国际标准化比值不同分层的慢加急性肝衰竭患者行人工肝治疗的预后及不良反应观察
DOI: 10.3969/j.issn.1001-5256.2022.10.020
Prognosis and adverse reactions of patients with acute-on-chronic liver failure receiving artificial liver support therapy stratified by international normalized ratio
-
摘要:
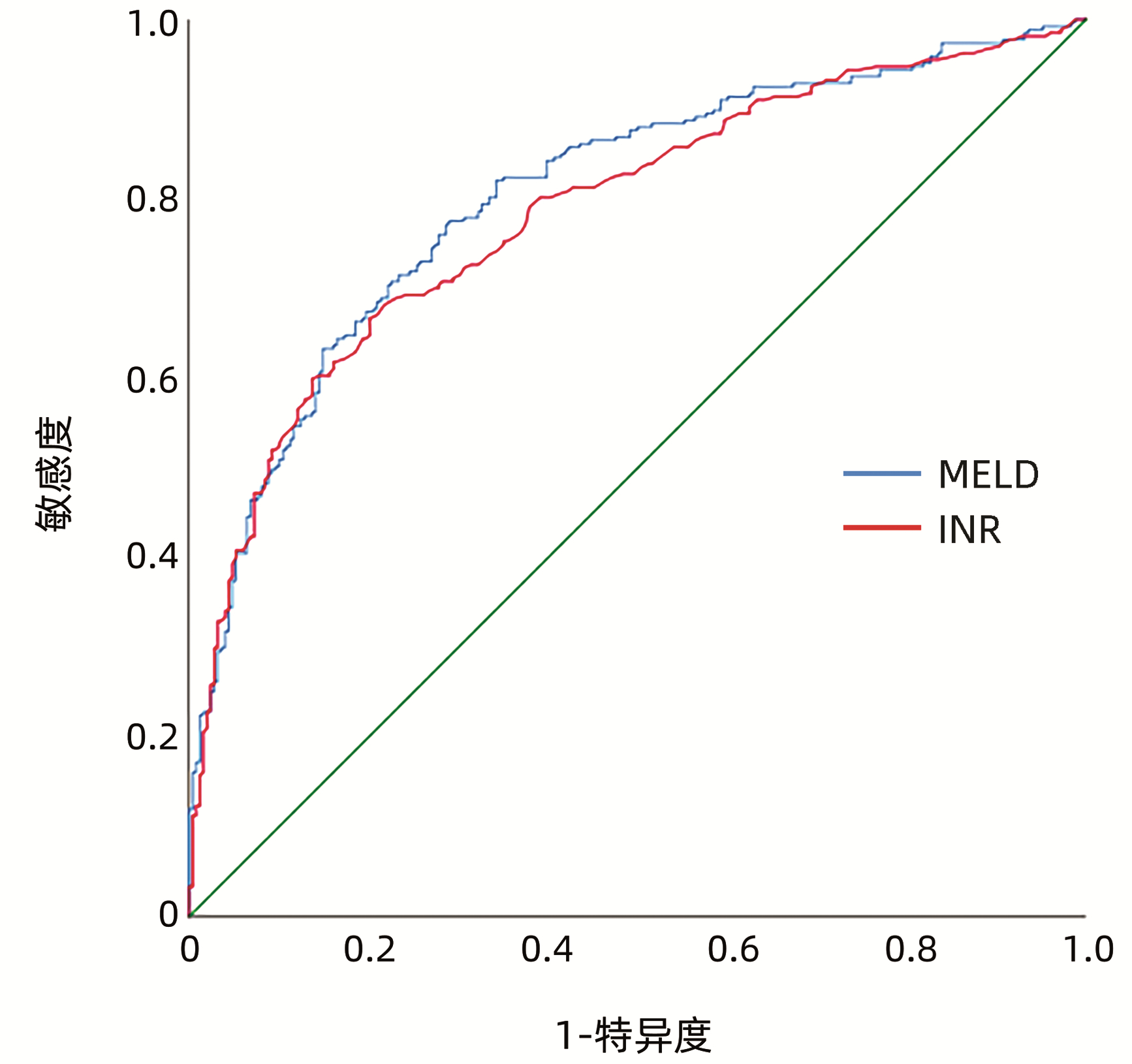
目的 观察不同国际标准化比值(INR)水平的慢加急性肝衰竭(ACLF)患者人工肝治疗的预后及不良反应。 方法 选取2010年1月—2020年5月于南昌市第九医院重症肝病科行人工肝治疗的ACLF患者共515例,根据INR水平将其分为6组:A组(INR<1.5)20例,B组(1.5≤INR<1.9)115例,C组(1.9≤INR<2.6)179例,D组(2.6≤INR<3.2)61例,E组(3.2≤INR<4.2)75例,F组(INR≥4.2)65例。所有患者均经内科综合治疗联合人工肝治疗。计量资料多组间采用方差分析或Kruskal-Wallis H检验,计数资料多组间比较采用χ2检验,进一步两两比较均采用Bonferroni校正法。通过受试者工作特征曲线评估INR及MELD评分系统对ACLF患者预后的预测价值。 结果 INR不同分层ACLF患者90 d病死率比较差异有统计学意义(χ2=124.84,P<0.001),其中A组(25.0%)、B组(25.2%)、C组(39.7%)之间病死率差异无统计学意义(P>0.05);D组(65.6%)、E组(82.7%)、F组(92.3%)与A、B、C组病死率比较,差异均有统计学意义(P值均<0.05);D组与E组患者病死率差异无统计学意义(P>0.05);D组与F组病死率差异有统计学意义(P<0.05);E组与F组病死率差异无统计学意义(P>0.05)。6组间人工肝术中不良反应的发生率差异无统计学意义(χ2=8.956,P=0.111)。INR预测人工肝治疗ACLF患者预后的曲线下面积为0.786(95%CI:0.746~0.825,P<0.001),敏感度、特异度分别为66.7%、79.8%。 结论 INR对ACLF患者行人工肝治疗的预后具有较好的预测价值,人工肝安全性良好。 Abstract:Objective To investigate the prognosis and adverse reactions of patients with acute-on-chronic liver failure (ACLF) receiving artificial liver support therapy stratified by international normalized ratio (INR). Methods A total of 515 ACLF patients who received artificial liver support therapy in Department of Severe liver Disease, The Ninth Hospital of Nanchang, from January 2010 to May 2020 were enrolled, and according to the level of INR, they were divided into group A with 20 patients (INR < 1.5), group B with 115 patients (1.5≤INR < 1.9), group C with 179 patients (1.9≤INR < 2.6), group D with 61 patients (2.6≤INR < 3.2), group E with 75 patients (3.2≤INR < 4.2), and group F with 65 patients (INR≥4.2). All patients received multimodality medical treatment combined with artificial liver support therapy. The one-way analysis of variance was used for comparison of normally distributed continuous data between multiple groups; the Kruskal-Wallis H test was used for comparison of non-normally distributed continuous data between multiple groups。The chi-square test was used for comparison of categorical data between groups. Bonferroni correction was used for further comparison between two groups. and the receiver operating characteristic (ROC) curve was used to evaluate the value of INR and MELD scoring system in predicting the prognosis of ACLF patients. Results As for 90-day mortality rate, there was a significant difference between the six groups stratified by INR (χ2=124.84, P < 0.001); there was no significant difference between groups A(25%), B(25.2%), and C(39.7%) (P > 0.05), and there was a significant difference between groups D/E/F(65.6%, 82.7%, and 92.3%, respectively) and groups A/B/C (all P < 0.05); there was no significant difference between groups D and E and between groups E and F (P > 0.05), and there was a significant difference between groups D and F (P < 0.05). There was no significant difference in the incidence rate of intraoperative adverse reactions between the six groups (χ2=8.956, P=0.111). INR had an area under the ROC curve of 0.786 (95% confidence interval: 0.746-0.825, P < 0.001) in predicting the prognosis of patients with ACLF receiving artificial liver support therapy, with a sensitivity of 66.7% and a specificity of 79.8%. Conclusion INR has a good value in predicting the prognosis of ACLF patients receiving artificial liver support therapy, and the artificial liver has good safety. -
表 1 INR不同分层ACLF患者的一般资料比较
Table 1. Comparison of general data of ACLF patients with different INR stratification
组别 例数 男/女
(例)年龄
(岁)MELD评分 INR PTA
(%)ALT
(U/L)SCr
(mg/dL)TBil
(mg/dL)B组 115 106/9 46.12±11.70 12.76(11.92~13.66) 1.66(1.61~1.77) 49.88±5.68 173.00(101.00~272.00) 0.76(0.66~0.92) 21.27±7.63 C组 179 161/18 44.66±12.07 14.25(13.19~15.24) 2.19(2.03~2.41) 36.83±5.27 225.00(103.00~476.00) 0.76(0.64~0.96) 23.48±16.97 D组 61 57/4 44.85±11.55 15.82(14.72~16.41) 2.80(2.72~2.93) 28.28±3.24 242.00(123.80~687.35) 0.80(0.67~0.93) 24.92±7.33 E组 75 62/13 43.35±11.87 16.73(15.67~18.38) 3.51(3.29~3.78) 22.65±4.14 273.00(86.00~602.90) 0.76(0.63~1.12) 24.13±7.66 F组 65 55/10 45.51±12.89 18.71(17.77~21.57) 5.31(4.76~6.30) 14.40±3.95 452.8(145.00~1 191.85) 0.79(0.63~1.24) 24.49±7.00 统计值 χ2=8.37 F=0.61 H=302.45 H=483.21 F=646.98 H=26.57 H=1.24 F=1.21 P值 0.137 0.692 <0.001 <0.001 <0.001 <0.001 0.941 0.303 表 2 INR不同分层ACLF患者90 d预后情况比较
Table 2. Comparison of 90 days prognosis of ACLF patients with different INR stratification
组别 例数 死亡[例(%)] A组 20 5(25.0) B组 115 29(25.2) C组 179 71(39.7) D组 61 40(65.6)1)2)3) E组 75 62(82.7)1)2)3) F组 65 60(92.3)1)2)3)4) χ2值 124.84 P值 <0.001 注:与A组比较,1)P<0.05;与B组比较,2)P<0.05;与C组比较,3)P<0.05;与D组比较,4)P<0.05。 表 3 INR不同分层ACLF患者人工肝术中不良反应比较
Table 3. Comparison of adverse reactions of ACLF patients with different INR stratification in artificial liver support system
组别 例数 无不良反应
[例(%)]有不良反应
[例(%)]A组 20 12(60.0) 8(40.0) B组 115 59(51.3) 56(48.7) C组 179 119(66.5) 60(33.5) D组 61 31(50.8) 30(49.2) E组 75 46(61.3) 29(38.7) F组 65 40(61.5) 25(38.5) χ2值 8.956 P值 0.111 -
[1] Chinese Society of Infectious Diseases, Chinese Medical Association; Chinese Society of Hepatology, Chinese Medical Association. Guidelines for the prevention and treatment of chronic hepatitis B (version 2019)[J]. J Clin Hepatol, 2019, 35(12): 2648-2669. DOI: 10.3969/j.issn.1001-5256.2019.12.007.中华医学会感染病学分会, 中华医学会肝病学分会. 慢性乙型肝炎防治指南(2019年版)[J]. 临床肝胆病杂志, 2019, 35(12): 2648-2669. DOI: 10.3969/j.issn.1001-5256.2019.12.007. [2] XIAO LL, XU XW, HUANG KZ, et al. Artificial liver support system improves short-term outcomes of patients with HBV-associated acute-on-chronic liver failure: A propensity score analysis[J]. Biomed Res Int, 2019, 2019: 3757149. DOI: 10.1155/2019/3757149. [3] FAN Q, LI Z. Liver transplantation for acute-on-chronic liver failure[J]. Ogran Transplant, 2022, 13(3): 333-337. DOI: 10.3969/j.issn.1674-7445.2022.03.008.范祺, 李照. 慢加急性肝衰竭的肝移植治疗[J]. 器官移植, 2022, 13(3): 333-337. DOI: 10.3969/j.issn.1674-7445.2022.03.008. [4] ALSHAMSI F, ALSHAMMARI K, BELLEY-COTE E, et al. Extracorporeal liver support in patients with liver failure: a systematic review and meta-analysis of randomized trials[J]. Intensive Care Med, 2020, 46(1): 1-16. DOI: 10.1007/s00134-019-05783-y. [5] Liver Failure and Artificial Liver Group, Chinese Society of Infectious Diseases, Chinese Medical Association; Severe Liver Disease and Artificial Liver Group, Chinese Society of Hepatology, Chinese Medical Association. Guideline for diagnosis and treatment of liver failure(2018)[J]. J Clin Hepatol, 2019, 35(1): 38-44. DOI: 10.3969/j.issn.1001-5256.2019.01.007.中华医学会感染病学分会肝衰竭与人工肝学组, 中华医学会肝病学分会重型肝病与人工肝学组. 肝衰竭诊治指南(2018年版)[J]. 临床肝胆病杂志, 2019, 35(1): 38-44. DOI: 10.3969/j.issn.1001-5256.2019.01.007. [6] Chinese Society of Hepatology, Chinese Medical Association. Chinese guidelines on the management of liver cirrhosis[J]. J Clin Hepatol, 2019, 35(11): 2408-2425. DOI: 10.3969/j.issn.1001-5256.2019.11.006.中华医学会肝病学分会. 肝硬化诊治指南[J]. 临床肝胆病杂志, 2019, 35(11): 2408-2425. DOI: 10.3969/j.issn.1001-5256.2019.11.006. [7] PORTE RJ, LISMAN T, TRIPODI A, et al. The International Normalized Ratio (INR) in the MELD score: problems and solutions[J]. Am J Transplant, 2010, 10(6): 1349-1353. DOI: 10.1111/j.1600-6143.2010.03064.x. [8] MOREAU R, JALAN R, GINES P, et al. Acute-on-chronic liver failure is a distinct syndrome that develops in patients with acute decompensation of cirrhosis[J]. Gastroenterology, 2013, 144(7): 1426-1437. DOI: 10.1053/j.gastro.2013.02.042. [9] CLÀRIA J, STAUBER RE, COENRAAD MJ, et al. Systemic inflammation in decompensated cirrhosis: Characterization and role in acute-on-chronic liver failure[J]. Hepatology, 2016, 64(4): 1249-1264. DOI: 10.1002/hep.28740. [10] RUESCHENBAUM S, CIESEK S, QUECK A, et al. Dysregulated adaptive immunity is an early event in liver cirrhosis preceding acute-on-chronic liver failure[J]. Front Immunol, 2021, 11: 534731. DOI: 10.3389/fimmu.2020.534731. [11] ZHANG CX, GENG JW, XIE Q. Global disease burden and regional differences of acute-on-chronic liver failure: a review[J]. Chin Hepatol, 2021, 26(4): 355-358. DOI: 10.3969/j.issn.1008-1704.2021.04.003.张宸溪, 耿嘉蔚, 谢青. 慢加急性肝衰竭全球疾病负担及地域性差异研究进展[J]. 肝脏, 2021, 26(4): 355-358. DOI: 10.3969/j.issn.1008-1704.2021.04.003. [12] YADAV SK, SARAF N, CHOUDHARY NS, et al. Living donor liver transplantation for acute-on-chronic liver failure[J]. Liver Transpl, 2019, 25(3): 459-468. DOI: 10.1002/lt.25395. [13] DU FJ, SHAO C, ZHENG GZ. Therapeutic effect and nursing observation of artificial liver plasma replacement in severe hepatitis[J]. J Changchun Univ Chin Med, 2020, 36(2): 381-384. DOI: 10.13463/j.cnki.cczyy.2020.02.051.杜粉静, 邵灿, 郑鸽之. 人工肝血浆置换术在重症肝炎中的治疗效果及护理[J]. 长春中医药大学学报, 2020, 36(2): 381-384. DOI: 10.13463/j.cnki.cczyy.2020.02.051. [14] NOVELLI G, ANNESINI MC, MORABITO V, et al. Cytokine level modifications: molecular adsorbent recirculating system versus standard medical therapy[J]. Transplant Proc, 2009, 41(4): 1243-1248. DOI: 10.1016/j.transproceed.2009.03.035. [15] XU KL, LEI M, YUAN WF, et al. Effect of dual plasma molecular adsorption system in the treatment of hyperbilirubinemia in patients with liver failure[J]. Traum Crit Med, 2020, 8(2): 91-93, 96. DOI: 10.16048/j.issn.2095-5561.2020.02.08.许开亮, 雷鸣, 袁维方, 等. 双重血浆分子吸附系统治疗肝衰竭高胆红素患者疗效研究[J]. 创伤与急危重病医学, 2020, 8(2): 91-93, 96. DOI: 10.16048/j.issn.2095-5561.2020.02.08. [16] WIESNER R, EDWARDS E, FREEMAN R, et al. Model for end-stage liver disease (MELD) and allocation of donor livers[J]. Gastroenterology, 2003, 124(1): 91-96. DOI: 10.1053/gast.2003.50016. [17] HERNAEZ R, SOLÀ E, MOREAU R, et al. Acute-on-chronic liver failure: an update[J]. Gut, 2017, 66(3): 541-553. DOI: 10.1136/gutjnl-2016-312670. [18] JALAN R, SALIBA F, PAVESI M, et al. Development and validation of a prognostic score to predict mortality in patients with acute-on-chronic liver failure[J]. J Hepatol, 2014, 61(5): 1038-1047. DOI: 10.1016/j.jhep.2014.06.012. [19] DU L, MA Y, ZHOU S, et al. A prognostic score for patients with acute-on-chronic liver failure treated with plasma exchange-centered artificial liver support system[J]. Sci Rep, 2021, 11(1): 1469. DOI: 10.1038/s41598-021-81019-8. [20] MA S, XIE Z, ZHANG H, et al. Characterization of an artificial liver support system-related vasovagal reaction[J]. Biomed Res Int, 2020, 2020: 6313480. DOI: 10.1155/2020/6313480. -



 PDF下载 ( 2000 KB)
PDF下载 ( 2000 KB)

 下载:
下载:


