Establishment of a predictive model for the prognosis of patients with hepatitis B virus-related acute-on-chronic liver failure treated with plasma exchange and double plasma molecular adsorption system alone or in combination
-
摘要:
目的 观察采用血浆置换(PE)与双重血浆分子吸附系统(DPMAS)单用或联合使用(PE+DPMAS)治疗的HBV感染相关慢加急性肝衰竭(HBV-ACLF)患者的24周生存情况,建立评估患者24周预后的预测模型。 方法 收集2015年1月—2019年12月在安徽医科大学附属省立医院行PE+DPMAS治疗的133例HBV-ACLF患者的临床资料,根据治疗后24周随访生存情况分为生存组(n=71)和死亡组(n=62)。另收集2018年1月—2020年1月在安徽医科大学第二附属医院接受PE+DPMAS治疗的55例HBV-ACLF患者为验证组验证模型效能。收集临床资料包括采用人工肝治疗的模式、性别、年龄、TBil、INR、肌酐(Cr)、血清钠、PLT、Alb,是否有腹水、肝肾综合征、肝性脑病和消化道出血。正态分布的计量资料2组间比较采用t检验,偏态分布的计量资料2组间比较采用Wilcoxon秩和检验; 计数资料组间比较采用χ2检验和Fisher检验;PE+DPMAS治疗的HBV-ACLF患者预后因素分析采用Cox回归分析法,并建立预测模型;绘制受试者工作特征曲线(ROC曲线),采用DeLong法比较新的预测模型与MELD及MELD-Na评分的ROC曲线下面积(AUC)。 结果 治疗后24周建模组共存活71例,死亡62例。Cox回归分析显示年龄(HR=1.030,P=0.013)、TBil(HR=1.018,P<0.001)、INR(HR=1.517,P<0.001)和PLT(HR=0.993,P=0.04)是患者24周生存的独立影响因素。根据Cox回归分析结果建立了行PE+DPMAS治疗的HBV-ACLF患者的预后模型(ATIP)=0.029×年龄(岁)+0.018×TBil(mg/dL)+0.417×INR-0.007×PLT(109/L)。建模组和验证组均显示ATIP模型的预测效能优于MELD和MELD-Na评分(P值均<0.05)。 结论 行PE+DPMAS治疗的HBV-ACLF患者的年龄、TBil、INR和PLT为24周生存的独立影响因素;ATIP模型对行PE+DPMAS治疗的HBV-ACLF患者的24周预后预测效能良好。 Abstract:Objective To observe the 24-week survival status of patients with hepatitis B virus-related acute-on-chronic liver failure (HBV-ACLF) treated with plasma exchange (PE) and double plasma molecular adsorption system (DPMAS) alone or in combination, and to establish a predictive model for 24-week prognosis. Methods Related clinical data were collected from 133 patients with HBV-ACLF who received PE and DPMAS alone or in combination in The Affiliated Provincial Hospital of Anhui Medical University from January 2015 to December 2019, and according to the survival status at the 24-week follow-up after treatment, they were divided into survival group with 71 patients and death group with 62 patients. A total of 55 patients with HBV-ACLF who received PE and DPMAS alone or in combination in The Second Affiliated Hospital of Anhui Medical University from January 2018 to January 2020 were enrolled as validation group to validate the performance of the model. Related clinical data included mode of artificial liver support therapy, age, sex, total bilirubin (TBil), international normalized ratio (INR), creatinine (Cr), serum sodium, platelet count (PLT), albumin (Alb), and presence or absence of ascites, hepatorenal syndrome, hepatic encephalopathy, and gastrointestinal bleeding. The t-test was used for comparison of normally distributed continuous data between two groups, and the Wilcoxon rank-sum test was used for comparison of continuous data with skewed distribution between two groups; the chi-square test and the Fisher's exact test were used for comparison of categorical data between groups. The Cox regression model was used to analyze the influencing factors for the prognosis of HBV-ACLF patients after PE and DPMAS alone or in combination and establish a predictive model; the receiver operator characteristic (ROC) curve was plotted and the DeLong method was used to compare the area under the ROC curve (AUC) between the new predictive model and Model for End-Stage Liver Disease (MELD)/MELD combined with serum sodium concentration (MELD-Na) scores. Results At 24 weeks after treatment, 71 patients survived and 62 patients died in the modeling group. The Cox regression analysis showed age (hazard ratio [HR]=1.030, P=0.013), TBil (HR=1.018, P < 0.001), INR (HR=1.517, P < 0.001), and PLT (HR=0.993, P=0.04) were independent influencing factors for 24-week survival. According to the results of the Cox regression analysis, a prognostic model for HBV-ACLF patients treated with PE and DPMAS alone or in combination was established as ATIP=0.029×age (years)+0.018×TBil (mg/dL)+0.417×INR-0.007×PLT (109/L). Both the modeling group and the validation group showed that the ATIP model had a better predictive performance than MELD and MELD-NA scores(all P < 0.05). Conclusion Age, TBil, INR, and PLT are independent influencing factors for the 24-week survival of HBV-ACLF patients treated with PE and DPMAS alone or in combination, and the ATIP model has a good performance in predicting the 24-week prognosis of HBV-ACLF patients treated with PE and DPMAS alone or in combination. -
Key words:
- Liver Failure /
- Hepatitis B virus /
- Liver, Artificial /
- Prognosis
-
表 1 建模组和验证组患者一般资料比较
指标 建模组(n=133) 验证组(n=55) 统计值 P值 年龄(岁) 47.89±11.98 51.15±14.83 t=1.575 0.153 女/男(例) 17/116 11/44 χ2=1.081 0.746 腹水[例(%)] 85(63.91) 31(56.36) χ2=0.645 0.422 肝肾综合征[例(%)] 21(15.79) 11(20.00) χ2=0.236 0.627 肝性脑病[例(%)] 28(21.05) 8(14.55) χ2=0.685 0.408 消化道出血[例(%)] 9(6.77) 1(1.82) 0.286 TBil(mg/dL) 23.88(18.92~27.44) 21.8(15.79~26.56) Z=-1.791 0.073 INR 2.18(1.66~2.72) 1.64(1.51~2.21) Z=-3.20 0.001 Alb(g/dL) 32.0(29.3~34.7) 32.6(29.8~36.5) Z=-1.416 0.157 Na(mmol/L) 136(134~139) 137(133~138) Z=-0.983 0.326 Cr(mg/dL) 0.78(0.66~0.96) 0.61(0.48~0.77) Z=-4.158 <0.001 PLT(109/L) 89(59~119) 106(56~181) Z=-1.513 0.131 MELD 24.67(21.00~29.36) 19.23(16.23~26.00) Z=-4.061 <0.001 MELD-Na 25.66(22.14~31.93) 21.03(16.75~28.16) Z=-3.042 0.002 ALSS模式[例(%)] χ2=1.600 0.449 PE 45(33.83) 21(38.18) DPMAS 39(29.32) 19(34.55) PE+DPMAS 49(36.84) 15(23.27) 表 2 建模组患者24周生存预后的单因素和多因素Cox回归分析
参数 单因素Cox回归分析 多因素Cox回归分析 B值 SE HR(95%CI) P值 B值 SE HR(95%CI) P值 年龄 0.044 0.010 1.045(1.025~1.065) <0.001 0.029 0.012 1.030(1.006~1.054) 0.013 性别 -0.201 0.401 0.818(0.372~1.795) 0.616 腹水 0.468 0.285 1.597(0.914~2.792) 0.100 肝肾综合征 0.814 0.291 2.257(1.276~3.995) 0.005 肝性脑病 0.833 0.273 2.301(1.348~3.928) 0.002 消化道出血 1.011 0.381 2.749(1.304~5.795) 0.008 TBil 0.017 0.005 1.017(1.007~1.026) 0.001 0.018 0.005 1.018(1.008~1.028) <0.001 INR 0.435 0.079 1.546(1.323~1.807) <0.001 0.417 0.109 1.517(1.226~1.877) <0.001 Alb -0.043 0.020 0.958(0.920~0.997) 0.035 Na -0.050 0.026 0.952(0.940~1.001) 0.057 Cr 0.765 0.316 2.149(1.157~3.991) 0.015 PLT -0.010 0.003 0.990(0.984~0.996) 0.002 -0.007 0.004 0.993(0.986~1.000) 0.040 ALSS模式 PE DPMAS 0.25653 0.31632 1.292(0.695~2.402) 0.417 PE+DPMAS 0.05008 0.30897 1.051(0.574~1.926) 0.871 表 3 行PE+DPMAS治疗的HBV-ACLF患者预后评分系统预测价值比较
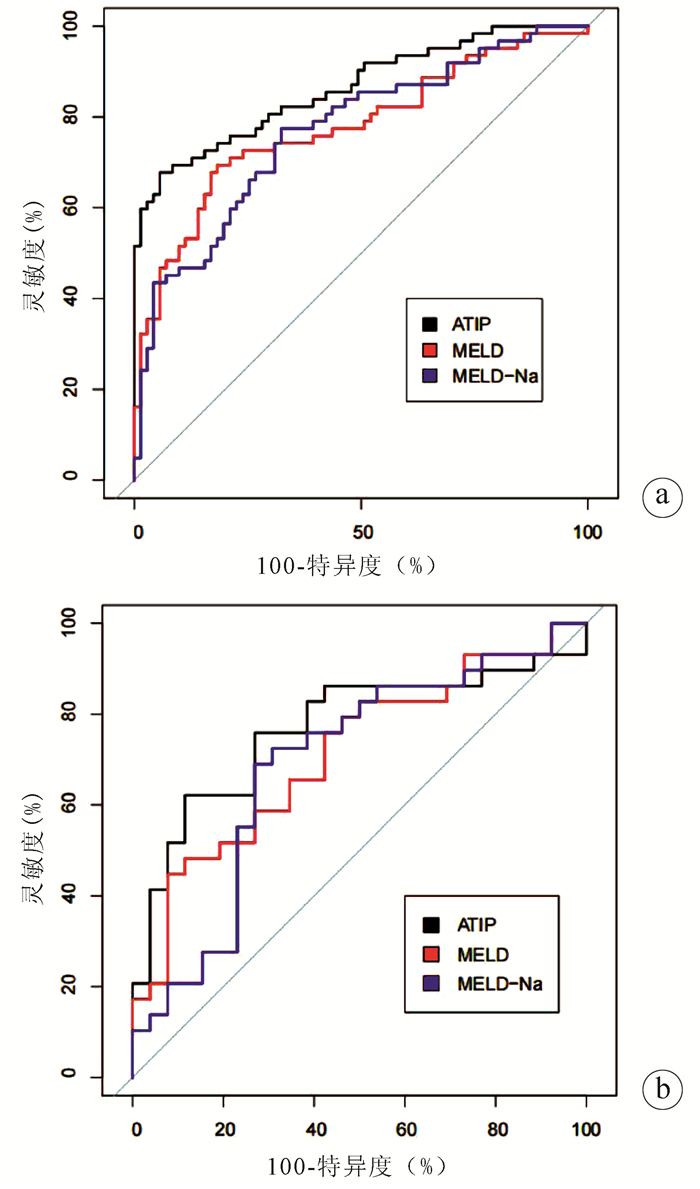
模型 建模组 验证组 灵敏度(%) 特异度(%) cut off值 AUC 95% CI AUC 95% CI ATIP 67.74 94.37 0.31 0.863 0.800~0.926 0.768 0.636~0.899 MELD 69.35 81.69 25.55 0.7771) 0.695~0.858 0.7121) 0.575~0.850 MELD-Na 77.42 67.61 25.22 0.7681) 0.688~0.849 0.6911) 0.545~0.837 注:与ATIP比较,1) P<0.05。 -
[1] LIU J, FAN D. Hepatitis B in China[J]. Lancet, 2007, 369(9573): 1582-1583. DOI: 10.1016/S0140-6736(07)60723-5. [2] Liver Failure and Artificial Liver Group, Chinese Society of Infectious Diseases, Chinese Medical Association; Severe Liver Disease and Artificial Liver Group, Chinese Society of Hepatology, Chinese Medical Association. Guideline for diagnosis and treatment of liver failure[J]. J Clin Hepatol, 2019, 35(1): 38-44. DOI: 10.3969/j.issn.1001-5256.2019.01.007.中华医学会感染病学分会肝衰竭与人工肝学组, 中华医学会肝病学分会重型肝病与人工肝学组. 肝衰竭诊治指南(2018年版)[J]. 临床肝胆病杂志, 2019, 35(1): 38-44. DOI: 10.3969/j.issn.1001-5256.2019.01.007. [3] SARIN SK, CHOUDHURY A. Acute-on-chronic liver failure: Terminology, mechanisms and management[J]. Nat Rev Gastroenterol Hepatol, 2016, 13(3): 131-149. DOI: 10.1038/nrgastro.2015.219. [4] REEVES HM, WINTERS JL. The mechanisms of action of plasma exchange[J]. Br J Haematol, 2014, 164(3): 342-351. DOI: 10.1111/bjh.12629. [5] YAO J, LI S, ZHOU L, et al. Therapeutic effect of double plasma molecular adsorption system and sequential half-dose plasma exchange in patients with HBV-related acute-on-chronic liver failure[J]. J Clin Apher, 2019, 34(4): 392-398. DOI: 10.1002/jca.21690. [6] KAMATH PS, WIESNER RH, MALINCHOC M, et al. A model to predict survival in patients with end-stage liver disease[J]. Hepatology, 2001, 33(2): 464-470. DOI: 10.1053/jhep.2001.22172. [7] BIGGINS SW, KIM WR, TERRAULT NA, et al. Evidence-based incorporation of serum sodium concentration into MELD[J]. Gastroenterology, 2006, 130(6): 1652-1660. DOI: 10.1053/j.gastro.2006.02.010. [8] ZOULIM F, LOCAMINI S. Hepatitis B virus resistance to nucleos(t)ide analogues[J]. Gastroenterology, 2009, 137: 1593-1608. e1-2. DOI: 10.1053/j.gastro.2009.08.063. [9] LING Q, XU X, WEI Q, et al. Downgrading MELD improves the outcomes after liver transplantation in patients with acute-on-chronic hepatitis B liver failure[J]. PLoS One, 2012, 7(1): e30322. DOI: 10.1371/journal.pone.0030322. [10] YU JW, SUN LJ, ZHAO YH, et al. Prediction value of model for end-stage liver disease scoring system on prognosis in patients with acute-on-chronic hepatitis B liver failure after plasma exchange and lamivudine treatment[J]. J Gastroenterol Hepatol, 2008, 23(8 Pt 1): 1242-1249. DOI: 10.1111/j.1440-1746.2008.05484.x. [11] CHEN JJ, HUANG JR, YANG Q, et al. Plasma exchange-centered artificial liver support system in hepatitis B virus-related acute-on-chronic liver failure: A nationwide prospective multicenter study in China[J]. Hepatobiliary Pancreat Dis Int, 2016, 15(3): 275-281. DOI: 10.1016/s1499-3872(16)60084-x. [12] XU X, LIU X, LING Q, et al. Artificial liver support system combined with liver transplantation in the treatment of patients with acute-on-chronic liver failure[J]. PLoS One, 2013, 8(3): e58738. DOI: 10.1371/journal.pone.0058738. [13] WIESNER R, EDWARDS E, FREEMAN R, et al. Model for end-stage liver disease (MELD) and allocation of donor livers[J]. Gastroenterology, 2003, 124(1): 91-96. DOI: 10.1053/gast.2003.50016. [14] ROUILLARD SS, BASS NM, ROBERTS JP, et al. Severe hyperbilirubinemia after creation of transjugular intrahepatic portosystemic shunts: Natural history and predictors of outcome[J]. Ann Intern Med, 1998, 128(5): 374-377. DOI: 10.7326/0003-4819-128-5-199803010-00006. [15] INTAGLIATA NM, DAVIS J, CALDWELL SH. Coagulation pathways, hemostasis, and thrombosis in liver failure[J]. Semin Respir Crit Care Med, 2018, 39(5): 598-608. DOI: 10.1055/s-0038-1673658. [16] HARUKI K, SHIBA H, SAITO N, et al. Risk stratification using a novel liver functional reserve score of combination prothrombin time-international normalized ratio to albumin ratio and albumin in patients with hepatocellular carcinoma[J]. Surgery, 2018, 164(3): 404-410. DOI: 10.1016/j.surg.2018.02.022. [17] HU XP, GAO J. International normalized ratio and Model for End-stage Liver Disease score predict short-term outcome in cirrhotic patients after the resolution of hepatic encephalopathy[J]. World J Gastroenterol, 2019, 25(26): 3426-3437. DOI: 10.3748/wjg.v25.i26.3426. [18] O'LEARY JG, GREENBERG CS, PATTON HM, et al. AGA clinical practice update: Coagulation in cirrhosis[J]. Gastroenterology, 2019, 157(1): 34-43. e1. DOI: 10.1053/j.gastro.2019.03.070. [19] XIA Q, DAI X, ZHANG Y, et al. A modified MELD model for Chinese pre-ACLF and ACLF patients and it reveals poor prognosis in pre-ACLF patients[J]. PLoS One, 2013, 8(6): e64379. DOI: 10.1371/journal.pone.0064379. [20] LU XC, LU D, PAN Y. Clinical significance of platelet parameters in diagnosis and treatment of acute leukemia[J]. Int J Lab Med, 2006, 27(10): 870-871, 873. DOI: 10.3969/j.issn.1673-4130.2006.10.003.陆小婵, 卢冬, 潘云. 血小板参数在急性白血病疗效观察中的应用[J]. 国际检验医学杂志, 2006, 27(10): 870-871, 873. DOI: 10.3969/j.issn.1673-4130.2006.10.003. [21] PECK-RADOSAVLJEVIC M, WICHLAS M, ZACHERL J, et al. Thrombopoietin induces rapid resolution of thrombocytopenia after orthotopic liver transplantation through increased platelet production[J]. Blood, 2000, 95(3): 795-801. DOI: 10.1182/blood.V95.3.795.003k25_795_801 [22] HITCHCOCK IS, KAUSHANSKY K. Thrombopoietin from beginning to end[J]. Br J Haematol, 2014, 165(2): 259-268. DOI: 10.1111/bjh.12772. [23] OLARIU M, OLARIU C, OLTEANU D. Thrombocytopenia in chronic hepatitis C[J]. J Gastrointestin Liver Dis, 2010, 19(4): 381-385. http://193.231.29.96/jgld/2010/4/7.pdf [24] SAKAI K, IWAO T, OHO K, et al. Propranolol ameliorates thrombocytopenia in patients with cirrhosis[J]. J Gastroenterol, 2002, 37(2): 112-118. DOI: 10.1007/s005350200005. [25] MA Z, WU Y. Current status of liver failure treatment[J]. J Clin Hepatol, 2016, 32(9) : 1668-1672. DOI: 10.3969/ j.issn.1001- 5256.2016.09.007.马臻, 乌云. 肝衰竭的治疗现状[J]. 临床肝胆病杂志, 2016, 32(9): 1668- 1672. DOI: 10.3969/ j.issn.1001- 5256.2016.09.007. [26] XU SS, WEI XH, LIN W, et al. Clinical significance of platelet count and its dynamic change in patients with acute- on-chronic liver failure[J]. J CIin Hepatol, 2018, 34(4): 810-813. DOI: 10. 3969/ j. issn. 1001- 5256. 2018. 04. 023.许姗姗, 韦新焕, 林伟, 等. 慢加急性肝衰竭患者血小板计数及其动态变化的临床意义[J]. 临床肝胆病杂志, 2018, 34(4): 810-813. DOI: 10.3969/j.issn.1001-5256.2018.04.023. [27] SHI XX, ZHANG YQ, ZHU P, et al. Prognostic risk factors in patients with acute- on- chronic hepatitis B liver failure[J]. J Clin Hepatol, 2016, 32(4): 700-705. DOI: 10.3969/j.issn.1001-5256. 2016. 04. 018.石新星, 张艳琼, 朱鹏, 等. 乙型肝炎相关慢加急性肝衰竭患者预后的危险因素分析[J]. 临床肝胆病杂志, 2016, 32(4): 700-705. DOI: 10.3969/j.issn.1001-5256.2016.04.018 [28] LI WY, ZHANG MX, QI TT, et al. The potential factors contributing to thrombocytopenia in acute-on-chronic liver failure patients[J]. Chin Hepatol, 2015, 20(6): 457-461. DOI: 10.3969/j.issn.1008-1704.2015.06.009李文燕, 张明霞, 祁婷婷, 等. 慢加急性肝衰竭患者血小板减少的可能原因[J]. 肝脏, 2015, 20(6): 457-461. DOI: 10.3969/j.issn.1008-1704.2015.06.009 [29] LESURTEL M, GRAF R, ALEIL B, et al. Platelet-derived serotonin mediates liver regeneration[J]. Science, 2006, 312(5770): 104-107. DOI: 10.1126/science.1123842. -



 PDF下载 ( 2400 KB)
PDF下载 ( 2400 KB)

 下载:
下载:


