳ 二线Ⅳ 11(45.8)Ⅳ 三线 (8.3) 表 2 24例入组患者不良反应情况
项目 合计[例(%)] 1级[例(%)] 2级[例(%)] 3级[例(%)] 食欲下降 3(12.50) 3(12.50) 体质量下降 1(4.17) 1(4.17) 疲乏 7(29.17) 4(16.67) 2(8.33) 1(4.17) 腹泻 3(12.50) 2(8.33) 1(4.17) 皮肤毛细血管增生症 1(4.17) 1(4.17) 皮疹 3(12.50) 1(4.17) 1(4.17) 1(4.17) 手足综合征 1(4.17) 1(4.17) 高血压 6(25.00) 1(4.17) 3(12.50) 2(8.33) 胆红素升高 1(4.17) 1(4.17) 尿蛋白 3(12.50) 3(12.50) 急性肾损伤 2(8.33) 1(4.17) 1(4.17) 肾上腺皮质功能减退 1(4.17) 1(4.17) 甲状腺功能减退 3(12.50) 2(8.33) 1(4.17) -
[1] BRAY F, FERLAY J, SOERJOMATARAM I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2018, 68(6): 394-424. DOI: 10.3322/caac.21492 [2] ZHENG R, QU C, ZHANG S, et al. Liver cancer incidence and mortality in China: Temporal trends and projections to 2030[J]. Chin J Cancer Res, 2018, 30(6): 571-579. DOI: 10.21147/j.issn.1000-9604.2018.06.01 [3] LLOVET JM, MONTAL R, SIA D, et al. Molecular therapies and precision medicine for hepatocellular carcinoma[J]. Nat Rev Clin Oncol, 2018, 15(10): 599-616. DOI: 10.1038/s41571-018-0073-4 [4] GRETEN TF, PAPENDORF F, BLECK JS, et al. Survival rate in patients with hepatocellular carcinoma: A retrospective analysis of 389 patients[J]. Br J Cancer, 2005, 92(10): 1862-1868. DOI: 10.1038/sj.bjc.6602590 [5] KUDO M, FINN RS, QIN S, et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: A randomised phase 3 non-inferiority trial[J]. Lancet, 2018, 391(10126): 1163-1173. DOI: 10.1016/S0140-6736(18)30207-1 [6] TOPALIAN SL, HODI FS, BRAHMER JR, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer[J]. N Engl J Med, 2012, 366(26): 2443-2454. DOI: 10.1056/NEJMoa1200690 [7] EL-KHOUEIRY AB, SANGRO B, YAU T, et al. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): An open-label, non-comparative, phase 1/2 dose escalation and expansion trial[J]. Lancet, 2017, 389(10088): 2492-2502. DOI: 10.1016/S0140-6736(17)31046-2 [8] ZHU AX, FINN RS, EDELINE J, et al. Pembrolizumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib (KEYNOTE-224): A non-randomised, open-label phase 2 trial[J]. Lancet Oncol, 2018, 19(7): 940-952. DOI: 10.1016/S1470-2045(18)30351-6 [9] FUKUMURA D, KLOEPPER J, AMOOZGAR Z, et al. Enhancing cancer immunotherapy using antiangiogenics: Opportunities and challenges[J]. Nat Rev Clin Oncol, 2018, 15(5): 325-340. DOI: 10.1038/nrclinonc.2018.29 [10] KATO Y, TABATA K, KIMURA T, et al. Lenvatinib plus anti-PD-1 antibody combination treatment activates CD8+ T cells through reduction of tumor-associated macrophage and activation of the interferon pathway[J]. PLoS One, 2019, 14(2): e0212513. DOI: 10.1371/journal.pone.0212513 [11] FINN RS, IKEDA M, ZHU AX, et al. Phase Ib study of lenvatinib plus pembrolizumab in patients with unresectable hepatocellular carcinoma[J]. J Clin Oncol, 2020, 38(26): 2960-2970. DOI: 10.1200/JCO.20.00808 [12] LENCIONI R, LLOVET JM. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma[J]. Semin Liver Dis, 2010, 30(1): 52-60. DOI: 10.1055/s-0030-1247132 [13] EISENHAUER EA, THERASSE P, BOGAERTS J, et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1)[J]. Eur J Cancer, 2009, 45(2): 228-247. DOI: 10.1016/j.ejca.2008.10.026 [14] WANG F, QIN S, SUN X, et al. Reactive cutaneous capillary endothelial proliferation in advanced hepatocellular carcinoma patients treated with camrelizumab: Data derived from a multicenter phase 2 trial[J]. J Hematol Oncol, 2020, 13(1): 47. DOI: 10.1186/s13045-020-00886-2 [15] HOOS A. Development of immuno-oncology drugs-from CTLA4 to PD1 to the next generations[J]. Nat Rev Drug Discov, 2016, 15(4): 235-247. DOI: 10.1038/nrd.2015.35 [16] SHEN H, YANG ES, CONRY M, et al. Predictive biomarkers for immune checkpoint blockade and opportunities for combination therapies[J]. Genes Dis, 2019, 6(3): 232-246. DOI: 10.1016/j.gendis.2019.06.006 [17] FINN RS, RYOO BY, MERLE P, et al. Pembrolizumab as second-line therapy in patients with advanced hepatocellular carcinoma in KEYNOTE-240: A randomized, double-blind, phase Ⅲ trial[J]. J Clin Oncol, 2020, 38(3): 193-202. DOI: 10.1200/JCO.19.01307 [18] RIVERA LB, MEYRONET D, HERVIEU V, et al. Intratumoral myeloid cells regulate responsiveness and resistance to antiangiogenic therapy[J]. Cell Rep, 2015, 11(4): 577-591. DOI: 10.1016/j.celrep.2015.03.055 [19] YASUDA S, SHO M, YAMATO I, et al. Simultaneous blockade of programmed death 1 and vascular endothelial growth factor receptor 2(VEGFR2) induces synergistic anti-tumour effect in vivo[J]. Clin Exp Immunol, 2013, 172(3): 500-506. DOI: 10.1111/cei.12069 [20] KIMURA T, KATO Y, OZAWA Y, et al. Immunomodulatory activity of lenvatinib contributes to antitumor activity in the Hepa1-6 hepatocellular carcinoma model[J]. Cancer Sci, 2018, 109(12): 3993-4002. DOI: 10.1111/cas.13806 [21] CHEN J, HU X, LI Q, et al. Effectiveness and safety of toripalimab, camrelizumab, and sintilimab in a real-world cohort of hepatitis B virus associated hepatocellular carcinoma patients[J]. Ann Transl Med, 2020, 8(18): 1187. DOI: 10.21037/atm-20-6063 -



 PDF下载 ( 1942 KB)
PDF下载 ( 1942 KB)

 下载:
下载: