抑郁对原发性胆汁性胆管炎患者熊去氧胆酸治疗应答和发生肝硬化的影响
DOI: 10.3969/j.issn.1001-5256.2023.12.011
Effect of depression on response to ursodeoxycholic acid and the occurrence of liver cirrhosis in patients with primary biliary cholangitis
-
摘要:
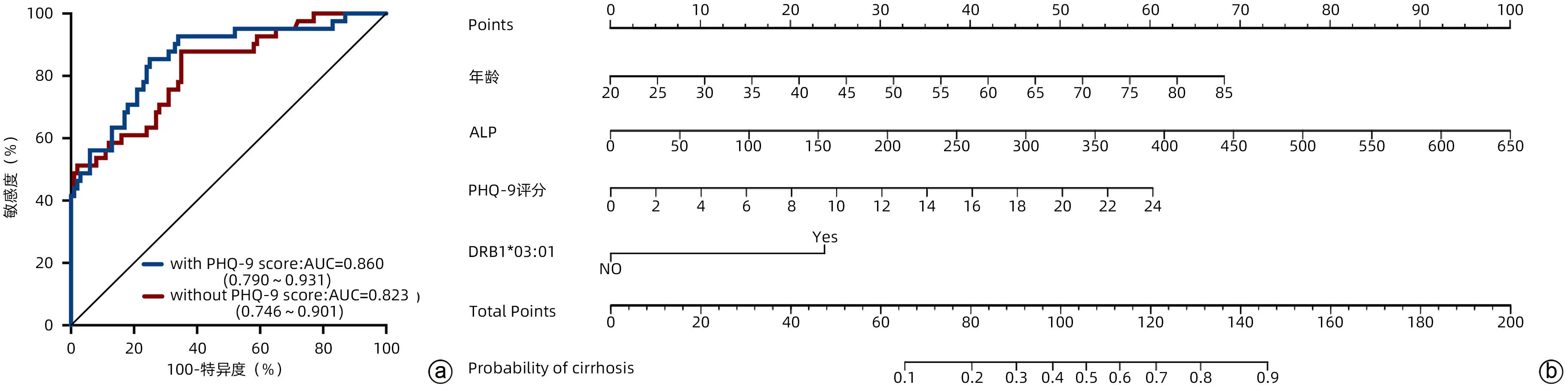
目的 原发性胆汁性胆管炎(PBC)患者常伴发抑郁,但抑郁在PBC疾病进展中的作用尚不清楚。本文拟探讨抑郁与PBC治疗应答的关系及其对PBC患者肝硬化的影响。 方法 回顾性分析2018年1月—2020年12月在天津医科大学总医院自身免疫性肝病门诊就诊的141例接受单药熊去氧胆酸(UDCA)标准治疗1年的PBC患者的临床资料。从健康体检中心选取的年龄及性别匹配的170例健康对照者作为对照组。应用患者健康问卷抑郁量表(PHQ-9)评估PBC患者及健康对照者的抑郁状态。正态分布的计量资料两组间比较采用成组t检验,非正态分布的计量资料两组间比较采用Mann-Whitney U检验。计数资料两组间比较采用χ2检验。应用二元Logistic回归模型及决策树模型分析PBC患者肝硬化的影响因素,分析抑郁及抑郁相关的HLA-DRB1基因对肝硬化的影响。通过计算受试者工作特征曲线(ROC曲线)下面积(AUC)和拟合优度统计值来评价模型效能。运用全部13个变量(年龄、性别、PHQ-9评分、DRB1*03∶01基因,以及血清ALT、AST、ALP、GGT、TBil、IgG、IgM、C3和C4水平)建立分类回归树(CART)模型。采用AUC、敏感度和特异度等指标在模型队列中评价CART模型的效能。 结果 PBC患者合并抑郁的比例(53.9%)明显高于正常对照组(15.3%)(χ2=57.836,P<0.001)。合并抑郁的PBC患者较不合并抑郁的PBC患者对UDCA的治疗应答较差(χ2=7.549,P=0.006),血清ALP(Z=-2.157,P=0.031)、GGT(Z=-2.180,P=0.029)、IgM(Z=-2.000,P=0.046)水平显著升高。合并抑郁的PBC患者较不合并抑郁的PBC患者携带HLA-DRB1*03∶01基因的PBC患者合并肝硬化的风险显著升高(P<0.001)。二元Logistic回归模型分析结果显示,PHQ-9评分(OR=1.148,95%CI:1.050~1.255,P=0.002)、HLA-DRB1*03∶01基因(OR=5.150,95%CI:1.362~19.478,P=0.016)、年龄(OR=1.057,95%CI:1.009~1.106,P=0.018)和血清ALP水平(OR=1.009,95%CI:1.001~1.017,P=0.020)为PBC患者发生肝硬化的独立危险因素。决策树分析显示,PHQ-9评分≥3.5亦为PBC患者肝硬化的危险因素。 结论 抑郁与PBC患者治疗应答不良相关,并且是PBC患者发生肝硬化的独立危险因素。本研究提示了在PBC患者中对抑郁状态进行识别和早期管理的重要临床意义。 Abstract:Objective Depression is common in patients with primary biliary cholangitis (PBC), but the role of depression in disease progression remains unclear. This study aims to investigate the association between depression and treatment response and the impact of depression on liver cirrhosis in PBC patients. Methods A retrospective analysis was performed for the clinical data of 141 patients with PBC who attended the outpatient service of autoimmune liver diseases in General Hospital of Tianjin Medical University from January 2018 to December 2020 and received standard ursodeoxycholic acid (UDCA) monotherapy for 1 year, and 170 healthy controls, matched for age and sex, who underwent physical examination in Physical Examination Center were enrolled as healthy control group. Patient Health Questionnaire-9 (PHQ-9) was used to evaluate depressive state in the patients with PBC and the healthy controls. The independent-samples t test was used for comparison of normally distributed continuous data between two groups, and the Mann-Whitney U test was used for comparison of non-normally distributed continuous data between two groups; the chi-square test was used for comparison of categorical data between two groups. The binary logistic regression model and the decision tree model were used to analyze the influencing factors for liver cirrhosis in patients with PBC, as well as the influence of depression and the HLA-DRB1 gene on liver cirrhosis. The receiver operating characteristic (ROC) curve, the area under the ROC curve (AUC), and goodness of fit were used to evaluate model performance. All 13 variables were used to establish a classification and regression tree (CART) model, i.e., age, sex, PHQ-9 score, the DRB1*03∶01 gene, and the serum levels of alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase (ALP), gamma-glutamyl transpeptidase (GGT), total bilirubin, immunoglobulin G, immunoglobulin M (IgM), C3, and C4. The indications including AUC, sensitivity, and specificity were used to evaluate the performance of CART model in the model cohort. Results Compared with the normal control group, the PBC group had a significantly higher proportion of the patients with depression (53.9% vs 15.3%, χ2=57.836, P<0.001). Compared with the PBC patients without depression, the PBC patients with depression had a significantly poorer response to UDCA treatment (χ2=7.549, P=0.006) and significant increases in the serum levels of ALP (Z=-2.157, P=0.031), GGT (Z=-2.180, P=0.029), and IgM (Z=-2.000, P=0.046). Compared with the PBC patients without depression, the PBC patients carrying the HLA-DRB1*03∶01 allele had a significant increase in the risk of liver cirrhosis (P<0.001). The binary logistic regression model analysis showed that PHQ-9 score (OR=1.148, 95%CI: 1.050 — 1.255, P=0.002), the HLA-DRB1*03∶01 gene (OR=5.150, 95%CI: 1.362 — 19.478, P=0.016), age (OR=1.057, 95%CI: 1.009 — 1.106, P=0.018), and serum ALP level (OR=1.009, 95%CI: 1.001 — 1.017, P=0.020) were independent risk factors for liver cirrhosis in patients with PBC. The decision tree analysis showed that PHQ-9 score ≥3.5 was also a risk factor for liver cirrhosis in PBC patients. Conclusion Depression is associated with poor treatment response in patients with PBC, and it is an independent risk factor for liver cirrhosis in patients with PBC. This study highlights the important clinical significance of the identification and early management of depressive state in patients with PBC. -
Key words:
- Primary Biliary Cholangitis /
- Liver Cirrhosis /
- Depression /
- Risk Factors
-
表 1 PBC患者和对照组合并抑郁状态比较
Table 1. Comparison of depressive symptoms between patients with PBC and controls
抑郁评估 对照组(n=170) PBC组(n=141) 统计值 P值 PHQ-9评分 1.0(0.0~2.0) 5.0(1.0~10.0) Z=-7.413 <0.001 抑郁比例[例(%)] 26(15.3) 76(53.9) χ2=57.836 <0.001 抑郁严重程度[例(%)] χ2=59.490 <0.001 轻度 14(53.8) 35(46.1) 中度 8(30.8) 21(27.6) 中重度 4(15.4) 14(18.4) 重度 0(0.0) 6(7.9) 表 2 抑郁状态对PBC患者UDCA治疗应答的影响
Table 2. Impact of depressive symptoms to the treatment response to UDCA in patients with PBC
指标 合并抑郁(n=76) 不合并抑郁(n=65) 统计值 P值 年龄(岁) 57.6±11.6 56.2±12.7 t=-0.700 0.485 女性[例(%)] 68(89.5) 51(78.5) χ2=3.226 0.072 肝硬化[例(%)] 34(44.7) 7(10.8) χ2=19.602 <0.001 ALT(U/L) 33.0(25.0~41.8) 31.0(17.0~40.0) Z=-1.460 0.144 AST(U/L) 30.5(24.0~41.8) 30.0(22.0~38.0) Z=-0.470 0.639 ALP(U/L) 121.5(82.5~237.3) 95.0(71.5~178.5) Z=-2.157 0.031 GGT(U/L) 65.5(34.3~104.3) 41.0(27.5~87.0) Z=-2.180 0.029 TBil(μmol/L) 14.0(10.6~19.9) 12.1(9.9~16.1) Z=-1.454 0.146 IgG(mg/dL) 1 367.9±221.2 1 310.6±217.4 t=-1.545 0.125 IgM(mg/dL) 292.5(163.6~397.4) 237.9(137.5~312.5) Z=-2.000 0.046 C3(mg/dL) 97.8±19.2 102.0±20.3 t=1.275 0.204 C4(mg/dL) 19.5(15.7~24.6) 21.4(17.4~25.4) Z=-1.688 0.091 UDCA应答[例(%)] 43(56.6) 51(78.5) χ2=7.549 0.006 表 3 PBC患者HLA-DRB1基因型与肝硬化的相关性分析
Table 3. Association between HLA-DRB1 genotyping and cirrhosis in patients with PBC
DRB1 无肝硬化(2n=200) 肝硬化(2n=82) P值 01∶01 9(4.5%) 4(4.9%) >0.05 01∶02 2(1.0%) 1(1.2%) >0.05 03∶01 7(3.5%) 14(17.1%) <0.001 04∶01 2(1.0%) 0(0.0) >0.05 04∶05 7(3.5%) 2(2.4%) >0.05 04∶06 2(1.0%) 0(0.0) >0.05 04∶07 1(0.5%) 0(0.0) >0.05 04∶10 1(0.5%) 0(0.0) >0.05 07∶01 38(19.0%) 11(13.4%) >0.05 08∶01 4(2.0%) 3(3.7%) >0.05 08∶03 16(8.0%) 6(7.3%) >0.05 09∶01 22(11.0%) 8(9.8%) >0.05 10∶01 5(2.5%) 2(2.4%) >0.05 11∶01 11(5.5%) 4(4.9%) >0.05 12∶01 4(2.0%) 2(2.4%) >0.05 12∶02 9(4.5%) 4(4.9%) >0.05 13∶01 5(2.5%) 2(2.4%) >0.05 14∶01 6(3.0%) 2(2.4%) >0.05 14∶05 9(4.5%) 3(3.7%) >0.05 14∶07 2(1.0%) 0(0.0) >0.05 14∶14 1(0.5%) 1(1.2%) >0.05 14∶54 3(1.5%) 1(1.2%) >0.05 15∶01 23(11.5%) 7(8.5%) >0.05 15∶02 7(3.5%) 3(3.7%) >0.05 16∶02 4(2.0%) 2(2.4%) >0.05 表 4 PBC患者发生肝硬化的单因素和多因素分析
Table 4. Univariate and multivariate analysis of risk factors for cirrhosis in patients with PBC
变量 单因素 多因素 OR(95%CI) P值 OR(95%CI) P值 年龄 1.036(1.003~1.071) 0.034 1.057(1.009~1.106) 0.018 性别(男=0,女=1) 1.111(0.402~3.074) 0.839 - - PHQ-9评分(≤4分=0,≥5分=1) 1.146(1.073~1.223) <0.001 1.148(1.050~1.255) 0.002 DRB1*03∶01(其他基因型=0,DRB1*03∶01=1) 6.889(2.526~18.789) <0.001 5.150(1.362~19.478) 0.016 ALT(ALT<40 U/L=0,ALT≥40 U/L=1) 1.022(0.999~1.047) 0.065 - - AST(AST<40 U/L=0,AST≥40 U/L=1) 1.026(0.999~1.053) 0.057 - - ALP(ALP<150 U/L=0,ALP≥150 U/L=1) 1.008(1.004~1.012) <0.001 1.009(1.001~1.017) 0.020 GGT(GGT<40 U/L=0,GGT≥40 U/L=1) 1.012(1.004~1.020) 0.003 0.993(0.980~1.005) 0.258 TBil(TBil<17 μmol/L=0,TBil≥17 μmol/L=1) 1.059(1.011~1.109) 0.015 0.956(0.883~1.035) 0.265 IgG(IgG<1 560 mg/dL=0,IgG≥1 560 mg/dL=1) 1.001(0.999~1.002) 0.470 - - IgM(IgM<304 mg/dL=0,IgM≥304 mg/dL=1) 1.006(1.003~1.009) <0.001 1.004(1.000~1.008) 0.063 C3(C3<79 mg/dL=0,C3≥79 mg/dL=1) 0.981(0.961~1.001) 0.060 - - C4(C4<16 mg/dL=0,C4≥16 mg/dL=1) 0.887(0.823~0.956) 0.002 0.924(0.844~1.012) 0.090 -
[1] CORDELL HJ, FRYETT JJ, UENO K, et al. An international genome-wide meta-analysis of primary biliary cholangitis: Novel risk loci and candidate drugs[J]. J Hepatol, 2021, 75( 3): 572- 581. DOI: 10.1016/j.jhep.2021.04.055. [2] WONG GL, LAW FM, WONG VW, et al. Health-related quality of life in Chinese patients with primary biliary cirrhosis[J]. J Gastroenterol Hepatol, 2008, 23( 4): 592- 598. DOI: 10.1016/j.jhep.2021.04.055. [3] BIAGINI MR, TOZZI A, MILANI S, et al. Fatigue in primary biliary cirrhosis: a possible role of comorbidities[J]. Eur J Gastroenterol Hepatol, 2008, 20( 2): 122- 126. DOI: 10.1097/MEG.0b013e3282f1cbda. [4] SHAHEEN AA, KAPLAN GG, ALMISHRI W, et al. The impact of depression and antidepressant usage on primary biliary cholangitis clinical outcomes[J]. PLoS One, 2018, 13( 4): e0194839. DOI: 10.1371/journal.pone.0194839. [5] BUGANZA-TORIO E, MITCHELL N, ABRALDES JG, et al. Depression in cirrhosis-a prospective evaluation of the prevalence, predictors and development of a screening nomogram[J]. Aliment Pharmacol Ther, 2019, 49( 2): 194- 201. DOI: 10.1111/apt.15068. [6] KRONSTEN VT, TRANAH TH, PARIANTE C, et al. Gut-derived systemic inflammation as a driver of depression in chronic liver disease[J]. J Hepatol, 2022, 76( 3): 665- 680. DOI: 10.1016/j.jhep.2021.11.008. [7] VOS T, BARBER RM, BELL B, et al. Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990—2013: a systematic analysis for the Global Burden of Disease Study 2013[J]. Lancet, 2015, 386( 9995): 743- 800. DOI: 10.1016/S0140-6736(15)60692-4. [8] MULLISH BH, KABIR MS, THURSZ MR, et al. Review article: depression and the use of antidepressants in patients with chronic liver disease or liver transplantation[J]. Aliment Pharmacol Ther, 2014, 40( 8): 880- 892. DOI: 10.1111/apt.12925. [9] HARMS MH, de VEER RC, LAMMERS WJ, et al. Number needed to treat with ursodeoxycholic acid therapy to prevent liver transplantation or death in primary biliary cholangitis[J]. Gut, 2020, 69( 8): 1502- 1509. DOI: 10.1136/gutjnl-2019-319057. [10] HIRSCHFIELD GM, LIU X, XU C, et al. Primary biliary cirrhosis associated with HLA, IL12A, and IL12RB2 variants[J]. N Engl J Med, 2009, 360( 24): 2544- 2555. DOI: 10.1056/NEJMoa0810440. [11] LINDOR KD, BOWLUS CL, BOYER J, et al. Primary biliary cholangitis: 2018 practice guidance from the American Association for the Study of Liver Diseases[J]. Hepatology, 2019, 69( 1): 394- 419. DOI: 10.1002/hep.30145. [12] PARÉS A, CABALLERÍA L, RODÉS J. Excellent long-term survival in patients with primary biliary cirrhosis and biochemical response to ursodeoxycholic acid[J]. Gastroenterology, 2006, 130( 3): 715- 720. DOI: 10.1053/j.gastro.2005.12.029. [13] NEGERI ZF, LEVIS B, SUN Y, et al. Accuracy of the Patient Health Questionnaire-9 for screening to detect major depression: updated systematic review and individual participant data meta-analysis[J]. BMJ, 2021, 375: n2183. DOI: 10.1136/bmj.n2183. [14] GINÈS P, KRAG A, ABRALDES JG, et al. Liver cirrhosis[J]. Lancet, 2021, 398( 10308): 1359- 1376. DOI: 10.1016/S0140-6736(21)01374-X. [15] SCHRAMM C, WAHL I, WEILER-NORMANN C, et al. Health-related quality of life, depression, and anxiety in patients with autoimmune hepatitis[J]. J Hepatol, 2014, 60( 3): 618- 624. DOI: 10.1016/j.jhep.2013.10.035. [16] LIAO J, KANG J, LI F, et al. A cross-sectional study on the association of anxiety and depression with the disease activity of systemic lupus erythematosus[J]. BMC Psychiatry, 2022, 22( 1): 591. DOI: 10.1186/s12888-022-04236-z. [17] MICHELSEN B, KRISTIANSLUND EK, SEXTON J, et al. Do depression and anxiety reduce the likelihood of remission in rheumatoid arthritis and psoriatic arthritis? Data from the prospective multicentre NOR-DMARD study[J]. Ann Rheum Dis, 2017, 76( 11): 1906- 1910. DOI: 10.1136/annrheumdis-2017-211284. [18] KOCHAR B, BARNES EL, LONG MD, et al. Depression is associated with more aggressive inflammatory bowel disease[J]. Am J Gastroenterol, 2018, 113( 1): 80- 85. DOI: 10.1038/ajg.2017.423. [19] ROBERTS AL, KUBZANSKY LD, MALSPEIS S, et al. Association of depression with risk of incident systemic lupus erythematosus in women assessed across 2 decades[J]. JAMA Psychiatry, 2018, 75( 12): 1225- 1233. DOI: 10.1001/jamapsychiatry.2018.2462. [20] ONUORA S. Depression linked to seronegative RA risk[J]. Nat Rev Rheumatol, 2020, 16( 12): 666. DOI: 10.1038/s41584-020-00532-8. [21] ANANTHAKRISHNAN AN. Epidemiology and risk factors for IBD[J]. Nat Rev Gastroenterol Hepatol, 2015, 12( 4): 205- 217. DOI: 10.1038/nrgastro.2015.34. [22] CAUCH-DUDEK K, ABBEY S, STEWART DE, et al. Fatigue in primary biliary cirrhosis[J]. Gut, 1998, 43( 5): 705- 710. DOI: 10.1136/gut.43.5.705. [23] UMEMURA T, JOSHITA S, ICHIJO T, et al. Human leukocyte antigen class II molecules confer both susceptibility and progression in Japanese patients with primary biliary cirrhosis[J]. Hepatology, 2012, 55( 2): 506- 511. DOI: 10.1002/hep.24705. [24] CZAJA AJ. Rapidity of treatment response and outcome in type 1 autoimmune hepatitis[J]. J Hepatol, 2009, 51( 1): 161- 167. DOI: 10.1016/j.jhep.2009.02.026. [25] MURATORI P, GRANITO A, QUARNETI C, et al. Autoimmune hepatitis in Italy: the Bologna experience[J]. J Hepatol, 2009, 50( 6): 1210- 1218. DOI: 10.1016/j.jhep.2009.01.020. [26] KATRINLI S, LORI A, KILARU V, et al. Association of HLA locus alleles with posttraumatic stress disorder[J]. Brain Behav Immun, 2019, 81: 655- 658. DOI: 10.1016/j.bbi.2019.07.016. [27] LE CLERC S, LOMBARDI L, BAUNE BT, et al. HLA-DRB1 and HLA-DQB1 genetic diversity modulates response to lithium in bipolar affective disorders[J]. Sci Rep, 2021, 11( 1): 17823. DOI: 10.1038/s41598-021-97140-7. [28] BANDINELLI F, BENUCCI M, SALAFFI F, et al. Do new and old biomarkers of early undifferentiated arthritis correlate with arthritis impact measurement scales?[J]. Clin Exp Rheumatol, 2021, 39( 1): 79- 83. DOI: 10.55563/clinexprheumatol/nqqx5k. -



 PDF下载 ( 868 KB)
PDF下载 ( 868 KB)

 下载:
下载:



