急性胰腺炎初期全身炎症反应综合征持续时间与感染性胰腺坏死的关系
DOI: 10.3969/j.issn.1001-5256.2021.03.029
Association of the duration of systemic inflammatory response syndrome with infectious pancreatic necrosis at the initial stage of acute pancreatitis
-
摘要:
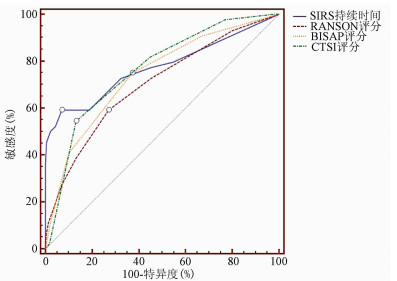
目的 探究急性胰腺炎 (AP) 患者早期炎症反应与晚期感染性胰腺坏死(IPN)的潜在关系。 方法 回顾性纳入西南医科大学附属医院2019年6月—2020年6月收治的中度重症急性胰腺炎(MSAP)患者219例,重症急性胰腺炎(SAP)患者53例,根据起病初期是否合并全身炎症反应综合征(SIRS),分为SIRS组160例,非SRIS组112例。纳入基线资料、血清学指标、并发症及病死率。计量资料两组间比较采用t检验或Mann-Whitney U检验;计数资料组间比较采用χ2检验,多组间进一步两两比较采用Bonferroni法。logistic回归分析筛选有价值的变量,受试者工作特征曲线(ROC曲线)用于比较变量的诊断价值,ROC曲线下面积(AUC)的两两比较采用Z检验。 结果 SIRS组患者的WBC、CRP、降钙素原更高(P值均<0.05),SIRS组合并急性胰周坏死物聚集(ANC)、IPN、胰腺坏死(PN)、器官功能障碍(OF)、多器官功能障碍(MODS)、重症急性胰腺炎(SAP)、危重症急性胰腺炎(CAP)、死亡、BISAP评分>2、CTSI评分>2、RANSON评分>2的比例均高于非SIRS组(P值均<0.05)。单因素分析显示,SIRS持续时间、肥胖、CRP、WBC、血尿素氮、PN、ANC、SAP、MODS、RANSON评分、BISAP评分、CTSI评分均是AP患者发生IPN的影响因素(P值均<0.05);多因素分析显示,SIRS持续时间(OR=1.307,95%CI:1.081~1.580,P=0.006)、ANC(OR=42.247,95%CI:10.829~164.818,P<0.001)是IPN的危险因素,排除ANC时,SIRS持续时间(OR=1.430,95%CI:1.207~1.694,P<0.001)、PN(OR=5.296,95%CI:1.845~15.203,P=0.002)是IPN的危险因素。预测IPN的ROC曲线显示:SIRS持续时间(AUC=0.772,约登指数:0.521)、RANSON评分(AUC=0.701,约登指数:0.319)、BISAP评分(AUC=0.741,约登指数:0.377)、CTSI评分(AUC=0.765,约登指数:0.414)的AUC两两比较差异均无统计学意义(P值均>0.05)。超长时间SIRS组(>4 d)患者发生PN、ANC、IPN、SAP、CAP的比例均显著高于无SIRS组(0)、短暂性SIRS组(1~2 d)和持续性SIRS组(3~4 d)(P值均<0.05),持续性SIRS组患者发生SAP的比例高于无SIRS组(P<0.05)。 结论 AP患者早期合并SIRS时更容易发生器官功能衰竭及局部并发症,当SIRS持续时间>4.5 d时,患者发生IPN的风险显著增加。 Abstract:Objective To investigate the potential association between early-stage inflammatory response and late-stage infectious pancreatic necrosis (IPN) in patients with acute pancreatitis (AP). Methods A retrospective analysis was performed for the clinical data of 219 patients with moderate-severe acute pancreatitis (MSAP) and 53 patients with severe acute pancreatitis (SAP) who were admitted to The Affiliated Hospital of Southwest Medical University from June 2019 to June 2020, and according to the presence or absence of systemic inflammatory response syndrome (SIRS) at the initial stage of the disease, they were divided into SIRS group with 160 patients and non-SRIS group with 112 patients. Baseline data, serological markers, complications, and mortality rate were included for analysis. The t-test or the Mann-Whitney U test was used for comparison of continuous data between two groups; the chi-square test was used for comparison of categorical data between multiple groups, and the Bonferroni method was used for further comparison between two groups. A logistic regression analysis was used to screen out valuable variables; the receiver operating characteristic (ROC) curve was used to compare the diagnostic value of variables, and the Z-test was used for pairwise comparison of area under the ROC curve (AUC). Results Compared with the non-SIRS group, the SIRS group had significantly higher white blood cell count (WBC), C-reactive protein (CRP), and procalcitonin (all P < 0.05) and a significantly higher proportion of patients with acute peripancreatic necrosis (ANC), IPN, pancreatic necrosis (PN), organ dysfunction, multiple organ dysfunction syndrome (MODS), SAP, critically-ill acute pancreatitis (CAP), death, BISAP score > 2, CTSI score > 2, or RANSON score > 2 (all P < 0.05). The univariate analysis showed that SIRS duration, obesity, CRP, WBC, blood urea nitrogen, PN, ANC, SAP, MODS, RANSON score, BISAP score, and CTSI score were risk factors for IPN in patients with AP (all P < 0.05), and the multivariate analysis showed that SIRS duration (odds ratio [OR]=1.307, 95% confidence interval [CI]: 1.081-1.580, P=0.006) and ANC (OR=42.247, 95% CI: 10.829-164.818, P < 0.001) were risk factors for IPN; when ANC was excluded, SIRS duration (OR=1.430, 95% CI: 1.207-1.694, P < 0.001) and PN (OR=5.296, 95% CI: 1.845-15.203, P=0.002) were risk factors for IPN. The ROC curve showed that SIRS duration (AUC=0.772, Youden index=0.521), RANSON score (AUC=0.701, Youden index=0.319), BISAP score (AUC=0.741, Youden index=0.377), and CTSI score (AUC=0.765, Youden Index=0.414) had a certain value in predicting IPN, and there was no significant difference in AUC between any two indices. The long-duration SIRS group (> 4 d) had a significantly higher proportion of patients with PN, ANC, IPN, SAP, or CAP than the non-SIRS group (0 d), the transient SIRS group (1~2 d), and the persistent SIRS group (3~4 d) (all P < 0.05), and the persistent SIRS group had a significantly higher proportion of patients with SAP than the non-SIRS group (P < 0.05). Conclusion AP patients with SIRS in the early stage are likely to develop organ failure and local complications, and there is a significant increase in the risk of IPN when SIRS duration is > 4.5 days. -
表 1 两组AP患者基线资料比较
项目 SIRS组(n=160) 非SIRS组(n=112) 统计值 P值 年龄(岁) 48(42~57) 48(42~54) Z=-0.343 0.731 男性[例(%)] 105(65.6) 67(59.8) χ2=0.954 0.329 病因[例(%)] χ2=1.067 0.785 胆源性 65(40.6) 51(45.5) 高脂血症性 64(40.0) 38(33.9) 酒精性 15(9.4) 11(9.8) 其他 16(10.0) 12(10.7) 肥胖[例(%)] 75(46.9) 45(40.2) χ2=1.198 0.274 吸烟史[例(%)] 64(40.0) 42(37.5) χ2=0.173 0.677 2型糖尿病[例(%)] 32(20.0) 21(18.8) χ2=0.066 0.798 高血压[例(%)] 51(31.9) 25(22.3) χ2=2.987 0.084 表 2 两组AP患者临床资料比较
项目 SIRS组(n=160) 非SIRS组(n=112) 统计值 P值 血清学指标 WBC(109/L) 14.88(12.25~18.68) 11.65(9.66~16.37) Z=-4.315 <0.001 CRP (mg/L) 99.68(36.02~173.00) 47.21(7.62~104.64) Z=-3.973 <0.001 PCT (ng/L) 0.35(0.13~1.46) 0.13(0.05~0.40) Z=-4.447 <0.001 BUN(mmol/L) 5.17(3.81~7.24) 5.16(3.88~6.49) Z=-0.570 0.569 并发症[例(%)] APFC 141(88.1) 105(93.8) χ2=2.411 0.120 ANC 48(30.0) 20(17.9) χ2=5.181 0.023 IPN 35(21.9) 9(8.0) χ2=9.306 0.002 PN 50(31.3) 12(10.7) χ2=18.788 <0.001 OF 45(28.1) 8(7.1) χ2=18.488 <0.001 MODS 39(24.4) 4(3.6) χ2=21.423 <0.001 疾病严重程度[例(%)] SAP 45(28.1) 8(7.1) χ2=10.210 0.001 CAP 19(11.9) 1(0.9) χ2=11.664 0.001 死亡 10(6.3) 1(0.9) χ2=4.872 0.027 AP相关评分[例(%)] BISAP评分>2 40(25.0) 1(0.9) χ2=29.909 <0.001 CTSI评分>2 140(87.5) 80(71.4) χ2=11.005 0.001 RANSON评分>2 75(46.9) 13(11.6) χ2=37.422 <0.001 表 3 IPN的单因素回归分析
项目 B值 SE Wald P值 OR 95%CI SIRS持续时间 0.429 0.067 40.674 <0.001 1.535 1.346~1.751 胆源性AP(是=1,否=0) 0.573 0.331 2.990 0.084 1.774 0.926~3.397 肥胖(是=1,否=0) 0.950 0.341 7.759 0.005 2.587 1.325~5.409 PCT 0.013 0.007 3.121 0.077 1.013 0.999~1.207 CRP 0.005 0.002 4.437 0.035 1.005 1.000~1.009 WBC 0.152 0.034 20.589 <0.001 1.164 1.090~1.244 BUN 0.083 0.040 4.411 0.036 1.087 1.006~1.174 SAP(是=1,否=0) 1.594 0.357 19.995 <0.001 4.924 2.448~9.904 APFC(是=1,否=0) 1.667 1.034 2.601 0.107 5.296 0.699~40.147 PN(是=1,否=0) 2.436 0.370 43.438 <0.001 11.424 5.537~23.572 ANC(是=1,否=0) 4.622 0.632 53.452 <0.001 101.741 29.466~351.289 MODS(是=1,否=0) 1.866 0.373 25.004 <0.001 6.460 3.109~13.422 BISAP评分 0.828 0.163 25.831 <0.001 2.288 1.663~3.149 CTSI评分 0.510 0.098 27.365 <0.001 1.665 1.376~2.016 RANSON评分 0.402 0.087 21.418 <0.001 1.494 1.261~1.772 表 4 IPN的多因素回归分析
项目 B值 SE Wald P值 OR 95%CI 回归方程1 SIRS持续时间 0.267 0.097 7.613 0.006 1.307 1.081~1.580 ANC(是=1,否=0) 3.744 0.695 29.050 <0.001 42.247 10.829~164.818 回归方程2 SIRS持续时间 0.358 0.086 17.135 <0.001 1.430 1.207~1.694 WBC 0.023 0.013 3.238 0.072 1.024 0.998~1.050 PN(是=1,否=0) 1.667 0.538 9.600 0.002 5.296 1.845~15.203 表 5 预测IPN的ROC曲线结果
项目 AUC P值 敏感度(%) 特异度(%) 约登指数 95%CI cut-off值 SIRS持续时间 0.772 <0.001 59.09 92.98 0.521 0.718~0.821 4.5 BISAP评分 0.741 <0.001 75.00 62.72 0.377 0.685~0.792 1.5 RANSON评分 0.701 <0.001 59.09 72.81 0.319 0.643~0.755 2.5 CTSI评分 0.765 <0.001 54.55 86.84 0.414 0.710~0.810 7.0 表 6 根据SIRS持续时间分组比较
项目 无SIRS组(n=112) 短暂性SIRS组(n=54) 持续性SIRS组(n=64) 超长时间SIRS组(n=42) χ2值 P值 PN[例(%)] 12(10.7)1) 6(11.1)1) 13(20.3)1) 31(73.8) 75.811 <0.001 ANC[例(%)] 20(17.9)1) 4(7.4)1) 11(17.2)1) 33(78.6) 78.330 <0.001 IPN[例(%)] 9(8.0)1) 3(5.6)1) 6(9.4)1) 26(61.9) 76.919 <0.001 SAP[例(%)] 8(7.1)1)2) 6(11.1)1) 15(23.4)1) 24(57.1) 51.890 <0.001 CAP[例(%)] 1(0.9)1) 1(1.9)1) 2(3.1)1) 16(38.1) 69.207 <0.001 注:与超长时间SIRS组比较,1)P<0.05;与持续性SIRS组比较,2)P<0.05。 -
[1] Pancreas Study Group, Chinese Society of Gastroenterology, Chinese Medical Association; Editorial Board of Chinese Journal of Pancreatology; Editorial Board of Chinese Journal of Digestion. Chinese guidelines for the management of acute pancreatitis (Shenyang, 2019)[J]. J Clin Hepatol, 2019, 35(12): 2706-2711. (in Chinese)中华医学会消化病学分会胰腺疾病学组, 《中华胰腺病杂志》编委会, 《中华消化杂志》编委会. 中国急性胰腺炎诊治指南(2019年, 沈阳)[J]. 临床肝胆病杂志, 2019, 35(12): 2706-2711. [2] LEPPÄNIEMI A, TOLONEN M, TARASCONI A, et al. 2019 WSES guidelines for the management of severe acute pancreatitis[J]. World J Emerg Surg, 2019, 14: 27. DOI: 10.1186/s13017-019-0247-0 [3] TAN C, YANG L, SHI F, et al. Early systemic inflammatory response syndrome duration predicts infected pancreatic necrosis[J]. J Gastrointest Surg, 2020, 24(3): 590-597. DOI: 10.1007/s11605-019-04149-5 [4] SILVA-VAZ P, ABRANTES AM, MORGADO-NUNES S, et al. Evaluation of prognostic factors of severity in acute biliary pancreatitis[J]. Int J Mol Sci, 2020, 21(12) : 4300. DOI: 10.3390/ijms21124300 [5] VENKATESH NR, VIJAYAKUMAR C, BALASUBRAMANIYAN G, et al. Comparison of different scoring systems in predicting the severity of acute pancreatitis: A prospective observational study[J]. Cureus, 2020, 12(2): e6943. http://www.researchgate.net/publication/339176836_Comparison_of_Different_Scoring_Systems_in_Predicting_the_Severity_of_Acute_Pancreatitis_A_Prospective_Observational_Study [6] SOULOUNTSI V, SCHIZODIMOS T. Use of antibiotics in acute pancreatitis: Ten major concerns[J]. Scand J Gastroenterol, 2020, 55(10): 1211-1218. DOI: 10.1080/00365521.2020.1804995 [7] CASE BM, JENSEN KK, BAKIS G, et al. Endoscopic interventions in acute pancreatitis: What the advanced endoscopist wants to know[J]. Radiographics, 2018, 38(7): 2002-2018. DOI: 10.1148/rg.2018180066 [8] ARVANITAKIS M, DUMONCEAU JM, ALBERT J, et al. Endoscopic management of acute necrotizing pancreatitis: European Society of Gastrointestinal Endoscopy (ESGE) evidence-based multidisciplinary guidelines[J]. Endoscopy, 2018, 50(5): 524-546. DOI: 10.1055/a-0588-5365 [9] TRIKUDANATHAN G, WOLBRINK D, van SANTVOORT HC, et al. Current concepts in severe acute and necrotizing pancreatitis: An evidence-based approach[J]. Gastroenterology, 2019, 156(7): 1994-2007. DOI: 10.1053/j.gastro.2019.01.269 [10] SHARMA D, JAKKAMPUDI A, REDDY R, et al. Association of systemic inflammatory and anti-inflammatory responses with adverse outcomes in acute pancreatitis: Preliminary results of an ongoing study[J]. Dig Dis Sci, 2017, 62(12): 3468-3478. DOI: 10.1007/s10620-017-4813-6 [11] SENDLER M, van den BRANDT C, GLAUBITZ J, et al. NLRP3 inflammasome regulates development of systemic inflammatory response and compensatory anti-inflammatory response syndromes in mice with acute pancreatitis[J]. Gastroenterology, 2020, 158(1): 253-269. DOI: 10.1053/j.gastro.2019.09.040 [12] YANG R, TENHUNEN J, TONNESSEN TI. HMGB1 and histones play a significant role in inducing systemic inflammation and multiple organ dysfunctions in severe acute pancreatitis[J]. Int J Inflam, 2017, 2017: 1817564. http://www.ncbi.nlm.nih.gov/pubmed/28316860 [13] CHEN Z, DONG WH, WU Q, et al. Two-layer regulation of TRAF6 mediated by both TLR4/NF-kB signaling and miR-589-5p increases proinflammatory cytokines in the pathology of severe acute pancreatitis[J]. Am J Transl Res, 2020, 12(6): 2379-2395. http://www.zhangqiaokeyan.com/academic-journal-foreign-pmc_american-journal-translational-research_thesis/040006415721.html [14] IYER S, BAWA EP, TARIQUE M, et al. Know thy enemy-understanding the role of inflammation in severe acute pancreatitis[J]. Gastroenterology, 2020, 158(1): 46-48. DOI: 10.1053/j.gastro.2019.11.039 [15] DIXIT A, CHEEMA H, GEORGE J, et al. Extracellular release of ATP promotes systemic inflammation during acute pancreatitis[J]. Am J Physiol Gastrointest Liver Physiol, 2019, 317(4): g463-g475. DOI: 10.1152/ajpgi.00395.2018 [16] DUMNICKA P, MADUZIA D, CERANOWICZ P, et al. The interplay between inflflammation, coagulation and endothelial injury in the early phase of acute pancreatitis: Clinical implications[J]. Int J Mol Sci, 2017, 18(2): 354. DOI: 10.3390/ijms18020354 [17] GAD MM, SIMONS-LINARES CR. Is aggressive intravenous fluid resuscitation beneficial in acute pancreatitis? A meta-analysis of randomized control trials and cohort studies[J]. World J Gastroenterol, 2020, 26(10): 1098-1106. DOI: 10.3748/wjg.v26.i10.1098 [18] YE B, MAO W, CHEN Y, et al. Aggressive resuscitation is associated with the development of acute kidney injury in acute pancreatitis[J]. Dig Dis Sci, 2019, 64(2): 544-552. DOI: 10.1007/s10620-018-5328-5 [19] CHOOSAKUL S, HARINWAN K, CHIRAPONGSATHORN S, et al. Comparison of normal saline versus Lactated Ringer's solution for fluid resuscitation in patients with mild acute pancreatitis, A randomized controlled trial[J]. Pancreatology, 2018, 18(5): 507-512. DOI: 10.1016/j.pan.2018.04.016 [20] FROGHI F, SOGGIU F, RICCIARDI F, et al. Ward-based goal-directed fluid therapy (GDFT) in acute pancreatitis (GAP) trial: Study protocol for a feasibility randomised controlled trial[J]. BMJ Open, 2019, 9(10): e028783. DOI: 10.1136/bmjopen-2018-028783 [21] WOLBRINK D, GRUNDSELL JR, WITTEMAN B, et al. Are omega-3 fatty acids safe and effective in acute pancreatitis or sepsis? A systematic review and meta-analysis[J]. Clin Nutr, 2020, 39(9): 2686-2694. DOI: 10.1016/j.clnu.2019.12.006 [22] RUIZ-REBOLLO ML, MUÑOZ-MORENO MF, MAYO-ISCAR A, et al. Statin intake can decrease acute pancreatitis severit[J]. Pancreatology, 2019, 19(6): 807-812. DOI: 10.1016/j.pan.2019.07.004 [23] JIANG X, SHI JY, WANG XY, et al. The impacts of infectious complications on outcomes in acute pancreatitis: A retrospective study[J]. Mil Med Res, 2020, 7(1): 38. http://www.researchgate.net/publication/343685598_The_impacts_of_infectious_complications_on_outcomes_in_acute_pancreatitis_a_retrospective_study [24] TIAN H, CHEN L, WU X, et al. Infectious complications in severe acute pancreatitis: Pathogens, drug resistance, and status of nosocomial infection in a university-affiliated teaching hospital[J]. Dig Dis Sci, 2020, 65(7): 2079-2088. DOI: 10.1007/s10620-019-05924-9 [25] BARON TH, DIMAIO CJ, WANG AY, et al. American Gastroenterological association clinical practice update: Management of pancreatic necrosis[J]. Gastroenterology, 2020, 158(1): 67-75. DOI: 10.1053/j.gastro.2019.07.064 [26] SUSAK YM, DIRDA OO, FEDORCHUK OG, et al. Infectious complications of acute pancreatitis is associated with peripheral blood phagocyte functional exhaustion[J]. Dig Dis Sci, 2020.[Online ahead of print] [27] LU JD, CAO F, DING YX, et al. Timing, distribution, and microbiology of infectious complications after necrotizing pancreatitis[J]. World J Gastroenterol, 2019, 25(34): 5162-5173. DOI: 10.3748/wjg.v25.i34.5162 [28] AUSANIA F, SENRA DEL RÍO P, BORIN A, et al. Factors associated with mortality in patients with infected pancreatic necrosis: The "surgery effect"[J]. Updates Surg, 2020, 72(4): 1097-1103. DOI: 10.1007/s13304-020-00764-z [29] ZHANG ZH, DING YX, WU YD, et al. A meta-analysis and systematic review of percutaneous catheter drainage in treating infected pancreatitis necrosis[J]. Medicine (Baltimore), 2018, 97(47): e12999. DOI: 10.1097/MD.0000000000012999 [30] DARRIVERE L, LAPIDUS N, COLIGNON N, et al. Minimally invasive drainage in critically ill patients with severe necrotizing pancreatitis is associated with better outcomes: An observational study[J]. Crit Care, 2018, 22(1): 321. DOI: 10.1186/s13054-018-2256-x [31] SUN B, LI GQ. Surgical intervention strategies for local complications of severe acute pancreatitis[J]. Chin J Dig Surg, 2020, 19(4): 379-383. (in Chinese) -



 PDF下载 ( 2124 KB)
PDF下载 ( 2124 KB)

 下载:
下载:


