肝硬化门静脉高压症患者脾切除后早期血栓形成与Janus激酶-信号转导和转录激活因子信号通路的关系
DOI: 10.3969/j.issn.1001-5256.2022.10.012
Association of JAK/STAT signaling pathway with portal vein thrombosis after splenectomy in cirrhotic patients with portal hypertension
-
摘要:
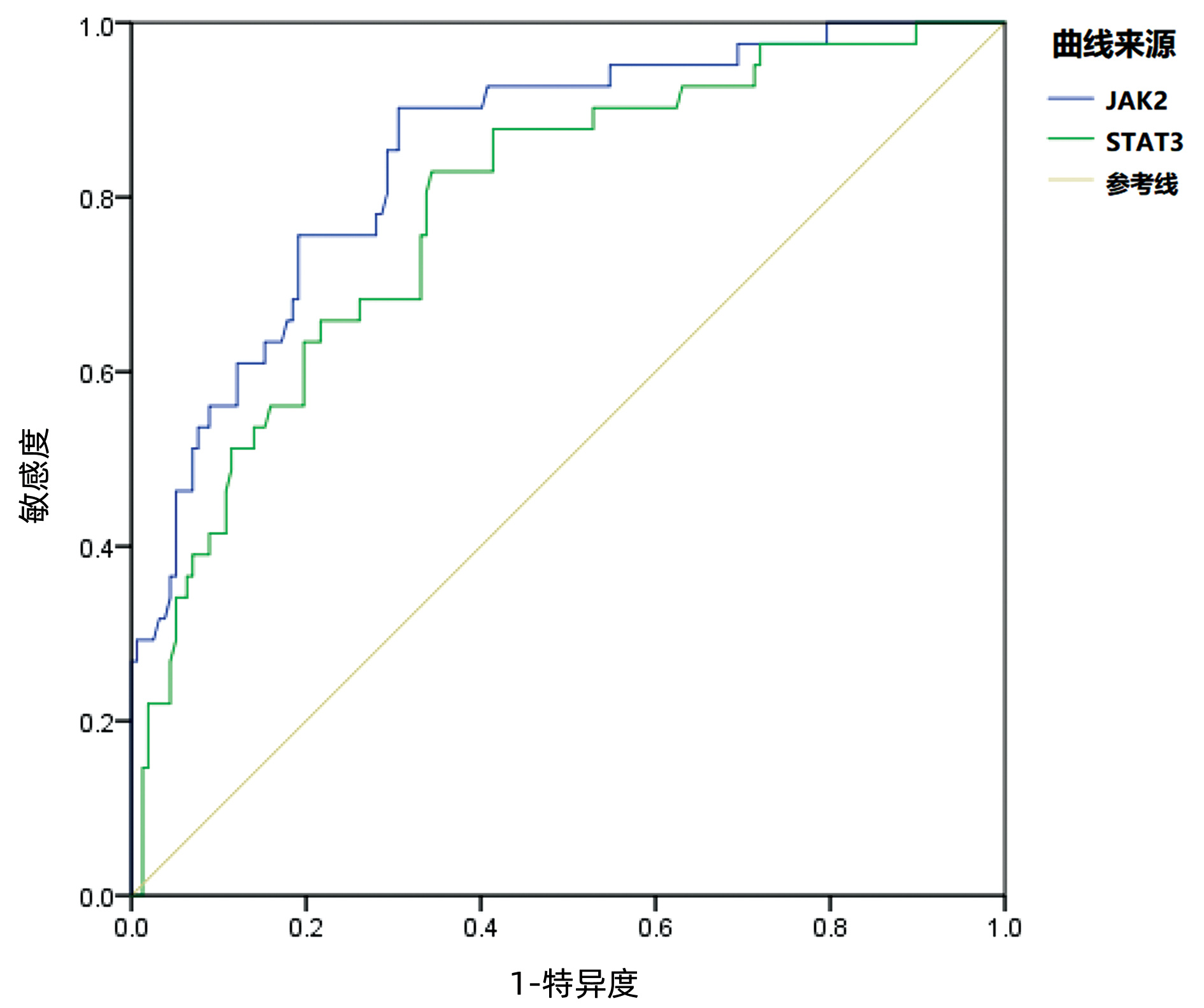
目的 探讨肝硬化门静脉高压症患者脾切除后早期血栓形成因素,及其与Janus激酶(JAK)-信号转导和转录激活因子(STAT)信号通路的关系。 方法 选择2018年3月—2020年4月在贵州医科大学附属医院因肝硬化门静脉高压行脾切除断流术患者198例,根据患者术后3个月内是否发生门静脉血栓将其分为血栓组(n=41)和非血栓组(n=157)。检测两组患者术后外周血单个核细胞中JAK2 mRNA及STAT3 mRNA表达情况。计量资料组间比较采用t检验,计数资料组间比较采用χ2检验,对可能影响患者术后门静脉血栓形成的各因素进行单因素及多因素Logistic回归分析;采用受试者工作曲线(ROC曲线)分析术后JAK2mRNA、STAT3 mRNA相对表达量对门静脉血栓的预测价值。 结果 单因素结果显示,血栓组患者术前门静脉直径、术后门静脉直径、门静脉流速差、脾容积均显著高于非血栓组(t值分别为4.718、3.945、8.671、2.006,P值均<0.05)。术后两组患者外周血单个核细胞JAK2 mRNA及STAT3 mRNA相对表达量较术前均显著升高(t值分别为12.933、15.442、14.386、10.896,P值均<0.05),同时血栓组患者JAK2 mRNA及STAT3 mRNA相对表达量升高程度均明显高于非血栓组(P值均<0.05)。多因素logistic回归分析结果显示,门静脉流速差、术后JAK2 mNRA、术后STAT3 mRNA及术前门静脉直径为影响患者术后门静脉血栓发生的重要因素(P值均<0.05)。患者术后JAK2 mRNA、STAT3 mRNA相对表达量预测门静脉血栓的ROC曲线下面积分别为0.850、0.787。 结论 JAK/STAT信号通路激活参与了肝硬化门静脉高压患者脾切除后血栓形成过程,可能是患者术后门静脉血栓形成的重要机制之一,有望成为患者术后门静脉血栓预防和治疗的潜在生物学途径。 Abstract:Objective To investigate the role of the Janus tyrosine kinase (JAK)- signal transducer and activators of transcription (STAT) signaling pathway in thrombosis formation after splenectomy in cirrhotic patients with portal hypertension. Methods A total of 198 liver cirrhosis patients with portal hypertension who underwent splenectomy and devascularization were recruited from March 2018 to April 2020 and then divided into thrombosis (n=41) and non-thrombosis groups (n=157) according to whether portal vein thrombosis occurred 3 months after surgery. mRNA levels of JAK2 and STAT3 in peripheral blood mononuclear cells (PBMCs) were assessed. Comparison of continuous data between groups was performed using t test, while comparison of categorial data between group was performed using chi square test. Clinicopathological data from patients were collected and analyzed for the potential risk factors of portal vein thrombosis by using univariate and multivariate Logistic regression analyses. The prediction values of JAK2 and STAT3 mRNA for portal vein thrombosis were evaluated by area under the receiver operating curve (AUC). Results The diameter of portal vein, the diameter of portal vein, the velocity difference of portal vein and the volume of spleen in the thrombus group were significantly higher than those in the non-thrombus group (t=4.718, 3.945, 8.671, and 2.006 respectively; P < 0.05).The relative mRNA levels of JAK2 and STAT3 in PBMCs of the two groups were significantly increased after surgery (t=12.933, 15.442, 14.386, 10.896; P < 0.05), and their levels were also much higher in the thrombosis group than in the non-thrombosis group of patients (P < 0.05). Multivariate Logistic regression analytic data showed that the portal vein diameter, the velocity difference of portal vein and mRNA level of JAK2 and STAT3 were all predictors for thrombosis in these patients (P < 0.05). The AUC of JAK2 and STAT3 mRNAs was 0.850 and 0.787, respectively in diagnosis of thrombosis in these patients. Conclusion The JAK / STAT signaling was activated in the process of thrombosis after splenectomy in cirrhotic patients with portal hypertension, which may be one of the important mechanisms of postoperative portal vein thrombosis, and may become a potential target for the prevention and treatment of postoperative portal vein thrombosis. -
Key words:
- Liver Cirrhosis /
- Hypertension /
- Portal /
- Splenectomy
-
表 1 单因素分析
Table 1. Single factor analysis
因素 血栓组(n=41) 非血栓组(n=157) 统计值 P值 性别(例) χ2=2.896 0.089 男 29 88 女 12 69 年龄(例) χ2=0.265 0.607 ≥50岁 23 81 <50岁 18 76 Child-Pugh分级(例) χ2=0.245 0.621 A级 25 89 B级 16 68 门静脉高压性胃病(例) χ2=0.469 0.493 是 36 131 否 5 26 病毒性肝炎(例) χ2=0.582 0.446 是 33 134 否 8 23 静脉曲张类型(例) χ2=0.037 0.848 GOV1 15 60 GOV2 26 97 术前门静脉直径(mm) 12.97±1.03 12.17±0.95 t=4.718 <0.001 术后门静脉直径(mm) 12.18±0.92 11.56±0.89 t=3.945 <0.001 门静脉流速差(cm/s) 8.95±2.02 6.27±1.69 t=8.671 <0.001 门静脉压力差(cmH2O) 9.37±1.98 8.96±2.31 t=1.041 0.299 脾容积(cm3) 1 382.48±560.28 1 202.83±497.03 t=2.006 0.046 腹水量(mL) 69.38±15.20 71.22±17.39 t=0.618 0.537 手术时间(min) 259.30±45.28 254.29±51.27 t=0.570 0.569 术中出血量(mL) 368.49±126.42 330.85±150.63 t=1.470 0.143 BMI(kg/m2) 23.26±3.04 22.37±3.79 t=1.391 0.166 术后7 d CRP 18.27±2.63 18.96±3.41 t=1.205 0.230 术后7 d VAS 1.30±0.31 1.21±0.40 t=1.339 0.182 表 2 两组患者手术前后JAK-STAT通路表达
Table 2. Expression of JAK-STAT pathway before and after operation in two groups
组别 例数 JAK2 mRNA STAT3 mRNA 血栓组 41 术前 0.793±0.167 0.842±0.188 术后 1.372±0.233 1.564±0.279 t值 12.933 15.442 P值 <0.001 <0.001 非血栓组 157 术前 0.762±0.179 0.860±0.205 术后 1.093±0.2261) 1.189±0.3181) t值 14.386 10.896 P值 <0.001 <0.001 注:与血栓组术后比较,1)P<0.05。 表 3 影响患者术后门静脉血栓发生的多因素Logistic回归分析
Table 3. Multivariate Logistic regression analysis on the incidence of postoperative portal vein thrombosis
因素 回归系数 标准误 Wald值 P值 OR(95%CI) 门静脉流速差 0.279 0.188 0.602 0.002 2.183(1.382~2.974) 术后JAK2 mRNA 0.318 0.173 0.583 0.013 1.795(1.028~3.118) 术后STAT3 mRNA 0.337 0.193 0.398 0.035 1.603(1.221~3.472) 术前门静脉直径 0.418 0.220 0.527 0.044 1.493(1.093~2.791) 表 4 术后JAK2 mRNA、STAT3 mRNA对门静脉血栓预测价值ROC曲线分析
Table 4. ROC curve analysis of the predictive value of JAK2 mRNA and STAT3 mRNA in postoperative portal vein thrombosis
指标 最佳截断值 标准误 P值 曲线下面积 95%CI JAK2 mRNA 1.228 0.033 <0.001 0.850 0.785~0.914 STAT3 mRNA 1.402 0.099 <0.001 0.787 0.710~0.864 -
[1] SIMONETTO DA, LIU M, KAMATH PS. Portal hypertension and related complications: diagnosis and management[J]. Mayo Clin Proc, 2019, 94(4): 714-726. DOI: 10.1016/j.mayocp.2018.12.020. [2] JIANG GQ, BAI DS, JIN SJ, et al. Modified vagus nerve-preserving versus conventional laparoscopic splenectomy and azygoportal disconnection for cirrhotic portal hypertension: a prospective randomized controlled trial[J]. Chin J Dig Surg, 2020, 19(12): 1280-1285. DOI: 10.3760/cma.j.cn115610-20201126-00741.蒋国庆, 柏斗胜, 金圣杰, 等. 改良保留迷走神经与传统腹腔镜脾切除联合贲门周围血管离断术治疗肝硬化门静脉高压症的前瞻性随机对照研究[J]. 中华消化外科杂志, 2020, 19(12): 1280-1285. DOI: 10.3760/cma.j.cn115610-20201126-00741. [3] HAN D, PENG JW, XIE HJ. Prevention of portal vein thrombosis in patients with cirrhotic portal hypertension aftersplenectomy and devascularization[J]. J Prac Hepatol, 2020, 23(3): 409-412. DOI: 10.3969/j.issn.1672-5069.2020.03.027.韩东, 彭经纬, 谢辉晋. 脾切除联合贲门周围血管离断术治疗肝硬化门脉高压症患者预防门静脉血栓形成研究[J]. 实用肝脏病杂志, 2020, 23(3): 409-412. DOI: 10.3969/j.issn.1672-5069.2020.03.027. [4] de FRANCHIS R, Baveno VI Faculty. Expanding consensus in portal hypertension: Report of the Baveno VI Consensus Workshop: Stratifying risk and individualizing care for portal hypertension[J]. J Hepatol, 2015, 63(3): 743-752. DOI: 10.1016/j.jhep.2015.05.022. [5] SONG ZM, DAI CL. Progress in diagnosis and treatment of portal vein thrombosis after splenectomy for cirrhosis and portal hypertension[J]. Chin J Curr Adv Gen Surg, 2019, 22(12): 958-962. DOI: 10.3969/j.issn.1009-9905.2019.12.009.宋子敏, 戴朝六. 肝硬化门脉高压症脾切除术后门静脉系统血栓形成的诊治进展[J]. 中国现代普通外科进展, 2019, 22(12): 958-962. DOI: 10.3969/j.issn.1009-9905.2019.12.009. [6] QI R, JIN X, SHI H, et al. Effect of laparoscopic splenectomy on portal vein thrombosis and serum YKL-40 in patients with cirrhotic portal hypertension[J]. Ann Hepatol, 2019, 18(6): 898-901. DOI: 10.1016/j.aohep.2019.06.009. [7] LI DY. Comparison of different intra-splenectomy splenic pedicle vascular treatments for postoperative portal vein thrombosis in cirrhotic portal hypertension[J]. Guangxi Med J, 2019, 41(8): 985-988. DOI: 10.11675/j.issn.0253-4304.2019.08.14.李丹阳. 肝硬化门静脉高压症脾切除术中不同脾蒂血管处理方法对术后门静脉血栓形成的对比研究[J]. 广西医学, 2019, 41(8): 985-988. DOI: 10.11675/j.issn.0253-4304.2019.08.14. [8] ZHANG XY, LI C, PENG W, et al. Platelet count-spleen volume ratio prediction of both moderate and severe portal hypertention in patients with cirrhosis by platelet count-spleen volume ratio[J]. J Abdominal Surg, 2016, 29(3): 187-192. DOI: 10.3969/j.issn.1003-5591.2016.03.009.张晓赟, 李川, 彭伟, 等. 血小板/脾脏体积比预测肝硬化门静脉高压症的严重程度[J]. 腹部外科, 2016, 29(3): 187-192. DOI: 10.3969/j.issn.1003-5591.2016.03.009. [9] XIONG ZJ, FAN HS, ZHU XN, et al. Application of FibroTouch combined with AST to platelet ratio index in diagnosis of portal hypertension in patients with hepatic cirrhosis[J/CD]. Chin J Hepat Surg(Electronic Edition), 2019, 8(4): 362-365. DOI: 10.3877/cma.j.issn.2095-3232.2019.04.019.熊志娇, 范红顺, 朱先女, 等. FibroTouch联合AST-血小板比值指数在肝硬化门静脉高压症诊断中的应用[J/CD]. 中华肝脏外科手术学电子杂志, 2019, 8(4): 362-365. DOI: 10.3877/cma.j.issn.2095-3232.2019.04.019. [10] SONG LJ, YUAN Y, WANG W. Clinical value of quantitative magnetic resonance imaging in the diagnosis of portal hypertension in patients with primary biliary cirrhosis[J]. J Prac Hepatol, 2020, 23(4): 568-571. DOI: 10.3969/j.issn.1672-5069.2020.04.029.宋丽俊, 袁雁, 王伟. 定量磁共振成像诊断原发性胆汁性肝硬化患者门静脉高压症价值分析[J]. 实用肝脏病杂志, 2020, 23(4): 568-571. DOI: 10.3969/j.issn.1672-5069.2020.04.029. [11] GIOIA S, NARDELLI S, PASQUALE C, et al. Natural history of patients with non cirrhotic portal hypertension: Comparison with patients with compensated cirrhosis[J]. Dig Liver Dis, 2018, 50(8): 839-844. DOI: 10.1016/j.dld.2018.01.132. [12] LYU SC, GU WQ. Research progress on prevention and treatment of portal vein thrombosis after splenectomy for patients with hepatic cirrhosis[J]. Chin J Hepatobiliary Surg, 2015, 21(1): 59-63. DOI: 10.3760/cma.j.issn.1007-8118.2015.01.019.吕少诚, 顾万清. 肝硬化患者脾切除术后门静脉血栓的防治研究进展[J]. 中华肝胆外科杂志, 2015, 21(1): 59-63. DOI: 10.3760/cma.j.issn.1007-8118.2015.01.019. [13] YANG S, HE C, FAN X, et al. Early prophylactic anticoagulation via transjugular intrahepatic route for portal vein thrombosis after splenectomy in cirrhotic portal hypertension[J]. J Vasc Interv Radiol, 2015, 26(7): 1009-1017. DOI: 10.1016/j.jvir.2015.03.008. [14] FENG Q, XU C, SUN MH. miR-141 inhibits thrombosis in vascular pathways through the TLR4 signaling pathway[J]. Med J West China, 2021, 33(2): 186-190. DOI: 10.3969/j.issn.1672-3511.2021.02.006.冯琦, 徐超, 孙明慧. miRNA-141通过调节TLR4信号通路抑制大鼠血栓形成[J]. 西部医学, 2021, 33(2): 186-190. DOI: 10.3969/j.issn.1672-3511.2021.02.006. [15] GU YN, XIE CY. Advancing investigate of deep vein thrombosis for inflammation and signa-ling pathway[J]. Chin J Immunol, 2020, 36(1): 113-118. DOI: 10.3969/j.issn.1000-484X.2020.01.022.顾燕妮, 谢春毅. 深静脉血栓形成炎症信号通路研究进展[J]. 中国免疫学杂志, 2020, 36(1): 113-118. DOI: 10.3969/j.issn.1000-484X.2020.01.022. [16] MALEMUD CJ. The role of the JAK/STAT signal pathway in rheumatoid arthritis[J]. Ther Adv Musculoskelet Dis, 2018, 10(5-6): 117-127. DOI: 10.1177/1759720X18776224. [17] BANERJEE S, BIEHL A, GADINA M, et al. JAK-STAT signaling as a target for inflammatory and autoimmune diseases: Current and future prospects[J]. Drugs, 2017, 77(5): 521-546. DOI: 10.1007/s40265-017-0701-9. [18] LI Y, LI KH, WEN SH, et al. Role of JAK/STAT in intestinal injury induced by intestinal ischemia/reperfusion in rats[J]. Chin J Pathophysioly, 2011, 27(12): 2338-2344. DOI: 10.3969/j.issn.1000-4718.2011.12.019.李毅, 李坤河, 温仕宏, 等. JAK/STAT通路在大鼠肠缺血再灌注所致肠损伤中的作用[J]. 中国病理生理杂志, 2011, 27(12): 2338-2344. DOI: 10.3969/j.issn.1000-4718.2011.12.019. -



 PDF下载 ( 1970 KB)
PDF下载 ( 1970 KB)

 下载:
下载:


