再代偿乙型肝炎肝硬化患者发生慢加急性肝衰竭的临床特点及预后分析
DOI: 10.3969/j.issn.1001-5256.2023.01.011
Clinical characteristics and prognosis of acute on chronic liver failure in patients with recompensatory hepatitis B cirrhosis
-
摘要:
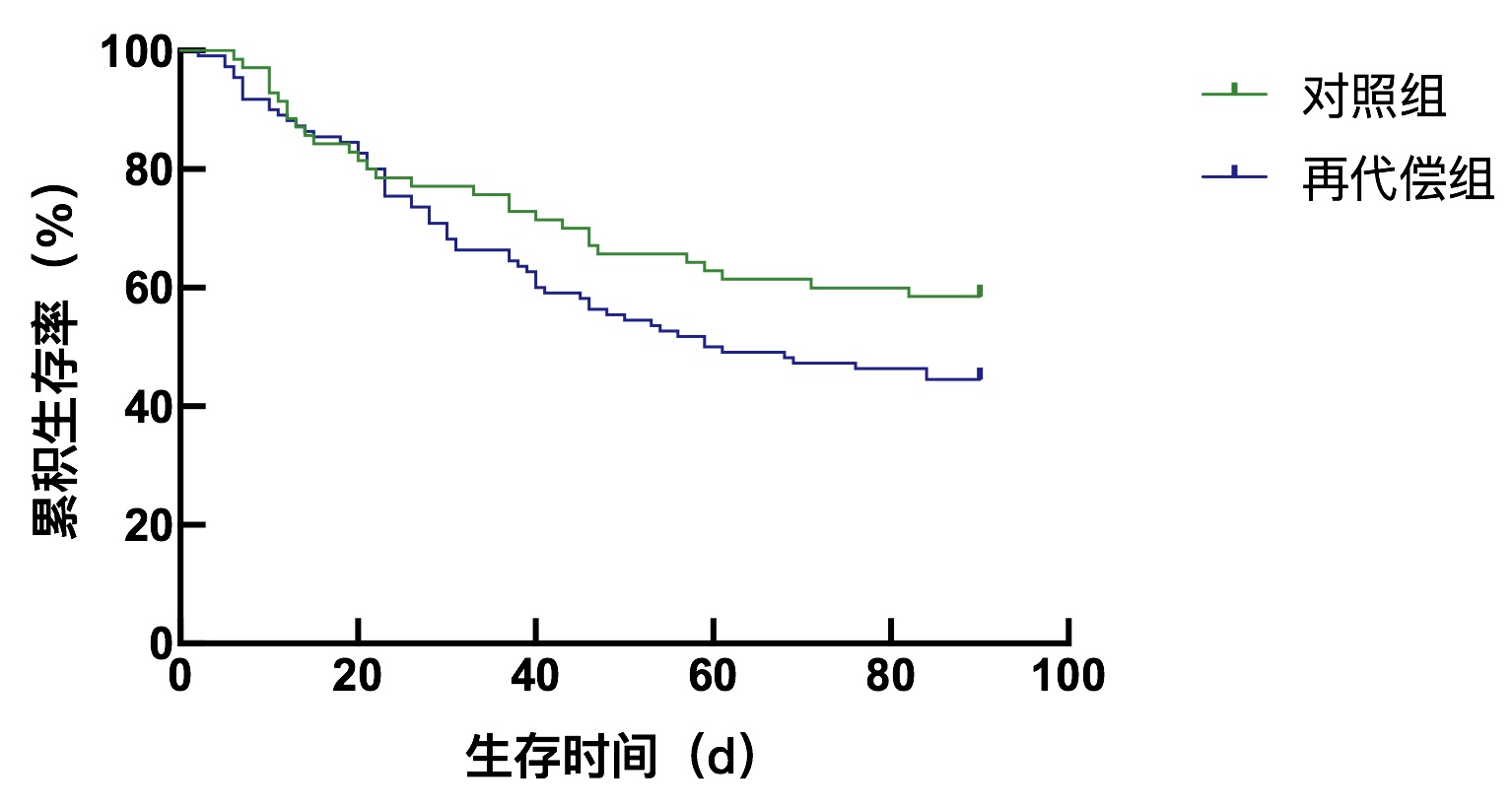
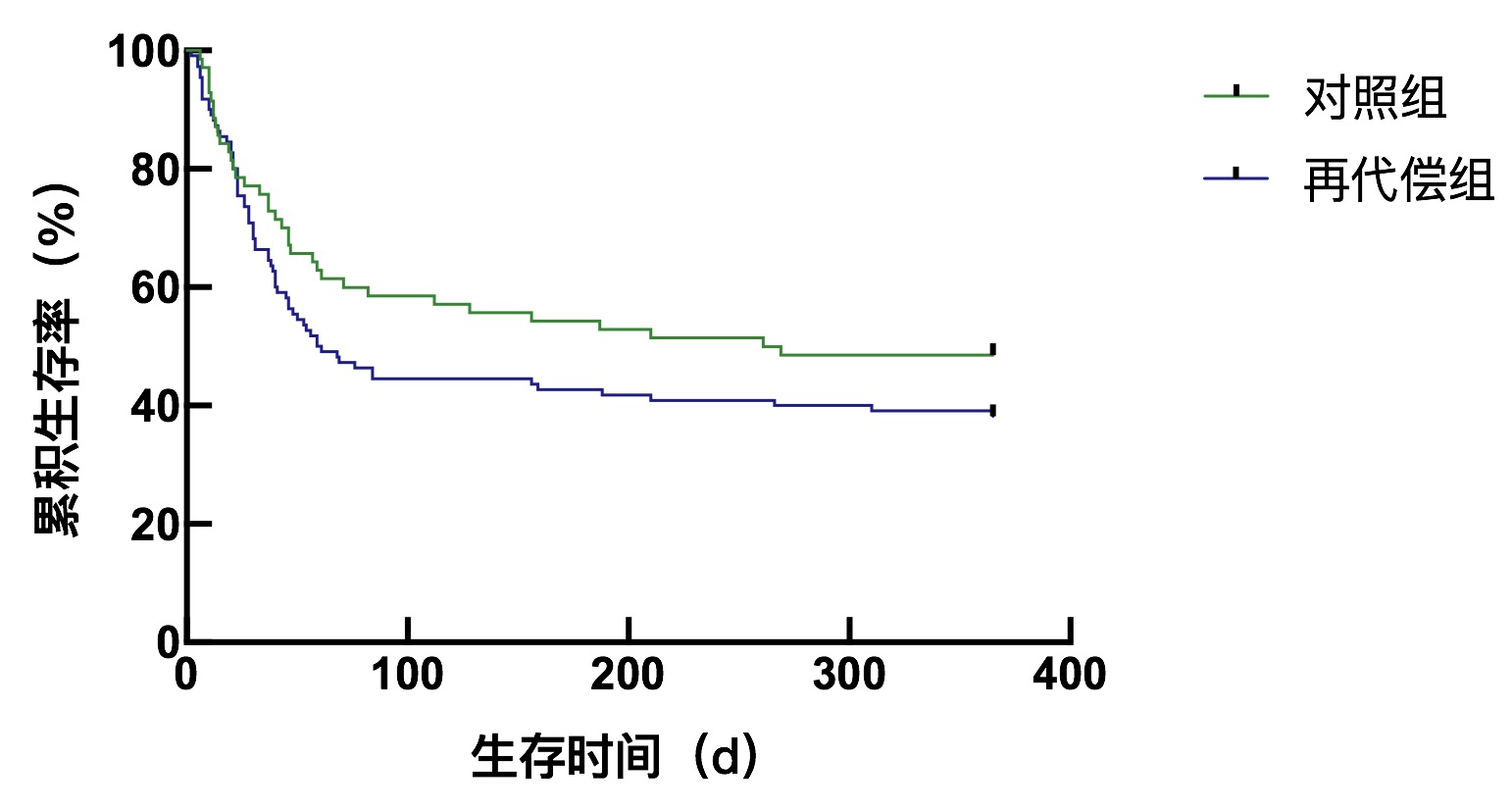
目的 探讨再代偿乙型肝炎肝硬化患者发生慢加急性肝衰竭的临床特点和预后情况。 方法 选取2013年9月—2021年9月于天津市第三中心医院住院治疗的慢加急性肝衰竭患者180例,其中乙型肝炎肝硬化再代偿患者110例,同期的代偿期乙型肝炎肝硬化患者70例作为对照组,比较其诱因、临床生化指标、并发症发生率和预后。两组间分类变量比较应用χ2检验或Fisher精确检验,对于连续性变量应用Mann-Whitney U检验。生存分析使用Kaplan-Meier方法,两组间比较采用Log-rank进行检验。 结果 再代偿组患者的肝肾综合征发生率(χ2=4.618,P=0.032)、感染发生率(χ2=6.712,P=0.010)、肌酐(Z=-4.508,P<0.001)、降钙素原(Z=-2.052,P=0.040)明显高于对照组。GGT(Z=-2.042,P=0.041)、Na(Z=-2.001,P=0.045)、空腹血糖(Z=-3.065,P=0.002)、TC(Z=-4.268,P<0.001)明显低于对照组。两组间90 d病死率(χ2=3.366,P=0.067)和1年病死率(χ2=1.893,P=0.169)比较差异均无统计学意义,经Log-rank检验,两组间90 d生存时间(χ2=2.680,P=0.100)和1年生存时间(χ2=2.074,P=0.150)比较差异均无统计学意义。 结论 与代偿期乙型肝炎肝硬化相比,再代偿后肝硬化患者发生慢加急性肝衰竭后肝肾综合征、感染及肌酐升高的发生风险增加,但90 d及1年的预后无明显差异。 Abstract:Objective To assess the clinical characteristics of acute-on-chronic liver failure in patients with recompensatory hepatitis B cirrhosis. Methods A total of 180 patients with acute-on-chronic liver failure hospitalized in Tianjin Third Central Hospital from September 2013 to September 2021 were retrospectively collected, with 110 patients had compensatory hepatitis B cirrhosis and 70 patients had compensatory hepatitis B cirrhosis and used as the control. Their causes, clinical biochemical indicators, complication rate, and prognosis were compared. The Chi-square test or Fisher's exact test was used for comparison of categorical variables between groups, and the Mann-Whitney U test was performed for analysis of the continuous variables. Kaplan-Meier curves and Log-rank test were used for survival of patients. Results The incidence of hepatorenal syndrome (χ2=4.618, P=0.032), infection (χ2=6.712, P=0.010), Cr (Z =-4.508, P < 0.001), and PCT (Z=-2.052, P=0.040) were all higher, whereas GGT (Z=-2.042, P=0.041), Na (Z=-2.001, P=0.045), FBS (Z=-3.065, P=0.002), and TC (Z=-4.268, P < 0.001) were all lower in the recompensation group than in the control group of patients. However, 90-day mortality rate (χ2=3.366, P=0.067) and 1-year mortality rate (χ2=1.893, P=0.169), 90-day survival (χ2=2.68, P=0.100), and 1-year survival (χ2=2.074, P=0.150) were not statistically significant difference. Conclusion Compared with compensatory hepatitis B cirrhosis, patients with recompensatory cirrhosis had an increased risk in developing hepatorenal syndrome, infection, and increased creatinine level after acute-on-chronic liver failure, although there was no statistically significant difference in 90-days and 1-year survival of patients. -
Key words:
- Hepatitis B /
- Liver Cirrhosis /
- Acute-On-Chronic Liver Failure /
- Prognosis
-
表 1 乙型肝炎肝硬化再代偿组和对照组人口学资料及入院前诱因比较
Table 1. Comparison of demographic data and prehospital indexes between the recompensatory hepatitis B cirrhosis group and the control group
指标 再代偿组(n=110) 对照组(n=70) 统计值 P值 男性[例(%)] 95(86.36) 58(82.86) χ2=0.413 0.521 年龄(岁) 49(43~58) 50(41~57) Z=-0.031 0.963 肝内因素[例(%)] HBV再激活 32(29.09) 25(35.71) χ2=0.867 0.352 酒精 8(7.27) 3(4.29) χ2=0.665 0.415 药物 4(3.64) 5(7.14) χ2=1.107 0.293 病毒再激活+酒精 15(13.64) 9(12.86) χ2=0.022 0.881 病毒再激活+药物 10(9.09) 5(7.14) χ2=0.213 0.645 肝外因素[例(%)] 感染 9(8.18) 12(17.14) χ2=3.333 0.068 出血 4(3.64) 4(5.71) χ2=0.435 0.510 过度劳累 10(9.09) 5(7.14) χ2=0.213 0.645 诱因不明 18(16.36) 2(2.86) χ2=7.901 0.005 表 2 乙型肝炎肝硬化再代偿组和对照组入院后各指标比较
Table 2. Comparison of indexes between the recompensatory hepatitis B cirrhosis group and the control group after admission
指标 再代偿组(n=110) 对照组(n=70) 统计值 P值 HE[例(%)] 26(23.64) 16(22.86) χ2=0.015 0.904 EVB[例(%)] 14(12.73) 8(11.43) χ2=0.336 0.562 HRS[例(%)] 49(44.55) 20(28.57) χ2=4.618 0.032 腹水[例(%)] 59(53.64) 45(64.29) χ2=1.989 0.158 感染[例(%)] 79(71.82) 37(52.86) χ2=6.712 0.010 呼吸道 28(25.45) 11(15.71) χ2=2.391 0.122 原发性腹膜炎 24(21.82) 9(12.86) χ2=2.294 0.130 泌尿道 10(9.09) 7(10.00) χ2=0.041 0.839 肠道 8(7.27) 5(7.14) χ2=0.001 0.974 皮肤软组织 9(8.18) 5(7.14) χ2=0.064 0.800 ALT (U/L) 121.50(61.68~291.75) 142.05(65.50~335.50) Z=-0.847 0.397 AST (U/L) 158.00(98.75~312.00) 169.00(96.90~360.25) Z=-0.021 0.984 ALP (U/L) 132.50(110.75~179.25) 146.50(113.40~184.73) Z=-0.847 0.397 GGT (U/L) 61.00(40.00~112.00) 92.00(48.68~139.15) Z=-2.042 0.041 TP (g/L) 59.00(54.00~65.00) 61.30(53.95~67.70) Z=-1.522 0.128 Alb (g/L) 29.25(26.00~33.00) 30.45(26.68~34.38) Z=-0.969 0.333 GLO (g/L) 29.55(25.00~34.35) 30.35(25.43~36.00) Z=-0.638 0.523 TBil (μmol/L) 320.35(209.88~414.95) 265.20(173.53~411.35) Z=-1.819 0.069 ChE (U/L) 2701(1848~3668) 2978(2021~3975) Z=-1.211 0.272 PTA (%) 35.20(28.15~41.05) 36.50(26.60~43.93) Z=-0.219 0.827 INR 2.05(1.76~2.45) 2.16(1.78~2.79) Z=-1.068 0.285 BUN (mmol/L) 5.65(4.15~8.08) 4.91(3.63~7.11) Z=-1.579 0.114 Cr (μmol/L) 82.50(69.50~100.25) 65.50(53.00~80.00) Z=-4.508 <0.001 Na (mmol/L) 134.80(130.30~137.00) 135.35(132.30~138.33) Z=-2.001 0.045 K (mmol/L) 3.99(3.62~4.40) 4.00(3.52~4.39) Z=-0.255 0.798 FBS (mmol/L) 4.75(4.00~6.45) 5.77(4.92~7.12) Z=-3.065 0.002 TG (mmol/L) 0.99(0.72~1.46) 1.11(0.84~1.55) Z=-0.722 0.413 TC (mmol/L) 1.42(1.07~2.27) 2.29(1.52~2.92) Z=-4.268 <0.001 Hb (g/L) 119.00(103.00~134.00) 128.50(108.75~140.25) Z=-1.916 0.055 WBC (×109/L) 6.66(4.70~9.32) 5.71(3.81~8.34) Z=-1.954 0.051 N (%) 73.35(63.80~80.13) 71.65(62.50~78.10) Z=-0.657 0.511 L (%) 15.88(10.18~22.18) 16.80(10.58~24.63) Z=-0.676 0.499 PLT (×109/L) 68.50(43.00~97.50) 77.00(46.75~103.75) Z=-0.734 0.463 NH3(μmol/L) 62(41~88) 76(49~101) Z=-1.769 0.084 AFP (ng/mL) 28.32(4.38~143.90) 56.59(10.14~216.63) Z=-1.732 0.085 PCT (ng/mL) 0.76(0.50~1.48) 0.50(0.34~1.08) Z=-2.052 0.040 CRP (μg/mL) 16.84(9.50~23.85) 12.53(7.37~20.60) Z=-1.503 0.123 Lac (mmol/L) 2.36(1.56~3.28) 2.49(2.10~3.04) Z=-0.693 0.433 HBV DNA阳性[例(%)] 57(51.82) 39(55.71) χ2=0.261 0.610 停药再激活[例(%)] 36(32.73) 22(31.43) χ2=0.033 0.856 变异耐药再激活[例(%)] 14(12.73) 11(15.71) χ2=0.319 0.572 免疫抑制剂再激活[例(%)] 7(6.36) 5(7.14) χ2=0.042 0.838 人工肝治疗[例(%)] 38(34.55) 29(41.43) χ2=0.867 0.352 90 d病死率[例(%)] 61(55.45) 29(41.43) χ2=3.366 0.067 1年病死率[例(%)] 68(61.82) 36(51.43) χ2=1.893 0.169 CTP评分 12(11~12) 11(10~12) Z=-1.232 0.263 MELD评分 26(24~29) 25(23~29) Z=-0.904 0.366 -
[1] LOK AS. Hepatitis: Long-term therapy of chronic hepatitis B reverses cirrhosis[J]. Nat Rev Gastroenterol Hepatol, 2013, 10(4): 199-200. DOI: 10.1038/nrgastro.2013.13. [2] MARCELLIN P, ASSELAH T. Long-term therapy for chronic hepatitis B: hepatitis B virus DNA suppression leading to cirrhosis reversal[J]. J Gastroenterol Hepatol, 2013, 28(6): 912-923. DOI: 10.1111/jgh.12213. [3] DIENSTAG JL, GOLDIN RD, HEATHCOTE EJ, et al. Histological outcome during long-term lamivudine therapy[J]. Gastroenterology, 2003, 124(1): 105-117. DOI: 10.1053/gast.2003.50013. [4] HADZIYANNIS SJ, TASSOPOULOS NC, HEATHCOTE EJ, et al. Long-term therapy with adefovir dipivoxil for HBeAg-negative chronic hepatitis B for up to 5 years[J]. Gastroenterology, 2006, 131(6): 1743-1751. DOI: 10.1053/j.gastro.2006.09.020. [5] KONG Y, SUN Y, ZHOU J, et al. Early steep decline of liver stiffness predicts histological reversal of fibrosis in chronic hepatitis B patients treated with entecavir[J]. J Viral Hepat, 2019, 26(5): 576-585. DOI: 10.1111/jvh.13058. [6] BUTI M, FUNG S, GANE E, et al. Long-term clinical outcomes in cirrhotic chronic hepatitis B patients treated with tenofovir disoproxil fumarate for up to 5 years[J]. Hepatol Int, 2015, 9(2): 243-250. DOI: 10.1007/s12072-015-9614-4. [7] SARIN SK, CHOUDHURY A, SHARMA MK, et al. Acute-on-chronic liver failure: consensus recommendations of the Asian Pacific association for the study of the liver (APASL): an update[J]. Hepatol Int, 2019, 13(4): 353-390. DOI: 10.1007/s12072-019-09946-3. [8] de FRANCHIS R, BOSCH J, GARCIA-TSAO G, et al. Baveno VⅡ - Renewing consensus in portal hypertension[J]. J Hepatol, 2022, 76(4): 959-974. DOI: 10.1016/j.jhep.2021.12.022. [9] Chinese Society of Hepatology, Chinese Society of Infectious Diseases, Chinese Medical Association. The guideline of prevention and treatment for chronic hepatitis B (2010 version)[J]. J Clin Hepatol, 2011, 27(1): Ⅰ-ⅩⅥ. https://www.cnki.com.cn/Article/CJFDTOTAL-LCGD201108002.htm中华医学会肝病学分会, 中华医学会感染病学分会. 慢性乙型肝炎防治指南(2010版)[J]. 临床肝胆病杂志, 2011, 27(1): Ⅰ-ⅩⅥ. https://www.cnki.com.cn/Article/CJFDTOTAL-LCGD201108002.htm [10] Chinese Society of Hepatology and Chinese Society of Infectious Diseases, Chinese Medical Association. The guideline of prevention and treatment for chronic hepatitis B: a 2015 update[J]. J Clin Hepatol, 2015, 31(12): 1941-1960. DOI: 10.3969/j.issn.1001-5256.2015.12.002.中华医学会肝病学分会, 中华医学会感染病学分会. 慢性乙型肝炎防治指南(2015年更新版)[J]. 临床肝胆病杂志, 2015, 31(12): 1941-1960. DOI: 10.3969/j.issn.1001-5256.2015.12.002. [11] Group of Liver Failure and Artificial Liver, Chinese Society of Infectious Diseases, Chinese Medical Association; Group of severe Liver disease and Artificial Liver, Chinese Society of Hepatology, Chinese Medical Association. Guidelines for diagnosis and treatment of hepatic failure (2012 edition)[J]. Inter J Epidemiol Infect Dis, 2013, 40(1): 1-7. DOI: 10.3760/cma.j.issn.1673-4149.2013.01.001.中华医学会感染病学分会肝衰竭与人工肝学组, 中华医学会肝病学分会重型肝病与人工肝学组. 肝衰竭诊治指南(2012年版)[J]. 国际流行病学传染病学杂志, 2013, 40(1): 1-7. DOI: 10.3760/cma.j.issn.1673-4149.2013.01.001. [12] Chinese Society of Hepatology, Chinese Medical Association. Chinese guidelines on the management of liver cirrhosis[J]. J Clin Hepatol, 2019, 35(11): 2408-2425. DOI: 10.3969/j.issn.1001-5256.2019.11.006.中华医学会肝病学分会. 肝硬化诊治指南[J]. 临床肝胆病杂志, 2019, 35(11): 2408-2425. DOI: 10.3969/j.issn.1001-5256.2019.11.006. [13] CAO ZJ, ZHANG CX, XIE Q. Paving the way to improve clinical management of acute-on-chronic liver failure using international criteria[J]. J Clin Hepatol, 2021, 37(4): 745-751. DOI: 10.3969/j.issn.1001-5256.2021.04.001.曹竹君, 张宸溪, 谢青. 慢加急性肝衰竭的国际标准与临床管理优化[J]. 临床肝胆病杂志, 2021, 37(4): 745-751. DOI: 10.3969/j.issn.1001-5256.2021.04.001. [14] MOREAU R, JALAN R, GINES P, et al. Acute-on-chronic liver failure is a distinct syndrome that develops in patients with acute decompensation of cirrhosis[J]. Gastroenterology, 2013, 144(7): 1426-1437. DOI: 10.1053/j.gastro.2013.02.042. [15] SHI Y, YANG Y, HU Y, et al. Acute-on-chronic liver failure precipitated by hepatic injury is distinct from that precipitated by extrahepatic insults[J]. Hepatology, 2015, 62(1): 232-242. DOI: 10.1002/hep.27795. [16] YIN S, WANG SJ, GU WY, et al. Risk of different precipitating events for progressing to acute-on-chronic liver failure in HBV-related cirrhotic patients[J]. J Dig Dis, 2017, 18(5): 292-301. DOI: 10.1111/1751-2980.12480. [17] CLÀRIA J, STAUBER RE, COENRAAD MJ, et al. Systemic inflammation in decompensated cirrhosis: Characterization and role in acute-on-chronic liver failure[J]. Hepatology, 2016, 64(4): 1249-1264. DOI: 10.1002/hep.28740. [18] DIRCHWOLF M, PODHORZER A, MARINO M, et al. Immune dysfunction in cirrhosis: Distinct cytokines phenotypes according to cirrhosis severity[J]. Cytokine, 2016, 77: 14-25. DOI: 10.1016/j.cyto.2015.10.006. [19] ALBILLOS A, LARIO M, ÁLVAREZ-MON M. Cirrhosis-associated immune dysfunction: distinctive features and clinical relevance[J]. J Hepatol, 2014, 61(6): 1385-1396. DOI: 10.1016/j.jhep.2014.08.010. [20] THABUT D, MASSARD J, GANGLOFF A, et al. Model for end-stage liver disease score and systemic inflammatory response are major prognostic factors in patients with cirrhosis and acute functional renal failure[J]. Hepatology, 2007, 46(6): 1872-1882. DOI: 10.1002/hep.21920. [21] FERNÁNDEZ J, ACEVEDO J, WIEST R, et al. Bacterial and fungal infections in acute-on-chronic liver failure: prevalence, characteristics and impact on prognosis[J]. Gut, 2018, 67(10): 1870-1880. DOI: 10.1136/gutjnl-2017-314240. [22] XU KC. Liver function detection technology and evaluation[M]//WANG BE, ZHANG DF. Modern Hepatology. Beijing: Science Press, 2003: 192.徐克成. 肝功能检测技术及评价[M]//王宝恩, 张定凤. 现代肝脏病学. 北京: 科学出版社, 2003: 192. [23] MEDZHITOV R, SCHNEIDER DS, SOARES MP. Disease tolerance as a defense strategy[J]. Science, 2012, 335(6071): 936-941. DOI: 10.1126/science.1214935. -



 PDF下载 ( 2214 KB)
PDF下载 ( 2214 KB)


 下载:
下载: