支链氨基酸在非酒精性脂肪性肝病发生发展中的作用
DOI: 10.3969/j.issn.1001-5256.2022.02.037
利益冲突声明:所有作者均声明不存在利益冲突。
作者贡献声明:谢小青负责课题设计,撰写论文;刘亚贤、陈顺参与修改论文;于晓辉负责拟定写作思路,指导撰写文章并最后定稿。
Role of branched-chain amino acids in the development and progression of nonalcoholic fatty liver disease
-
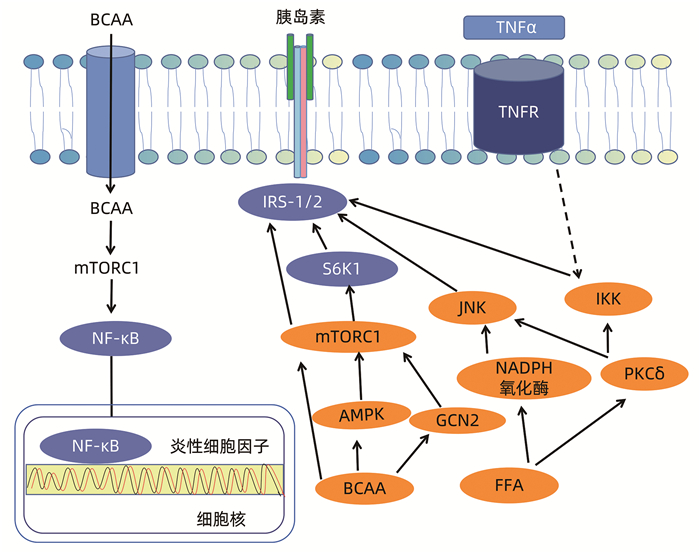
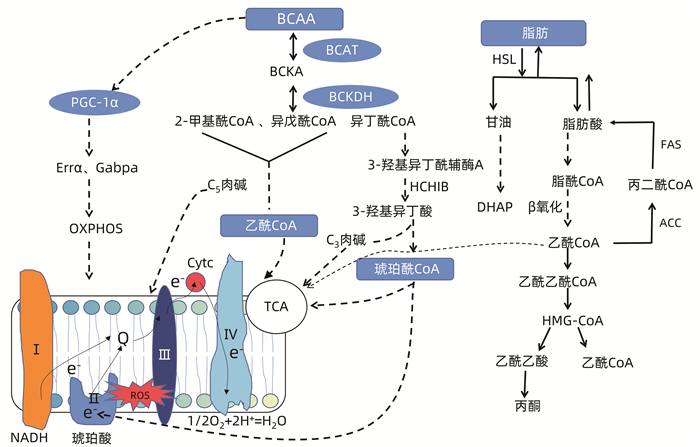
摘要: 非酒精性脂肪性肝病是常见慢性肝病,有进展为非酒精性肝炎、肝纤维化和肝癌的风险,发病机制多样,其中支链氨基酸代谢异常可引起肝细胞氧化应激、自噬、线粒体功能障碍等,是导致非酒精性脂肪性肝病发生发展的最主要机制,对其进展进行综述,分析支链氨基酸代谢异常对非酒精性脂肪性肝病发生与发展的可能作用,以期提高临床认知与诊治水平。Abstract: Nonalcoholic fatty liver disease is a common chronic liver disease with the risk of progression to nonalcoholic hepatitis, liver fibrosis, and hepatocellular carcinoma. Nonalcoholic fatty liver disease has various pathogeneses, among which abnormal metabolism of branched-chain amino acids can induce oxidative stress, autophagy, and mitochondrial dysfunction in hepatocytes and is the most important mechanism in the development and progression of nonalcoholic fatty liver disease. This article reviews related research advances and analyzes the possible role of abnormal metabolism of branched-chain amino acids in the development and progression of nonalcoholic fatty liver disease, in order to improve clinical awareness and diagnosis.
-
-
[1] YOUNOSSI ZM, KOENIG AB, ABDELATIF D, et al. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes[J]. Hepatology, 2016, 64(1): 73-84. DOI: 10.1002/hep.28431. [2] CHOLANKERIL G, PERUMPAIL RB, PHAM EA, et al. Nonalcoholic fatty liver disease: Epidemiology, natural history, and diagnostic challenges[J]. Hepatology, 2016, 64(3): 954. DOI: 10.1002/hep.28719. [3] European Association for the Study of the Liver (EASL), European Association for the Study of Diabetes (EASD), European Association for the Study of Obesity (EASO). EASL-EASD-EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease[J]. J Hepatol, 2016, 64(6): 1388-1402. DOI: 10.1016/j.jhep.2015.11.004. [4] NEINAST M, MURASHIGE D, ARANY Z. Branched chain amino acids[J]. Annu Rev Physiol, 2019, 81: 139-164. DOI: 10.1146/annurev-physiol-020518-114455. [5] SHIHA G, ALSWAT K, AL KHATRY M, et al. Nomenclature and definition of metabolic-associated fatty liver disease: A consensus from the Middle East and north Africa[J]. Lancet Gastroenterol Hepatol, 2021, 6(1): 57-64. DOI: 10.1016/S2468-1253(20)30213-2. [6] ESLAM M, GEORGE J. Reply to: Correspondence on "A new definition for metabolic associated fatty liver disease: An international expert consensus statement": MAFLD: Moving from a concept to practice[J]. J Hepatol, 2020, 73(5): 1268-1269. DOI: 10.1016/j.jhep.2020.06.036. [7] ZHENYUKH O, CIVANTOS E, RUIZ-ORTEGA M, et al. High concentration of branched-chain amino acids promotes oxidative stress, inflammation and migration of human peripheral blood mononuclear cells via mTORC1 activation[J]. Free Radic Biol Med, 2017, 104: 165-177. DOI: 10.1016/j.freeradbiomed.2017.01.009. [8] ZHENYUKH O, GONZÁLEZ-AMOR M, RODRIGUES-DIEZ RR, et al. Branched-chain amino acids promote endothelial dysfunction through increased reactive oxygen species generation and inflammation[J]. J Cell Mol Med, 2018, 22(10): 4948-4962. DOI: 10.1111/jcmm.13759. [9] SOLON-BIET SM, COGGER VC, PULPITEL T, et al. Branched chain amino acids impact health and lifespan indirectly via amino acid balance and appetite control[J]. Nat Metab, 2019, 1(5): 532-545. DOI: 10.1038/s42255-019-0059-2. [10] KIM SJ, TANG T, ABBOTT M, et al. AMPK phosphorylates desnutrin/ATGL and hormone-sensitive lipase to regulate lipolysis and fatty acid oxidation within adipose tissue[J]. Mol Cell Biol, 2016, 36(14): 1961-1976. DOI: 10.1128/MCB.00244-16. [11] ZHANG F, ZHAO S, YAN W, et al. Branched chain amino acids cause liver injury in obese/diabetic mice by promoting adipocyte lipolysis and inhibiting hepatic autophagy[J]. EBioMedicine, 2016, 13: 157-167. DOI: 10.1016/j.ebiom.2016.10.013. [12] GALARREGUI C, CANTERO I, MARIN-ALEJANDRE BA, et al. Dietary intake of specific amino acids and liver status in subjects with nonalcoholic fatty liver disease: Fatty liver in obesity (FLiO) study[J]. Eur J Nutr, 2021, 60(4): 1769-1780. DOI: 10.1007/s00394-020-02370-6. [13] TANAKA H, FUKAHORI S, BABA S, et al. Branched-chain amino acid-rich supplements containing microelements have antioxidant effects on nonalcoholic steatohepatitis in mice[J]. JPEN J Parenter Enteral Nutr, 2016, 40(4): 519-528. DOI: 10.1177/0148607114555160. [14] de PASQUALE V, CATERINO M, COSTANZO M, et al. Targeted metabolomic analysis of a mucopolysaccharidosis IIIB mouse model reveals an imbalance of branched-chain amino acid and fatty acid metabolism[J]. Int J Mol Sci, 2020, 21(12): 4211. DOI: 10.3390/ijms21124211. [15] YAN X, SUN Q, JI J, et al. Reconstitution of leucine-mediated autophagy via the mTORC1-Barkor pathway in vitro[J]. Autophagy, 2012, 8(2): 213-221. DOI: 10.4161/auto.8.2.18563. [16] SON SM, PARK SJ, LEE H, et al. Leucine signals to mTORC1 via its metabolite Acetyl-coenzyme A[J]. Cell Metab, 2019, 29(1): 192-201.e7. DOI: 10.1016/j.cmet.2018.08.013. [17] ZHENG R, HUANG S, ZHU J, et al. Leucine attenuates muscle atrophy and autophagosome formation by activating PI3K/AKT/mTOR signaling pathway in rotator cuff tears[J]. Cell Tissue Res, 2019, 378(1): 113-125. DOI: 10.1007/s00441-019-03021-x. [18] WU H, DRIDI S, HUANG Y, et al. Leucine decreases intramyocellular lipid deposition in an mTORC1-independent manner in palmitate-treated C2C12 myotubes[J]. Am J Physiol Endocrinol Metab, 2020, 318(2): E152-E163. DOI: 10.1152/ajpendo.00241.2019. [19] PENG KY, WATT MJ, RENSEN S, et al. Mitochondrial dysfunction-related lipid changes occur in nonalcoholic fatty liver disease progression[J]. J Lipid Res, 2018, 59(10): 1977-1986. DOI: 10.1194/jlr.M085613. [20] VOS M, GEENS A, BÖHM C, et al. Cardiolipin promotes electron transport between ubiquinone and complex I to rescue PINK1 deficiency[J]. J Cell Biol, 2017, 216(3): 695-708. DOI: 10.1083/jcb.201511044. [21] PÉREZ-CARRERAS M, DEL HOYO P, MARTÍN MA, et al. Defective hepatic mitochondrial respiratory chain in patients with nonalcoholic steatohepatitis[J]. Hepatology, 2003, 38(4): 999-1007. DOI: 10.1053/jhep.2003.50398. [22] PATTERSON RE, KALAVALAPALLI S, WILLIAMS CM, et al. Lipotoxicity in steatohepatitis occurs despite an increase in tricarboxylic acid cycle activity[J]. Am J Physiol Endocrinol Metab, 2016, 310(7): E484-E494. DOI: 10.1152/ajpendo.00492.2015. [23] WANG J, LIU Y, LIAN K, et al. BCAA catabolic defect alters glucose metabolism in lean mice[J]. Front Physiol, 2019, 10: 1140. DOI: 10.3389/fphys.2019.01140. [24] CHENG S, WIKLUND P, AUTIO R, et al. Adipose tissue dysfunction and altered systemic amino acid metabolism are associated with non-alcoholic fatty liver disease[J]. PLoS One, 2015, 10(10): e0138889. DOI: 10.1371/journal.pone.0138889. [25] MUYYARIKKANDY MS, MCLEOD M, MAGUIRE M, et al. Branched chain amino acids and carbohydrate restriction exacerbate ketogenesis and hepatic mitochondrial oxidative dysfunction during NAFLD[J]. FASEB J, 2020, 34(11): 14832-14849. DOI: 10.1096/fj.202001495R. [26] GAGGINI M, CARLI F, ROSSO C, et al. Altered amino acid concentrations in NAFLD: Impact of obesity and insulin resistance[J]. Hepatology, 2018, 67(1): 145-158. DOI: 10.1002/hep.29465. [27] XIAO F, HUANG Z, LI H, et al. Leucine deprivation increases hepatic insulin sensitivity via GCN2/mTOR/S6K1 and AMPK pathways[J]. Diabetes, 2011, 60(3): 746-756. DOI: 10.2337/db10-1246. [28] BUSH NC, BASU R, RIZZA RA, et al. Insulin-mediated FFA suppression is associated with triglyceridemia and insulin sensitivity independent of adiposity[J]. J Clin Endocrinol Metab, 2012, 97(11): 4130-4138. DOI: 10.1210/jc.2012-2285. [29] PEREIRA S, PARK E, MORI Y, et al. FFA-induced hepatic insulin resistance in vivo is mediated by PKCδ, NADPH oxidase, and oxidative stress[J]. Am J Physiol Endocrinol Metab, 2014, 307(1): E34-E46. DOI: 10.1152/ajpendo.00436.2013. [30] BISWAS D, DAO KT, MERCER A, et al. Branched-chain ketoacid overload inhibits insulin action in the muscle[J]. J Biol Chem, 2020, 295(46): 15597-15621. DOI: 10.1074/jbc.RA120.013121. [31] SOLON-BIET SM, COGGER VC, PULPITEL T, et al. Branched chain amino acids impact health and lifespan indirectly via amino acid balance and appetite control[J]. Nat Metab, 2019, 1(5): 532-545. DOI: 10.1038/s42255-019-0059-2. [32] HONG SH, LEE KS, KWAK SJ, et al. Minibrain/Dyrk1a regulates food intake through the Sir2-FOXO-sNPF/NPY pathway in drosophila and mammals[J]. PLoS Genet, 2012, 8(8): e1002857. DOI: 10.1371/journal.pgen.1002857. -



 PDF下载 ( 3176 KB)
PDF下载 ( 3176 KB)

 下载:
下载: