首乌制剂相关肝损伤伴或不伴自身抗体阳性患者临床特征比较
DOI: 10.3969/j.issn.1001-5256.2022.10.018
Clinical features of Polygonum multiflorum preparation-related liver injury with or without positive autoantibody
-
摘要:
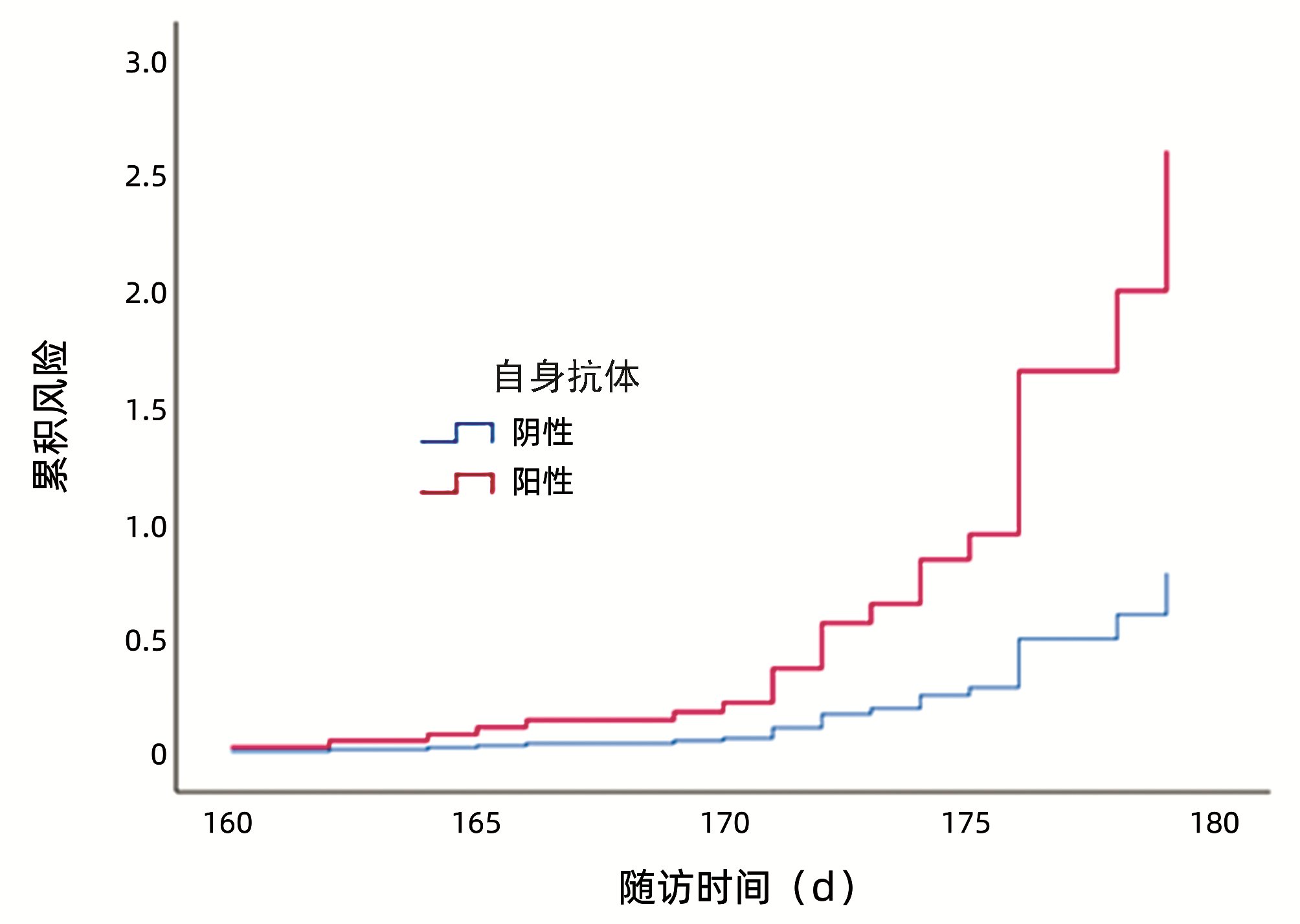
目的 基于倾向性评分匹配对比分析首乌制剂相关肝损伤伴自身抗体阳性患者与不伴自身抗体阳性患者的临床特征,探讨合并自身抗体对首乌制剂相关肝损伤患者预后的影响。 方法 选取首都医科大学附属北京地坛医院2008年8月—2021年6月首乌制剂相关肝损伤住院患者364例,根据是否检测到自身抗体分为自身抗体阴性组(H0组)和自身抗体阳性组(H1组),其中H0组157例,H1组207例,采用倾向性评分匹配方法进行混杂因素校正,比较两组患者肝损伤达峰值时生化指标、肝损伤严重程度、肝损伤分型及疾病转归情况的差异。正态分布的计量资料两组间比较采用t检验;非正态分布的计量资料两组间比较采用Mann-Whitney U秩和检验。计数资料或等级资料组间比较采用χ2检验或Mann-Whitney U秩和检验。应用Cox回归模型分析肝功能恢复的影响因素。通过Kaplan-Meier法绘制生存曲线,并用Log-rank检验进行比较。 结果 倾向性评分匹配后,成功匹配98对病例。两组肝损伤达峰值时生化指标比较结果显示,H1组ALP、球蛋白、总胆汁酸水平均高于H0组,Alb水平低于H0组,差异均有统计学意义(P值均<0.05)。两组患者肝损伤分型差异无统计学意义(P>0.05)。肝损伤严重程度方面两组比较差异有统计学意义(Z=1.710,P=0.045)。自身抗体以抗核抗体(ANA)阳性为主,占比49%;不同ANA抗体滴度患者肝损伤严重程度比较,差异有统计学意义(Z=20.252,P=0.001)。疾病转归以半年内肝功能恢复正常进行比较,H0、H1组半年肝功能复常率分别为90.8%和75.5%,H1组肝功能复常率偏低(χ2=8.199,P=0.004)。Cox回归分析显示自身抗体(HR=5.248,95%CI:1.554~17.718,P=0.008)和ALP(HR=1.013,95%CI:1.002~1.025,P=0.026)是肝功能恢复的独立危险因素。Kaplan-Meier生存曲线结果显示,半年后自身抗体阳性组患者肝功能未恢复的风险较自身抗体阴性组更高(χ2=8.802,P=0.003)。 结论 自身抗体对首乌制剂相关肝损伤分型无明显影响,与自身抗体阴性的首乌制剂肝损伤患者相比,伴自身抗体阳性的首乌制剂肝损伤患者有更加严重的肝损伤以及更明显的慢性化倾向。 -
关键词:
- 化学性与药物性肝损伤 /
- 何首乌 /
- 自身抗体
Abstract:Objective To investigate the clinical features of patients with Polygonum multiflorum preparation-related liver injury with or without positive autoantibody based on propensity score matching, as well as the influence of positive autoantibody on the prognosis of such patients. Methods A total of 364 patients with Polygonum multiflorum preparation-related liver injury who were hospitalized in Beijing Ditan Hospital, Capital Medical University, from August 2008 to June 2021 were enrolled, and according to whether autoantibodies were detected, they were divided into negative autoantibody group (H0 group) with 157 patients and positive autoantibody group (H1 group) with 207 patients. After adjustment for confounding factors by propensity score matching, the two groups were compared in terms of biochemical parameters, severity of liver injury, classification of liver injury, and disease outcome when liver injury reached the peak. The t-test was used for comparison of normally distributed continuous data between two groups, and the Mann-Whitney U rank sum test was used for comparison of non-normally distributed continuous data between two groups; the chi-square test or the Mann-Whitney U rank sum test was used for comparison of categorical data or ranked data between two groups. The Cox regression model was used to analyze the influencing factors for liver function recovery. The Kaplan-Meier method was used to plot survival curves, and the Log-rank test was used for comparison between groups. Results A total of 98 pairs of cases were successfully matched after propensity score matching. Comparison of biochemical parameters between the two groups at the peak of liver injury showed that compared with the H0 group, the H1 group had significantly higher levels of alkaline phosphatase (ALP), globulin, and total bile acid and a significantly lower level of albumin (all P < 0.05). There was no significant difference in the classification of liver injury between the two groups (P > 0.05), and there was a significant difference in the severity of liver injury between the two groups (Z=1.710, P=0.045). Antinuclear antibody (ANA) was the main positive autoantibody and accounted for 49%, and there was a significant difference in the severity of liver injury between the patients with different ANA antibody titers (Z=20.252, P=0.001). Comparison of disease outcome in terms of whether liver function returned to normal within 6 months showed that the normalization rate of liver function within 6 months was 90.8% in the H0 group and 75.5% in the H1 group, and the H1 group had a lower normalization rate of liver function (χ2=8.199, P=0.004). The Cox regression analysis showed that autoantibody (hazard ratio [HR]=5.248, 95% confidence interval [CI]: 1.554-17.718, P=0.008) and ALP (HR=1.013, 95%CI: 1.002-1.025, P=0.026) were independent risk factors for liver function recovery. The Kaplan-Meier survival curve analysis showed that compared with the negative autoantibody group, the positive autoantibody group had a significantly higher risk of failure in liver function recovery after 6 months (χ2=8.802, P=0.003). Conclusion Autoantibody has no significant influence on the classification of Polygonum multiflorum preparation-related liver injury, and compared with the patients with negative autoantibody, the patients with positive autoantibody tend to have more severe liver injury and a more obvious tendency of chronicity. -
表 1 两组患者基线资料比较
Table 1. Comparison of baseline data between the two groups
项目 H0组(n=98) H1组(n=98) 统计值 P值 性别[例(%)] χ2=0.023 0.880 男 34(34.7) 33(33.7) 女 64(65.3) 65(66.3) 年龄(岁) 45.17±12.14 45.34±12.52 t=0.053 0.958 过敏史[例(%)] χ2=0.191 0.662 有 13(13.3) 15(15.3) 无 85(86.7) 83(84.7) 用药时间(d) 20.93±7.19 21.45±11.15 t=0.485 0.706 用药剂型[例(%)] χ2=0.473 0.925 汤剂 30(30.6) 29(29.6) 丸剂 18(18.4) 16(16.3) 胶囊 23(23.5) 27(27.6) 其他 27(27.6) 26(26.5) 表 2 自身抗体分布结果
Table 2. Distribution results of autoantibodies
自身抗体 例数 阳性率(%) ANA 48 49.0 抗SS-A抗体 12 12.2 抗Ro-52抗体 10 10.2 抗线粒体抗体 9 9.2 抗Scl-70抗体 6 6.1 抗双链DNA抗体 3 3.1 其余 10 10.2 表 3 两组患者肝损伤类型及严重程度比较
Table 3. Comparison of liver injury types and severity between the two groups
项目 H0组
(n=98)H1组
(n=98)统计值 P值 肝损伤类型[例(%)] χ2=3.974 0.281 肝细胞损伤型 80(81.6) 78(79.6) 胆汁淤积型 5(5.1) 8(8.2) 混合型 9(9.2) 4(4.1) 肝脏生化学检查异常 4(4.1) 8(8.2) 严重程度[例(%)] Z=1.710 0.045 1级 43(43.9) 23(23.5) 2级 10(10.2) 13(13.3) 3级 40(40.8) 57(58.2) 4级 5(5.1) 5(5.1) 表 4 两组患者肝损伤达峰值时生化指标比较
Table 4. Comparison of biochemical parameters between the two groups when liver injury reached its peak
项目 H0组(n=98) H1组(n=98) 统计值 P值 ALT(U/L) 710.30(417.55~1 019.15) 662.3(364.8~1 080.95) Z=0.397 0.919 AST(U/L) 335.70(189.00~706.15) 465.55(239.25~676.65) Z=0.848 0.397 TBil(μmol/L) 64.50(20.75~154.20) 88.25(21.88~244.13) Z=-1.384 0.166 Alb(g/L) 39.84±4.42 36.75±6.16 t=-2.182 0.033 Glo(g/L) 25.24±3.95 29.45±6.76 t=2.851 0.006 GGT(U/L) 110.80(70.75~252.65) 128.35(103.70~276.35) Z=-1.117 0.264 ALP(U/L) 139.13±55.73 160.68±88.79 t=1.093 0.029 ChE(U/L) 5 960.48±2 022.82 5 997.96±1 680.09 t=0.076 0.940 TBA(μmol/L) 47.80(9.00~177.40) 135.9(35.55~276.83) Z=-1.963 0.050 TC(mmol/L) 4.51±3.09 3.75±1.26 t=-1.200 0.236 TG(mmol/L) 1.40(0.95~2.00) 1.60(1.23~2.28) Z=-1.419 0.156 PT(s) 12.10(11.30~13.25) 12.00(11.33~13.90) Z=-0.335 0.738 PTA(%) 86.77±20.71 83.16±22.11 t=-0.641 0.524 INR 1.14±0.21 1.15±0.30 t=0.102 0.919 AFP(ng/mL) 3.70(2.55~13.00) 4.75(3.43~10.55) Z=-0.962 0.336 Cr(μmol/L) 61.10±14.92 62.73±17.55 t=0.380 0.706 BUN(mmol/L) 3.75±1.18 3.60±1.09 t=1.276 0.207 注:GLO,球蛋白;ChE,胆碱酯酶;TBA,总胆汁酸;INR,国际标准化比值;Cr,肌酐;BUN,尿素氮。 表 5 不同ANA滴度患者肝损伤分型及严重程度比较
Table 5. Comparison of liver injury types and severity among different antinuclear antibody titers
项目 低滴度(n=22) 中滴度(n=12) 高滴度(n=14) 统计值 P值 肝损伤类型[例(%)] χ2=1.641 0.440 肝细胞损伤型 18(81.8) 9(75.0) 8(57.1) 胆汁淤积型 0 2(16.7) 4(28.6) 混合型 1(4.5) 0 1(7.1) 肝生化学检查异常 3(13.6) 1(8.3) 1(7.1) 严重程度[例(%)] Z=20.252 0.001 1级 12(54.5) 2(16.7) 1(7.1) 2级 6(27.3) 1(8.3) 0 3级 4(18.2) 9(75.0) 10(71.4) 4级 0 0 3(21.4) 表 6 两组患者半年后生化指标比较
Table 6. Comparison of biochemical indexes between the two groups after six months
项目 H0组(n=98) H1组(n=98) 统计值 P值 ALT(U/L) 23.05(18.30~30.70) 37.70(15.30~56.15) Z=-2.131 0.033 AST(U/L) 22.30(18.03~30.80) 29.30(20.80~47.15) Z=-2.698 0.007 TBil(μmol/L) 15.25(10.93~19.93) 14.70(10.55~26.05) Z=-1.050 0.290 Alb(g/L) 44.95±4.14 42.69±3.67 t=-2.204 0.032 Glo(g/L) 27.79±4.33 31.20±6.64 t=-2.308 0.025 GGT(U/L) 33.25(16.73~42.30) 50.30(12.85~73.65) Z=-0.860 0.390 ALP(U/L) 81.24±28.34 93.17±33.04 t=-1.427 0.046 ChE(U/L) 7 020.45±1 808.56 7 237.69±1 627.41 t=-0.481 0.633 TBA(μmol/L) 4.25(2.00~6.83) 5.10(2.25~22.85) Z=-1.300 0.190 住院时间(d) 17.93±7.18 21.45±11.15 t=1.428 0.159 肝功能恢复正常[例(%)] χ2=8.199 0.004 是 89(90.8) 74(75.5) 否 9(9.2) 24(24.5) 表 7 不同ANA滴度对患者预后的影响
Table 7. Effects of different antinuclear antibody titers on prognosis of patients
组别 例数 肝功能恢复
[例(%)]肝功能未恢复
[例(%)]低滴度 22 19(86.4) 3(13.6) 中滴度 12 10(83.3) 2(16.7) 高滴度 14 6(42.9) 8(57.1) 表 8 影响肝功能恢复的Cox回归分析
Table 8. Cox regression analysis model for liver function recovery
项目 单因素分析 多因素分析 HR 95%CI P值 HR 95%CI P值 性别 1.011 0.466~2.191 0.979 年龄 1.004 0.975~1.034 0.764 住院时间 1.038 0.992~1.086 0.108 用药时间 0.720 0.440~1.177 0.090 用药剂型 1.291 0.937~1.778 0.118 肝损伤类型 1.184 0.784~1.790 0.422 自身抗体 3.629 1.376~9.567 0.009 5.248 1.554~17.718 0.008 ALT(U/L) 1.000 0.999~1.001 0.673 AST(U/L) 1.002 0.999~1.005 0.522 TBil(μmol/L) 1.000 0.998~1.003 0.799 Alb(g/L) 0.963 0.869~1.067 0.475 Glo(g/L) 1.038 0.969~1.113 0.283 GGT(U/L) 1.001 0.996~1.006 0.655 ALP(U/L) 1.012 1.001~1.023 0.031 1.013 1.002~1.025 0.026 ChE(U/L) 1.000 0.467~1.032 0.062 TBA(μmol/L) 1.016 0.998~1.035 0.082 -
[1] Branch of Hepatobiliary Diseases, China Association of Chinese Medicine; Branch of Chinese Patent Medicine, China Association of Chinese Medicine. Guideline for diagnosis and treatment of herb-induced liver injury[J]. J Clin Hepatol, 2016, 32(5): 835-843. DOI: 10.3969/j.issn.1001-5256.2016.05.003.中华中医药学会肝胆病分会, 中华中医药学会中成药分会. 中草药相关肝损伤临床诊疗指南[J]. 临床肝胆病杂志, 2016, 32(5): 835-843. DOI: 10.3969/j.issn.1001-5256.2016.05.003. [2] AN XD, WEI Y, LIAN FM. Clinical application and dosage of fleeceflower root[J]. J Changchun Univ Chin Med, 2020, 36(2): 219-221. DOI: 10.13463/j.cnki.cczyy.2020.02.005.安学冬, 韦宇, 连凤梅. 何首乌的临床应用及其用量[J]. 长春中医药大学学报, 2020, 36(2): 219-221. DOI: 10.13463/j.cnki.cczyy.2020.02.005. [3] CAO YN, WANG NN, ZHOU GQ, et al. Analysis of influencing factors of clinical outcome in 186 patients with the drug-in-duced liver injury with positive autoantibodies[J]. Chin J Integr Tradit West Med Liver Dis, 2021, 31(1): 26-29, 33. DOI: 10.3969/j.issn.1005-0264.2021.01.007.曹亦楠, 王娜娜, 周桂琴, 等. 自身抗体阳性药物性肝损伤患者临床转归影响因素分析[J]. 中西医结合肝病杂志, 2021, 31(1): 26-29, 33. DOI: 10.3969/j.issn.1005-0264.2021.01.007. [4] YIN JY. Brief analysis of clinical value of detecting autoimmune antibody in patients with drug-induced liver injury[J]. China Prac Med, 2021, 16(7): 77-79. DOI: 10.14163/j.cnki.11-5547/r.2021.07.031.尹金燕. 浅析药物性肝损伤患者检测自身免疫性抗体的临床价值[J]. 中国实用医药, 2021, 16(7): 77-79. DOI: 10.14163/j.cnki.11-5547/r.2021.07.031. [5] Drug-induced Liver Disease Study Group, Chinese Society of Hepatology, Chinese Medical Association. Guidelines for the management of drug-induced liver injury[J]. J Clin Hepatol, 2015, 31(11): 1752-1769. DOI: 10.3969/j.issn.1001-5256.2015.11.002.中华医学会肝病学分会药物性肝病学组. 药物性肝损伤诊治指南[J]. 临床肝胆病杂志, 2015, 31(11): 1752-1769. DOI: 10.3969/j.issn.1001-5256.2015.11.002. [6] Chinese Society of Hepatology, Chinese Medical Association. Guidelines on the diagnosis and management of autoimmune hepatitis (2021)[J]. J Clin Hepatol, 2022, 38(1): 42-49. DOI: 10.3760/cma.j.cn112138-20211112-00796.中华医学会肝病学分会. 自身免疫性肝炎诊断和治疗指南(2021)[J]. 临床肝胆病杂志, 2022, 38(1): 42-49. DOI: 10.3760/cma.j.cn112138-20211112-00796. [7] SRIVASTAVA A, MAGGS JL, ANTOINE DJ, et al. Role of reactive metabolites in drug-induced hepatotoxicity[J]. Handb Exp Pharmacol, 2010, 196: 165-194. DOI: 10.1007/978-3-642-00663-0_7. [8] VINKEN M, MAES M, VANHAECKE T, et al. Drug-induced liver injury: mechanisms, types and biomarkers[J]. Curr Med Chem, 2013, 20(24): 3011-3021. DOI: 10.2174/0929867311320240006. [9] FONTANA RJ. Pathogenesis of idiosyncratic drug-induced liver injury and clinical perspectives[J]. Gastroenterology, 2014, 146(4): 914-928. DOI: 10.1053/j.gastro.2013.12.032. [10] REN MX, CHEN J, HUANG CY, et al. The clinical and pathological characteristics of drug-induced liver injury accompanied by autoimmune phenomena[J]. J China-Japan Friendship Hosp, 2018, 32(5): 279-283. DOI: 10.3969/j.issn.1001-0025.2018.05.006.任美欣, 陈杰, 黄春洋, 等. 伴自身免疫现象的药物性肝损伤的临床和病理特点[J]. 中日友好医院学报, 2018, 32(5): 279-283. DOI: 10.3969/j.issn.1001-0025.2018.05.006. [11] DENG ML, YANG LJ, SHI XL, et al. The immunologic and clinic features of 128 cases of drug-induced liver injury[J]. Chin Hosp Pharm J, 2019, 39(1): 63-66. DOI: 10.13286/j.cnki.chinhosppharmacyj.2019.01.14.邓茂林, 杨丽君, 石先利, 等. 128例药物性肝损伤患者的免疫特点及临床分析[J]. 中国医院药学杂志, 2019, 39(1): 63-66. DOI: 10.13286/j.cnki.chinhosppharmacyj.2019.01.14. [12] STINE JG, CHALASANI N. Chronic liver injury induced by drugs: a systematic review[J]. Liver Int, 2015, 35(11): 2343-2353. DOI: 10.1111/liv.12958. [13] MENG YK, LI CY, LI RY, et al. Cis-stilbene glucoside in Polygonum multiflorum induces immunological idiosyncratic hepatotoxicity in LPS-treated rats by suppressing PPAR-γ[J]. Acta Pharmacol Sin, 2017, 38(10): 1340-1352. DOI: 10.1038/aps.2017.32. [14] TENG SS, SUN Z, QIU Y, et al. Investigation, optimization and evaluation of traditional Chinese medicine processing technology of nine steaming and nine drying[J]. J Changchun Univ Chin Med, 2022, 38(1): 109-113. DOI: 10.13463/j.cnki.cczyy.2022.01.026.滕杉杉, 孙震, 邱野, 等. 中药炮制传统工艺九蒸九晒的调研、优化及评价[J]. 长春中医药大学学报, 2022, 38(1): 109-113. DOI: 10.13463/j.cnki.cczyy.2022.01.026. -



 PDF下载 ( 1991 KB)
PDF下载 ( 1991 KB)

 下载:
下载:


