mRECIST应答对经肝动脉化疗栓塞术治疗不可切除肝细胞癌患者预后的预测价值
DOI: 10.3969/j.issn.1001-5256.2021.03.022
Value of Modified Response Evaluation Criteria in Solid Tumors response in predicting the prognosis of patients with unresectable hepatocellular carcinoma patients after transarterial chemoembolization
-
摘要:
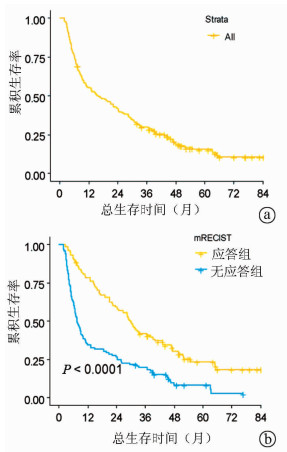
目的 探讨实体瘤疗效评价标准(mRECIST)应答对经肝动脉化疗栓塞术(TACE)治疗的不可切除肝细胞癌的预后价值。 方法 回顾性纳入2010年1月—2014年12月在空军军医大学第一附属医院消化介入科行TACE治疗连续的不可切除肝细胞癌患者190例。应用mRECIST标准评估TACE术后影像学应答,将肿瘤完全缓解(CR)和部分缓解(PR)患者定义为有应答组(n=89),将疾病进展(PD)和疾病稳定(SD)定义为无应答组(n=101)。采用Kaplan-Meier法计算中位生存时间,log-rank检验进行组间比较。利用Cox回归进行预后影响因素分析。 结果 根据mRECIST标准,CR、PR、SD和PD的患者分别有39(20.5%)例、50(26.3%)例、67(35.3%)例和34(17.9%)例。总人群的mRECIST客观应答率为46.8%。应答组的生存期明显高于无应答组,中位生存时间分别为29.9(95%CI:25.0~34.8)个月和7.5(95%CI:5.7~9.3)个月(P<0.001)。多因素分析结果显示,mRECIST应答(HR=2.02, P<0.001)、乙型肝炎(HR=4.03, P<0.001)和门静脉侵犯(HR=2.12, P=0.008)是影响患者生存的独立危险因素。 结论 mRECIST应答对经TACE治疗的不可切除肝细胞癌患者的预后具有一定的预测价值。 -
关键词:
- 癌,肝细胞 /
- 化学栓塞,治疗性 /
- 实体肿瘤疗效评价标准 /
- 预后
Abstract:Objective To investigate the association of Modified Response Evaluation Criteria in Solid Tumors (mRECIST) response with the prognosis of patients with unresectable hepatocellular carcinoma (HCC) after transarterial embolization (TACE). Methods A retrospective analysis was performed for the clinical data of 190 patients with unresectable HCC who were consecutively admitted to Department of Liver Disease and Digestive Interventional Radiology, The First Affiliated Hospital of Air Force Medical University, and treated with TACE from January 2010 to December 2014. The mRECIST criteria were used to evaluate imaging response after TACE; the patients with complete response (CR) or partial response (PR) were enrolled as response group (n=89), and those with progressive disease (PD) or stable disease (SD) were enrolled as non-response group (n=101). The Kaplan-Meier method was used to calculate median survival time, and the log-rank test was used for comparison between groups; the Cox regression model was used to identify the influencing factors for prognosis. Results According to the mRECIST criteria, 39 patients (20.5%) achieved CR, 50 (26.3%) achieved PR, 67 (35.3%) had SD, and 34 (17.9%) had PD. The objective response rate based on mRECIST was 46.8% for the whole population. The response group had a significantly longer survival time than the non-response group, and the median survival time was 29.9 (95% confidence interval [CI]: 25.0-34.8) months for the response group and 7.5 (95% CI: 5.7-9.3) months for the non-response group (P < 0.001). The multivariate analysis showed that mRECIST response (hazard ratio [HR]=2.02, P < 0.001), hepatitis B (HR=4.03, P < 0.001), and portal invasion (HR=2.12, P=0.008) were independent risk factors for survival. Conclusion The mRECIST response has a certain value in predicting the prognosis of patients with unresectable HCC after TACE. -
表 1 190例患者的基线特征(n=190)
指标 结果 年龄(岁) 52(45~62) 男/女(例) 155/35 病因(乙型肝炎/非乙型肝炎, 例) 175/15 ECOG评分(0/1, 例) 117/73 BCLC分期(A/AB/B/C,例) 22/49/33/86 肿瘤大小(cm,例) 8.3(5.5~11.5) 肿瘤数目(单个/多个,例) 122/68 门静脉侵犯(有/无,例) 41/149 AFP(≤400/>400 ng/ml,例) 89/101 血小板计数(×109/L) 123.5(83.8~176.3) 国际标准化比值 1.08(1.02~1.20) AST (U/L) 49(32~69) 白蛋白(g/L) 39.2(36.1~43.0) 胆红素(μmol/L) 15.8(11.1~21.9) 肌酐(μmol /L) 82(70~93) Child-Pugh评分(5/6,例) 144/46 ALBI分级(1/2,例) 90/100 mRECIST应答(CR/PR/SD/PD,例) 39/50/67/34 表 2 不同BCLC分期的影像学应答情况
应答情况 A期(n=22) AB期(n=49) B期(n=33) C期(n=86) ORR[例(%)] 20 (90.9) 25 (51.0) 15 (45.5) 29 (33.7) CR[例(%)] 10 (45.5) 13 (26.5) 5 (15.2) 11 (12.8) PR[例(%)] 10 (45.5) 12 (24.5) 10 (30.3) 18 (20.9) SD[例(%)] 1 (4.5) 20 (40.8) 10 (30.3) 36 (41.9) PD[例(%)] 1 (4.5) 4 (8.2) 8 (24.2) 21 (24.4) 表 3 Cox单因素及多因素回归分析
因素 单因素分析 多因素分析 HR (95%CI) P值 HR (95%CI) P值 男性(Ref: 女性) 1.15 (0.77~1.74) 0.491 年龄 0.98 (0.97~1.00) 0.012 0.99 (0.97~1.00) 0.173 病因(Ref: 非乙肝) 2.77 (1.36~5.66) 0.005 4.03 (1.87~8.71) <0.001 ECOG评分1分(Ref: 0分) 1.83 (1.33~2.51) <0.001 1.54 (0.73~3.23) 0.257 肿瘤大小 1.11 (1.07~1.16) <0.001 1.05 (0.99~1.11) 0.102 肿瘤数目 1.34 (1.17~1.52) <0.001 0.95 (0.80~1.13) 0.563 门静脉侵犯(Ref: 无) 4.30 (2.93~6.30) <0.001 2.12 (1.22~3.68) 0.008 Child-Pugh 6分(Ref: 5分) 1.28 (0.89~1.83) 0.176 ALBI 2级(Ref: 1级) 1.38 (1.01~1.90) 0.046 1.40 (1.00~1.97) 0.054 AFP (Ref: ≤400 ng/ml) 1.96 (1.42~2.70) <0.001 1.35 (0.95~1.93) 0.096 肌酐 0.99 (0.98~1.00) 0.052 1.00 (1.00~1.00) 0.960 白蛋白 0.97 (0.94~1.01) 0.104 胆红素 1.02 (0.99~1.04) 0.151 AST 1.01 (1.00~1.01) 0.001 1.00 (1.00~1.00) 0.986 血小板计数 1.00 (1.00~1.00) 0.856 国际标准化比值 2.23 (0.74~6.67) 0.152 BCLC分期(Ref: BCLC A) AB期 1.51 (0.80~2.85) 0.206 0.95 (0.46~1.96) 0.892 B期 2.49 (1.30~4.79) 0.006 1.73 (0.83~3.64) 0.145 C期 3.54 (1.96~6.39) <0.001 1.18 (0.45~3.12) 0.740 mRECIST应答情况(Ref: 应答) 2.33 (1.69~3.21) <0.001 2.02 (1.31~3.11) <0.001 -
[1] MARRERO JA, KULIK LM, SIRLIN C, et al. Diagnosis, staging, and management of hepatocellular carcinoma: 2018 practice guidance by the american association for the study of liver diseases[J]. Hepatology, 2018, 68(2): 723-50. DOI: 10.1002/hep.29913 [2] European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma[J]. J Hepatol, 2018, 69(1): 182-236. DOI: 10.1016/j.jhep.2018.03.019 [3] Bureau of Medical Administration, National Health Commission of the People's Republic of China. Guidelines for diagnosis and treatment of primary liver cancer in China (2019 edition)[J]. J Clin Hepatol, 2020, 36(2): 277-292. (in Chinese) DOI: 10.3969/j.issn.1001-5256.2020.02.007中华人民共和国国家卫生健康委员会医政医管局. 原发性肝癌诊疗规范(2019年版)[J]. 临床肝胆病杂志, 2020, 36(2): 277-292. DOI: 10.3969/j.issn.1001-5256.2020.02.007 [4] SIEGHART W, HUCKE F, PECK-RADOSAVLJEVIC M. Transarterial chemoembolization: Modalities, indication, and patient selection[J]. J Hepatol, 2015, 62(5): 1187-1195. DOI: 10.1016/j.jhep.2015.02.010 [5] LENCIONI R, LLOVET JM. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma[J]. Semin Liver Dis, 2010, 30(1): 52-60. DOI: 10.1055/s-0030-1247132 [6] GILLMORE R, STUART S, KIRKWOOD A, et al. EASL and mRECIST responses are independent prognostic factors for survival in hepatocellular cancer patients treated with transarterial embolization[J]. J Hepatol, 2011, 55(6): 1309-1316. DOI: 10.1016/j.jhep.2011.03.007 [7] JUNG ES, KIM JH, YOON EL, et al. Comparison of the methods for tumor response assessment in patients with hepatocellular carcinoma undergoing transarterial chemoembolization[J]. J Hepatol, 2013, 58(6): 1181-1187. DOI: 10.1016/j.jhep.2013.01.039 [8] KIM BK, KIM KA, PARK JY, et al. Prospective comparison of prognostic values of modified Response Evaluation Criteria in Solid Tumours with European Association for the Study of the Liver criteria in hepatocellular carcinoma following chemoembolisation[J]. Eur J Cancer, 2013, 49(4): 826-834. DOI: 10.1016/j.ejca.2012.08.022 [9] PRAJAPATI HJ, SPIVEY JR, HANISH SI, et al. mRECIST and EASL responses at early time point by contrast-enhanced dynamic MRI predict survival in patients with unresectable hepatocellular carcinoma (HCC) treated by doxorubicin drug-eluting beads transarterial chemoembolization (DEB TACE)[J]. Ann Oncol, 2013, 24(4): 965-973. DOI: 10.1093/annonc/mds605 [10] KIM CJ, KIM HJ, PARK JH, et al. Radiologic response to transcatheter hepatic arterial chemoembolization and clinical outcomes in patients with hepatocellular carcinoma[J]. Liver Int, 2014, 34(2): 305-312. DOI: 10.1111/liv.12270 [11] KIM BK, KIM SU, KIM KA, et al. Complete response at first chemoembolization is still the most robust predictor for favorable outcome in hepatocellular carcinoma[J]. J Hepatol, 2015, 62(6): 1304-1310. DOI: 10.1016/j.jhep.2015.01.022 [12] VINCENZI B, MAIO MD, SILLETTA M, et al. Prognostic relevance of objective response according to EASL Criteria and mRECIST Criteria in hepatocellular carcinoma patients treated with loco-regional therapies: A literature-based Meta-analysis[J]. Plos One, 2015, 10(7): 1-12. DOI: 10.1371/journal.pone.0133488 [13] TACHER V, LIN M, DURAN R, et al. Comparison of existing response criteria in patients with hepatocellular carcinoma treated with transarterial chemoembolization using a 3D quantitative approach[J]. Radiology, 2016, 278(1): 275-284. DOI: 10.1148/radiol.2015142951 [14] GEORGIADES C, GESCHWIND JF, HARRISON N, et al. Lack of response after initial chemoembolization for hepatocellular carcinoma: Does it predict failure of subsequent treatment?[J]. Radiology, 2012, 265(1): 115-123. DOI: 10.1148/radiol.12112264 [15] LIU L, WANG W, CHEN H, et al. EASL- and mRECIST-evaluated responses to combination therapy of sorafenib with transarterial chemoembolization predict survival in patients with hepatocellular carcinoma[J]. Clin Cancer Res, 2014, 20(6): 1623-1631. DOI: 10.1158/1078-0432.CCR-13-1716 [16] SHIM JH, LEE HC, KIM SO, et al. Which response criteria best help predict survival of patients with hepatocellular carcinoma following chemoembolization? A validation study of old and new models[J]. Radiology, 2012, 262(2): 708-718. DOI: 10.1148/radiol.11110282 [17] JUNG YK, JUNG CH, SEO YS, et al. BCLC stage B is a better designation for single large hepatocellular carcinoma than BCLC stage A[J]. J Gastroenterol Hepatol, 2016, 31(2): 467-474. DOI: 10.1111/jgh.13152 [18] ZHAO Y, WANG WJ, GUAN S, et al. Sorafenib combined with transarterial chemoembolization for the treatment of advanced hepatocellular carcinoma: A large-scale multicenter study of 222 patients[J]. Ann Oncol, 2013, 24(7): 1786-1792. DOI: 10.1093/annonc/mdt072 [19] WANG YY, ZHONG JH, XU HF, et al. A modified staging of early and intermediate hepatocellular carcinoma based on single tumour > 7 cm and multiple tumours beyond up-to-seven criteria[J]. Aliment Pharmacol Ther, 2019, 49(2): 202-210. DOI: 10.1111/apt.15074 -



 PDF下载 ( 1927 KB)
PDF下载 ( 1927 KB)

 下载:
下载:


