细胞毒性T淋巴细胞来源外泌体抑制肝星状细胞活化的机制
DOI: 10.3969/j.issn.1001-5256.2023.10.011
Mechanism of cytotoxic T lymphocyte-derived exosomes inhibiting hepatic stellate cell activation
-
摘要:
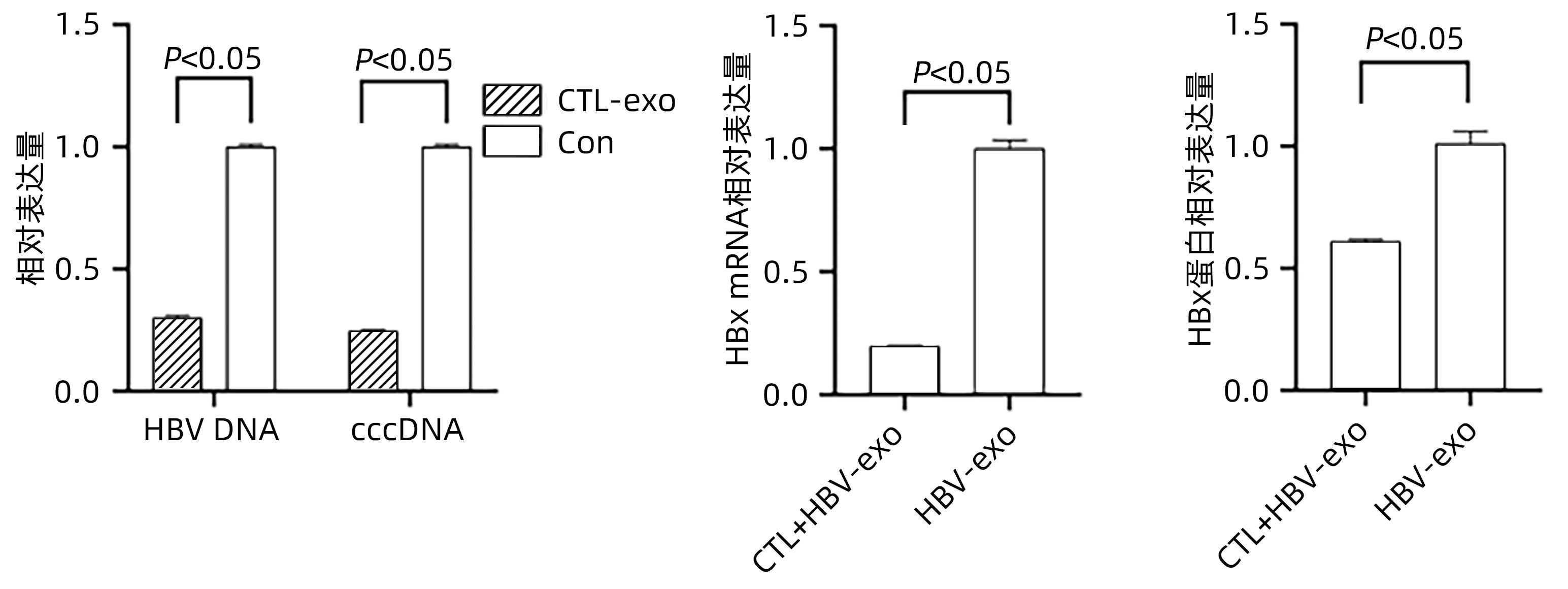
目的 探讨细胞毒性T淋巴细胞(CTL)来源外泌体能否下调HBx表达从而抑制肝星状细胞(HSC)活化。 方法 收集HepG2、HepGA14、CTL细胞上清液提取外泌体(分别简写为NC-exo、HBV-exo、CTL-exo),透射电镜观察其形态,Western Blot检测外泌体标志物CD63和TSG101的表达。将氟硼二吡咯染料(BODIPY)标记的NC-exo、HBV-exo以及CTL-exo与HBV-exo按不同比例混合后,分别与HSC LX-2(HSC-LX2)共培养,荧光显微镜观察外泌体能否进入LX-2细胞,倒置显微镜观察细胞形态学改变,实时荧光定量PCR (qPCR)检测LX-2细胞中TGF-β1、α-平滑肌肌动蛋白(α-SMA)、Ⅰ型胶原蛋白(Collagen1)等活化生物标志物的表达。将CTL-exo加入到HepGA14培养体系中,qPCR检测HepGA14细胞内HBV DNA、cccDNA及外泌体中HBx mRNA表达水平,Western Blot检测外泌体HBx蛋白表达水平。符合正态分布的计量资料两组间比较采用成组t检验,多组间比较采用单因素方差分析,进一步两两比较采用LSD-t检验。 结果 外泌体均为双层膜结构的微囊,呈圆形或椭圆形,囊泡的直径为50~100 nm,表达标志性蛋白CD63和TSG101。荧光显微镜观察示外泌体可进入LX-2细胞,并且HBV-exo进入LX-2细胞后,HSC胞体增大、胞突伸展。qPCR结果示NC-exo、HBV-exo、NC-exo+HBV-exo和Con组LX-2细胞中TGF-β1、α-SMA、Collagen1基因表达水平差异均有统计学意义(F值分别为444.678、417.144、571.508,P值均<0.05)。CTL-exo干预HepGA14细胞后,qPCR结果显示HepGA14细胞中HBV DNA、cccDNA表达水平较对照组明显下降(P值均<0.05),外泌体中HBx mRNA相对表达量明显下降(P<0.05);HBx蛋白表达水平与对照组相比也明显下降,差异有统计学意义(P<0.05)。CTL-exo和HBV-exo按照不同比例(2∶1、5∶1、10∶1)进行混合后干预LX-2细胞,qPCR结果显示各组间LX-2细胞中TGF-β1、α-SMA和Collagen1基因表达随着CTL-exo比例的增加而逐渐减弱,差异有统计学意义(P值均<0.05)。 结论 CTL-exo可下调HBV-exo中HBx蛋白表达抑制HSC活化,提示CTL-exo有抗乙型肝炎肝纤维化作用。 -
关键词:
- 肝纤维化 /
- T淋巴细胞, 细胞毒性 /
- 外泌体 /
- 肝星状细胞 /
- 乙型肝炎病毒X蛋白
Abstract:Objective To investigate whether cytotoxic T lymphocyte (CTL)-derived exosomes can downregulate HBx expression and inhibit hepatic stellate cell (HSC) activation. Methods The supernatants of HepG2, HepGA14, and CTL cells were collected to extract exosomes, which were referred to as NC-exo, HBV-exo, and CTL-exo, respectively). Transmission electron microscopy was used to observe their morphology, and Western Blot was used to measure the expression of the markers of exosomes CD63 and TSG101. NC-exo, HBV-exo, and CTL-exo labeled by BODIPY dye were mixed with HBV-exo at different ratios and were then co-cultured with HSC LX-2 (HSC-LX2). A fluorescence microscope was used to observe whether exosomes could enter LX-2 cells, and an fluorescence microscope was used to observe cell morphological changes; quantitative real-time PCR (qPCR) was used to measure the expression of the activated biomarkers such as transforming growth factor-β1 (TGF-β1), ɑ-smooth muscle actin (ɑ-SMA), and collagen type I (Collagen I) in LX-2 cells. CTL-exo was added to the HepGA14 culture system; then qPCR was used to measure the mRNA expression level of HBV DNA, cccDNA, and HBx in exosomes in HepGA14 cells, and Western Blot was used to measure the protein expression level of HBx in exosomes. The t-test was used for comparison of normally distributed continuous data between two groups; a one-way analysis of variance was used for comparison between multiple groups, and the least significant difference t-test was used for further comparison between two groups. Results The exosomes were all microcysts with a double-layer membrane structure and were circular or elliptical in shape, with the expression of the signature proteins CD63 and TSG101, and the vesicles had a diameter of 50-100 nm. The fluorescence microscope showed that exosomes could enter LX-2 cells, and HSC were enlarged with extended cell processes. The results of qPCR showed that there were significant differences in the expression levels of TGF-β1, ɑ-SMA, and Collagen I genes between the NC-exo, HBV-exo, NC-exo+HBV-exo, and Con groups (F=444.678, 417.144, and 571.508, all P<0.05). After the intervention of HepGA14 cells with CTL-exo, qPCR results showed that compared with the control group, there were significant reductions in the expression levels of HBV DNA and cccDNA in HepGA14 cells (all P<0.05), the relative mRNA expression level of HBx in exosomes (P<0.05), and the protein expression level of HBx (P<0.05). CTL-exo and HBV-exo were mixed at different ratios (2∶1, 5∶1, 10∶1) and were then used for the intervention of LX-2 cells, and qPCR results showed that the expression levels of TGF-β1, ɑ-SMA, and Collagen I genes in LX-2 cells gradually decreased with the increase in the ratio of CTL-exo between groups (P<0.05). Conclusion CTL-exo can downregulate the protein expression of HBx in HBV-exo to inhibit HSC activation, suggesting that CTL-exo has an anti-hepatitis B liver fibrosis effect. -
表 1 qPCR引物序列
Table 1. Real-time PCR primer sequences
基因 引物 HBV DNA F:5′-CTCGTGGTGGACTTCTCTC-3′ R:5′-CAGCAGGATGAAGAGGAA-3′ cccDNA F:5′-CTCCCCGTCTGTGCCTTCT-3′ R:5′-GCCCCAAAGCCACCCAAG-3′ HBx F:5′-TCTGTGCCTTCTCATCTGC-3′ R:5′-TCGGTCGTTGACATTGCTG-3′ TGF-β1 F:5′-GAGAAGCGGTACCTGAACCC-3′ R:5′-TGAACCCGTTGATGTCCACT-3′ Collagen1 F:5′-CATAAAGGGTCACCGTGGCT-3′ R:5′-AGTCCATCTTTGCCAGGAGC-3′ α-SMA F:5′-GGCAAGTGATCACCATCGGA-3′ R:5′-GTGGTTTCATGGATGCCAGC-3′ actin F:5′-ACACCCCAGCCATGTACGT-3′ R:5′-ATGGGCACAGTGTGGGTGA-3′ 表 2 qPCR检测不同组别中TGF-β1、α-SMA、Collagen1基因相对表达量
Table 2. qPCR was used to detect the relative expression of TGF-β1, α-SMA and Collagen1 genes in different groups
组别 TGF-β1 α-SMA Collagen1 NC-exo组 1.05 ± 0.03 1.07 ± 0.04 1.10 ± 0.03 NC-exo+HBV-exo组 1.29 ± 0.011)2)3) 1.48 ± 0.051)2)3) 1.44 ± 0.021)2)3) HBV-exo组 1.74 ± 0.051)2) 2.03 ± 0.041)2) 1.81 ± 0.031)2) Con组 1.00 ± 0.021) 1.00 ± 0.02 1.00 ± 0.031) F值 444.678 417.144 571.508 P值 <0.05 <0.05 <0.05 注:与NC-exo组比较,1)P<0.05;与Con组比较,2)P<0.05;与HBV-exo组比较,3)P<0.05。 表 3 不同HBV-exo、CTL-exo浓度比例干预后HSC-LX2细胞中TGF-β1、α-SMA、Collagen1的相对表达量
Table 3. Relative expressions of TGF-β1, α-SMA and Collagen1 in HSC-LX2 cells after different HBV-exo and CTL-exo concentration ratios
组别 TGF-β1 α-SMA Collagen1 HBV-exo组 3.18 ± 0.06 3.33 ± 0.11 2.76 ± 0.03 CTL-exo∶HBV-exo=2∶1组 2.19 ± 0.021)2) 2.32 ± 0.061)2) 1.94 ± 0.091)2) CTL-exo∶HBV-exo=5∶1组 1.47 ± 0.031)2)3) 1.55 ± 0.071)2)3) 1.34 ± 0.041)2)3) CTL-exo∶HBV-exo=10∶1组 1.20 ± 0.021)2)3)4) 1.17 ± 0.041)3)4) 1.07 ± 0.021)3)4) Con组 1.00 ± 0.021) 1.00 ± 0.001) 1.00 ± 0.031) F值 2 166.655 641.874 712.279 P值 <0.05 <0.05 <0.05 注:与HBV-exo组比较,1)P<0.05;与Con组比较,2)P<0.05;与CTL-exo∶HBV-exo=2∶1组比较,3)P<0.05;与CTL-exo∶HBV-exo=5∶1组比较,4)P<0.05。 -
[1] LI T, YAN Y, WANG B, et al. Exosomes derived from human umbilical cord mesenchymal stem cells alleviate liver fibrosis[J]. Stem Cells Dev, 2013, 22( 6): 845- 854. DOI: 10.1089/scd.2012.0395. [2] Chinese Society of Infectious Diseases, Chinese Medical Association, Chinese Society of Hepatology, Chinese Medical Association. Guidelines for the prevention and treatment of chronic hepatitis B(version 2019)[J]. J Clin Hepatol, 2019, 35( 12): 2648- 2669. DOI: 10.3969/j.issn.1001-5256.2019.12.007.中华医学会感染病学分会, 中华医学会肝病学分会. 慢性乙型肝炎防治指南(2019年版)[J]. 临床肝胆病杂志, 2019, 35( 12): 2648- 2669. DOI: 10.3969/j.issn.1001-5256.2019.12.007. [3] LIU J, LIANG W, JING W, et al. Countdown to 2030: eliminating hepatitis B disease, China[J]. Bull World Health Organ, 2019, 97( 3): 230- 238. DOI: 10.2471/BLT.18.219469. [4] LIU JW, XIE J, LI TS. Specific T-lymphocyte response and clinical outcome of patients with hepatitis B virus infection[J]. Med J Peking Union Med Coll Hosp, 2012, 3( 3): 350- 353. DOI: 10.3969/j.issn.1674-9081.2012.03.021.刘佳文, 谢静, 李太生. 乙型肝炎病毒感染者特异性T淋巴细胞应答与临床转归[J]. 协和医学杂志, 2012, 3( 3): 350- 353. DOI: 10.3969/j.issn.1674-9081.2012.03.021. [5] MA S, CHEN X, TAN Q, et al. An engineered novel lentivector specifically transducing dendritic cells and eliciting robust HBV-specific CTL response by upregulating autophagy in T cells[J]. Cell Cycle, 2018, 17( 10): 1220- 1234. DOI: 10.1080/15384101.2018.1471312. [6] CHENG ST, REN JH, CAI XF, et al. HBx-elevated SIRT2 promotes HBV replication and hepatocarcinogenesis[J]. Biochem Biophys Res Commun, 2018, 496( 3): 904- 910. DOI: 10.1016/j.bbrc.2018.01.127. [7] CHEN HY, CHEN ZX, HUANG RF, et al. Hepatitis B virus X protein activates human hepatic stellate cells through upregulating TGFβ1[J]. Genet Mol Res, 2014, 13( 4): 8645- 8656. DOI: 10.4238/2014.October.27.4. [8] QIU H, LIN DY, LI JY. Screening and identification of dominant monoclonal HepG2 cell strain with 1.3-fold HBV genome[J]. World Chin J Dig, 2021, 29( 16): 934- 944. DOI: 10.11569/wcjd.v29.i16.934.邱华, 林栋毅, 李锦源. HBV 1.3倍基因组HepG2稳转细胞模型优势单克隆株的筛选及鉴定[J]. 世界华人消化杂志, 2021, 29( 16): 934- 944. DOI: 10.11569/wcjd.v29.i16.934. [9] National Health Commission. China health year book 2019[M]. Beijing: Peking Union Medical College Press, 2019.国家卫生健康委员会. 2019 中国卫生健康统计年鉴[M]. 北京: 中国协和医科大学出版社, 2019. [10] ROCKEY DC. Liver fibrosis reversion after suppression of hepatitis B virus[J]. Clin Liver Dis, 2016, 20( 4): 667- 679. DOI: 10.1016/j.cld.2016.06.003. [11] CHANG TT, LIAW YF, WU SS, et al. Long-term entecavir therapy results in the reversal of fibrosis/cirrhosis and continued histological improvement in patients with chronic hepatitis B[J]. Hepatology, 2010, 52( 3): 886- 893. DOI: 10.1002/hep.23785. [12] TZIOMALOS K. Effect of antiviral treatment on the risk of hepatocellular carcinoma in patients with chronic hepatitis B[J]. World J Hepatol, 2010, 2( 3): 91- 93. DOI: 10.4254/wjh.v2.i3.91. [13] CHEN BW, ZHU XJ, ZHANG X, et al. Influence of virologic response on disease progression in patients with compensated hepatitis B cirrhosis[J]. J Clin Hepatol, 2021, 37( 8): 1811- 1816. DOI: 10.3969/j.issn.1001-5256.2021.08.014.陈博武, 朱晓骏, 张鑫, 等. 病毒学应答状态对代偿期乙型肝炎肝硬化患者疾病进展的影响[J]. 临床肝胆病杂志, 2021, 37( 8): 1811- 1816. DOI: 10.3969/j.issn.1001-5256.2021.08.014. [14] GIERSCH K, ALLWEISS L, VOLZ T, et al. Serum HBV pgRNA as a clinical marker for cccDNA activity[J]. J Hepatol, 2017, 66( 2): 460- 462. DOI: 10.1016/j.jhep.2016.09.028. [15] MOHAMMADI A, TAJIK N, SHAH-HOSSEINI A, et al. FAS and FAS-ligand promoter polymorphisms in hepatitis B virus infection[J]. Hepat Mon, 2015, 15( 10): e26490. DOI: 10.5812/hepatmon.26490. [16] KONDO Y, KOBAYASHI K, ASABE S, et al. Vigorous response of cytotoxic T lymphocytes associated with systemic activation of CD8 T lymphocytes in fulminant hepatitis B[J]. Liver Int, 2004, 24( 6): 561- 567. DOI: 10.1111/j.1478-3231.2004.0982.x. [17] HAQUE M, LEI F, XIONG X, et al. Stem cell-derived viral antigen-specific T cells suppress HBV replication through production of IFN-γ and TNF-α[J]. iScience, 2020, 23( 7): 101333. DOI: 10.1016/j.isci.2020.101333. [18] MOON B, CHANG S. Exosome as a delivery vehicle for cancer therapy[J]. Cells, 2022, 11( 3): 316. DOI: 10.3390/cells11030316. [19] COCUCCI E, MELDOLESI J. Ectosomes and exosomes: shedding the confusion between extracellular vesicles[J]. Trends Cell Biol, 2015, 25( 6): 364- 372. DOI: 10.1016/j.tcb.2015.01.004. [20] JIA Y, LI Y, WANG YJ. Research progress on exosome targeted delivery of nucleic acid molecules[J]. China Med Herald, 2023, 20( 3): 33- 36, 49. DOI: 10.20047/j.issn1673-7210.2023.03.07.贾岳, 李妍, 王英骥. 外泌体靶向递送核酸类分子研究进展[J]. 中国医药导报, 2023, 20( 3): 33- 36, 49. DOI: 10.20047/j.issn1673-7210.2023.03.07. [21] SUN JS, LU MY. Research progress on the formation of exosomes and their rolesin immune regulation[J]. J Tianjin Normal University(Natural Science Edition), 2020, 40( 1): 9- 15. DOI: 10.19638/j.issn1671-1114.20200102.孙金生, 陆美伊. 外泌体形成及在免疫调节中的作用研究进展[J]. 天津师范大学学报(自然科学版), 2020, 40( 1): 9- 15. DOI: 10.19638/j.issn1671-1114.20200102. [22] YANG F, YANG T, ZHOU WJ, et al. Macrophage-derived exosomes inhibit HBV DNA replication[J]. Infect Dis Info, 2018, 31( 2): 125- 130. DOI: 10.3969/j.issn.1007-8134.2018.02.008.杨帆, 杨涛, 周文靖, 等. 巨噬细胞外泌体抑制HBV DNA复制[J]. 传染病信息, 2018, 31( 2): 125- 130. DOI: 10.3969/j.issn.1007-8134.2018.02.008. [23] ABDULKARIM AS, CAO H, HUANG B, et al. The large GTPase dynamin is required for hepatitis B virus protein secretion from hepatocytes[J]. J Hepatol, 2003, 38( 1): 76- 83. DOI: 10.1016/s0168-8278(02)00326-4. -



 PDF下载 ( 1118 KB)
PDF下载 ( 1118 KB)


 下载:
下载: