肝星状细胞在慢加急性肝衰竭小鼠模型发病进程中的作用及机制
DOI: 10.3969/j.issn.1001-5256.2021.03.027
利益冲突声明:本研究不存在研究者、伦理委员会成员、受试者监护人以及与公开研究成果有关的利益冲突,特此声明。
作者贡献说明:田臻负责课题设计、拟定写作思路;王丽莎、姚耐娟负责实验操作、论文撰写;田臻、王丽莎负责数据分析;田臻、赵英仁、阮骊涛负责论文审定。
Role and mechanism of hepatic stellate cells in the pathogenesis of mice with acute-on-chronic liver failure
-
摘要:
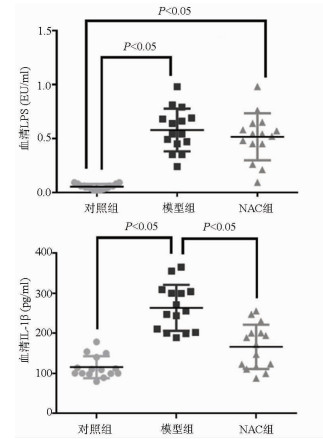
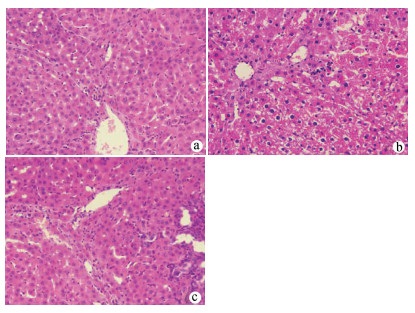
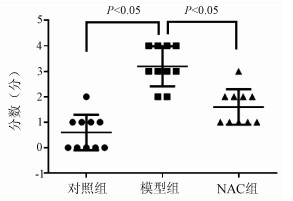
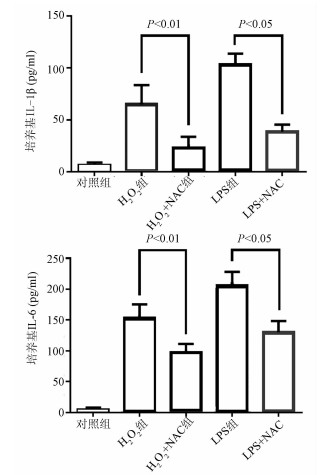
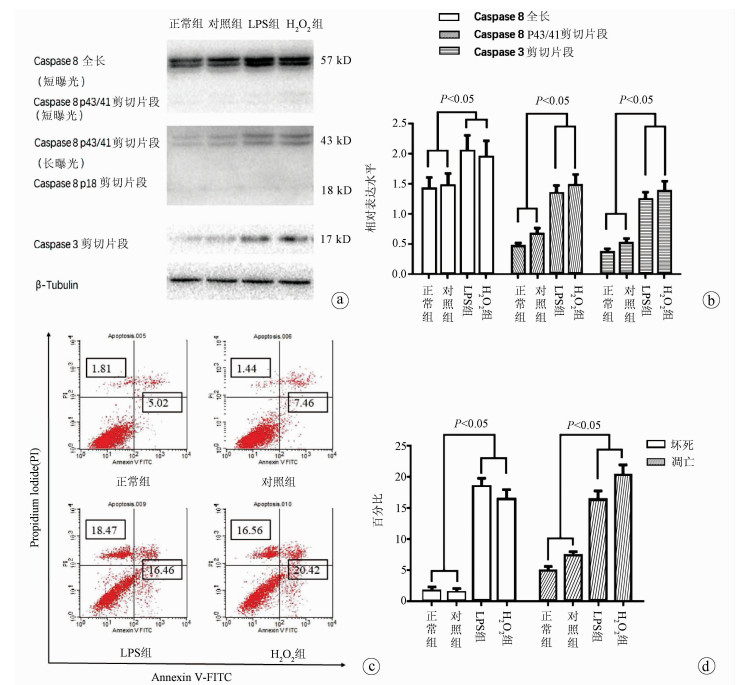
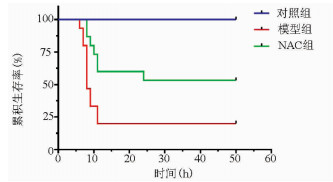
目的 探究肝星状细胞(HSC)炎症在慢加急性肝衰竭(ACLF)小鼠模型发病过程中的作用及机制。 方法 45只雄性昆明种小鼠随机分为对照组、模型组及N-乙酰-L-半胱氨酸(NAC)组,每组15只。模型组、NAC组注射人血白蛋白构建慢性肝病模型,之后腹腔注射内毒素(LPS)+D-GlaN诱导ACLF,对照组注射等量生理盐水, NAC组诱导ACLF前1周开始应用NAC。模型组及NAC组小鼠腹腔注射LPS+D-GlaN 48 h后处死。ELISA法检测血清AST、ALT及肝组织丙二醛及超氧化物歧化酶水平,肝组织行HE染色并进行病理评分,ELISA法检测小鼠血清LPS、IL-1β水平。在有或无NAC的情况下用LPS、H2O2刺激LX2细胞,ELISA法检测培养基中IL-1β及IL-6水平。用LPS、H2O2刺激LX2细胞,收取LX2培养基培养HL7702细胞,Western Blot检测HL7702细胞Caspase 8和Caspase 3表达,流式细胞法检测细胞凋亡。计量资料多组间比较采用单因素方差分析,方差齐时用LSD-t检验进行两两比较,方差不齐时采用Tamhane’s T2进行检验。Kaplan-Meier法绘制生存曲线,生存分析采用log-rank检验。 结果 48 h时对照组小鼠全部存活,模型组小鼠存活3只、NAC组存活8只。NAC组小鼠48 h累积生存率优于模型组(P<0.001);模型组血清AST、ALT及肝组织丙二醛水平较对照组和NAC组显著升高,肝组织超氧化物歧化酶水平显著降低(P值均<0.001);模型组小鼠肝脏病理评分明显高于对照组和NAC组(P值均<0.05)。LPS、H2O2均可促进LX2细胞产生IL-1β、IL-6,NAC能够有效抑制LPS、H2O2的促炎作用(P值均<0.05);H2O2、LPS作用于LX2细胞促进HL7702细胞凋亡(P值均<0.05)。 结论 LPS通过ROS促进HSC炎症,诱导肝细胞凋亡参与肝衰竭的疾病进程。 Abstract:Objective To investigate the role of hepatic stellate cell (HSC) inflammation in the pathogenesis of acute-on-chronic liver failure (ACLF). Methods A total of 45 male Kunming mice were randomly divided into control group, model group, and N-acetylcysteine (NAC) group. The mice in the model group and the NAC group were given injection of human serum albumin to establish a model of chronic liver disease, followed by intraperitoneal injection of the endotoxins lipopolysaccharide (LPS) and D-galactosamine (D-GlaN) to induce ACLF, and those in the control group were given injection of an equal volume of normal saline; the mice in the NAC group were given NAC since 1 week before the induction of NAC. The mice in the model group and the NAC group were sacrificed at 48 hours after the injection of LPS and D-GlaN. ELISA was used to measure the serum levels of aspartate aminotransferase (AST) and alanine aminotransferase (ALT) and the levels of malondialdehyde (MDA) and superoxide dismutase (SOD) in liver tissue; HE staining was used to determine liver pathological score; ELISA was used to measure the serum levels of LPS and interleukin-1β (IL-1β). LX2 cells were stimulated by LPS and H2O2 with the presence or absence of NAC, and ELISA was used to measure the levels of IL-1β and interleukin-6 (IL-6) in medium. LX2 cells were stimulated by LPS and H2O2, and then HL7702 cells were cultured with LX2 medium; Western blot was used to measure the expression of caspase-3 and caspase-8 in HL7702 cells, and flow cytometry was used to measure the apoptosis of HL7702 cells. A one-way analysis of variance was used for comparison of continuous data between multiple groups; the least significant difference t-test was used for comparison of data with homogeneity of variance between two groups, and the Tamhane's T2 test was used for comparison of data with heterogeneity of variance. The Kaplan-Meier survival analysis was used to evaluate survival time, and the log-rank test was used for comparison. Results At 48 hours, all mice in control group survived, while 3 mice in the model group and 8 mice in the NAC group survived, suggesting that the NAC group had a better survival rate of mice than the model group (P < 0.001). Compared with the control group and the NAC group, the model group had significant increases in the serum levels of AST and ALT and the level of MDA in liver tissue, as well as a significant reduction in the level of SOD in liver tissue (all P < 0.01). The model group had a significantly higher liver pathological score than the control group and the NAC group (both P < 0.05). Both LPS and H2O2 promoted the secretion of IL-1β and IL-6 in LX2 cells, and NAC effectively inhibited the pro-inflammatory effect of H2O2 and LPS (all P < 0.05). H2O2 and LPS acted on LX2 cells and promoted the apoptosis of HL7702 cells (all P < 0.05). Conclusion LPS can promote HSC inflammation via reactive oxygen species and participates in the progression of liver failure by inducing hepatocyte apoptosis. -
Key words:
- Acute-On-Chronic Liver Failure /
- Hepatic Stellate Cells /
- Inflammation /
- Apoptosis
-
表 1 各组小鼠血清ALT、AST及肝组织MDA和SOD水平
组别 小鼠数(只) AST(U/L) ALT(U/L) MDA(mmol/g) SOD(U/mg) 对照组 15 25.95±3.96 21.21±2.30 2529.21±143.32 152.16±17.13 模型组 15 6184.62±1123.121)2) 5012.41±988.351)2) 9721.34±215.381)2) 51.67±5.591)2) NAC组 15 965.51±106.011) 845.32±115.751) 4231.79±157.051) 83.34±7.561) F值 628.342 896.813 119.820 126.351 P值 <0.001 <0.001 <0.001 <0.001 注:与对照组相较,1)P<0.01;与NAC组相较,2)P<0.01。 -
[1] Liver Failure and Artificial Liver Group, Chinese Society of Infectious Diseases, Chinese Medical Association; Severe Liver Disease and Artificial Liver Group, Chinese Society of Hepatology, Chinese Medical Association. Guideline for diagnosis and treatment of liver failure(2018)[J]. J Clin Hepatol, 2019, 35(1): 38-44. (in Chinese)中华医学会感染病学分会肝衰竭与人工肝学组, 中华医学会肝脏病学分会重型肝病与人工肝学组. 肝衰竭诊治指南(2018年版)[J]. 临床肝胆病杂志, 2019, 35(1): 38-44. [2] ZHANG J, ZHOU XM. Value of different scoring systems in predicting short-term mortality of patients with acute-on-chronic liver failure[J]. J Clin Hepatol, 2019, 35(9): 1990-1994. (in Chinese) DOI: 10.3969/j.issn.1001-5256.2019.09.021张静, 周新民. 5种评分系统对慢加急性肝衰竭患者短期病死率的预测价值[J]. 临床肝胆病杂志, 2019, 35(9): 1990-1994. DOI: 10.3969/j.issn.1001-5256.2019.09.021 [3] MEHTA G, MOOKERJEE RP, SHARMA V, et al. Systemic inflammation is associated with increased intrahepatic resistance and mortality in alcohol-related acute-on-chronic liver failure[J]. Liver Int, 2015, 35(3): 724-34. DOI: 10.1111/liv.12559 [4] PAN C, GU Y, ZHANG W, et al. Dynamic changes of lipopolysaccharide levels in different phases of acute on chronic hepatitis B liver failure[J]. PLoS One, 2012, 7(11): e49460. DOI: 10.1371/journal.pone.0049460 [5] FUJITA T, SOONTRAPA K, ITO Y, et al. Hepatic stellate cells relay inflammation signaling from sinusoids to parenchyma in mouse models of immune-mediated hepatitis[J]. Hepatology, 2016, 63(4): 1325-1339. DOI: 10.1002/hep.28112 [6] LI J, ZHAO YR, TIAN Z. Roles of hepatic stellate cells in acute liver failure: From the perspective of inflammation and fibrosis[J]. World J Hepatol, 2019, 11(5): 412-420. DOI: 10.4254/wjh.v11.i5.412 [7] JIN L, GAO H, WANG J, et al. Role and regulation of autophagy and apoptosis by nitric oxide in hepatic stellate cells during acute liver failure[J]. Liver Int, 2017, 37(11): 1651-1659. DOI: 10.1111/liv.13476 [8] TIAN Z, CHEN Y, YAO N, et al. Role of mitophagy regulation by ROS in hepatic stellate cells during acute liver failure[J]. Am J Physiol Gastrointest Liver Physiol, 2018, 315(3): g374-g384. DOI: 10.1152/ajpgi.00032.2018 [9] DAS J, GHOSH J, MANNA P, et al. Acetaminophen induced acute liver failure via oxidative stress and JNK activation: Protective role of taurine by the suppression of cytochrome P450 2E1[J]. Free Radic Res, 2010, 44(3): 340-55. DOI: 10.3109/10715760903513017 [10] MONIAUX N, DARNAUD M, GARBIN K, et al. The reg3α (HIP/PAP) lectin suppresses extracellular oxidative stress in a murine model of acute liver failure[J]. PLoS One, 2015, 10(5): e0125584. DOI: 10.1371/journal.pone.0125584 [11] KINUGASA H, WHELAN KA, TANAKA K, et al. Mitochondrial SOD2 regulates epithelial-mesenchymal transition and cell populations defined by differential CD44 expression[J]. Oncogene, 2015, 34(41): 5229-39. DOI: 10.1038/onc.2014.449 [12] KIM SR, KIM DI, KIM SH, et al. NLRP3 inflammasome activation by mitochondrial ROS in bronchial epithelial cells is required for allergic inflammation[J]. Cell Death Dis, 2014, 5: e1498. DOI: 10.1038/cddis.2014.460 [13] GAO YD, TIAN Y, ZHANG XY, et al. Effect of magnesium isoglycyrrhizinate on concanavalin A-induced acute liver failure in mice[J]. J Clin Hepatol, 2020, 36(7): 1571-1576. (in Chinese) DOI: 10.3969/j.issn.1001-5256.2020.07.024高钰迪, 田原, 张向颖, 等. 异甘草酸镁对刀豆蛋白A诱导的急性肝衰竭小鼠模型的影响[J]. 临床肝胆病杂志, 2020, 36(7): 1571-1576. DOI: 10.3969/j.issn.1001-5256.2020.07.024 [14] EDWARDS S, LALOR PF, NASH GB, et al. Lymphocyte traffic through sinusoidal endothelial cells is regulated by hepatocytes[J]. Hepatology, 2005, 41(3): 451-459. DOI: 10.1002/hep.20585 [15] BIEGHS V, VERHEYEN F, van GORP PJ, et al. Internalization of modified lipids by CD36 and SR-A leads to hepatic inflammation and lysosomal cholesterol storage in Kupffer cells[J]. PLoS One, 2012, 7(3): e34378. DOI: 10.1371/journal.pone.0034378 [16] DECHÂNE A, SOWA JP, GIESELER RK, et al. Acute liver failure is associated with elevated liver stiffness and hepatic stellate cell activation[J]. Hepatology, 2010, 52(3): 1008-1016. DOI: 10.1002/hep.23754 -



 PDF下载 ( 4302 KB)
PDF下载 ( 4302 KB)

 下载:
下载: