T淋巴细胞在原发性硬化性胆管炎中的作用
DOI: 10.3969/j.issn.1001-5256.2023.12.026
利益冲突声明:本文不存在任何利益冲突。
作者贡献声明:李子怡、毛小荣、李俊峰对文章的思路和设计有关键贡献,并参与修改文章关键内容;李子怡、张万洁、王富春均参与了文献检索及起草文章初稿。
-
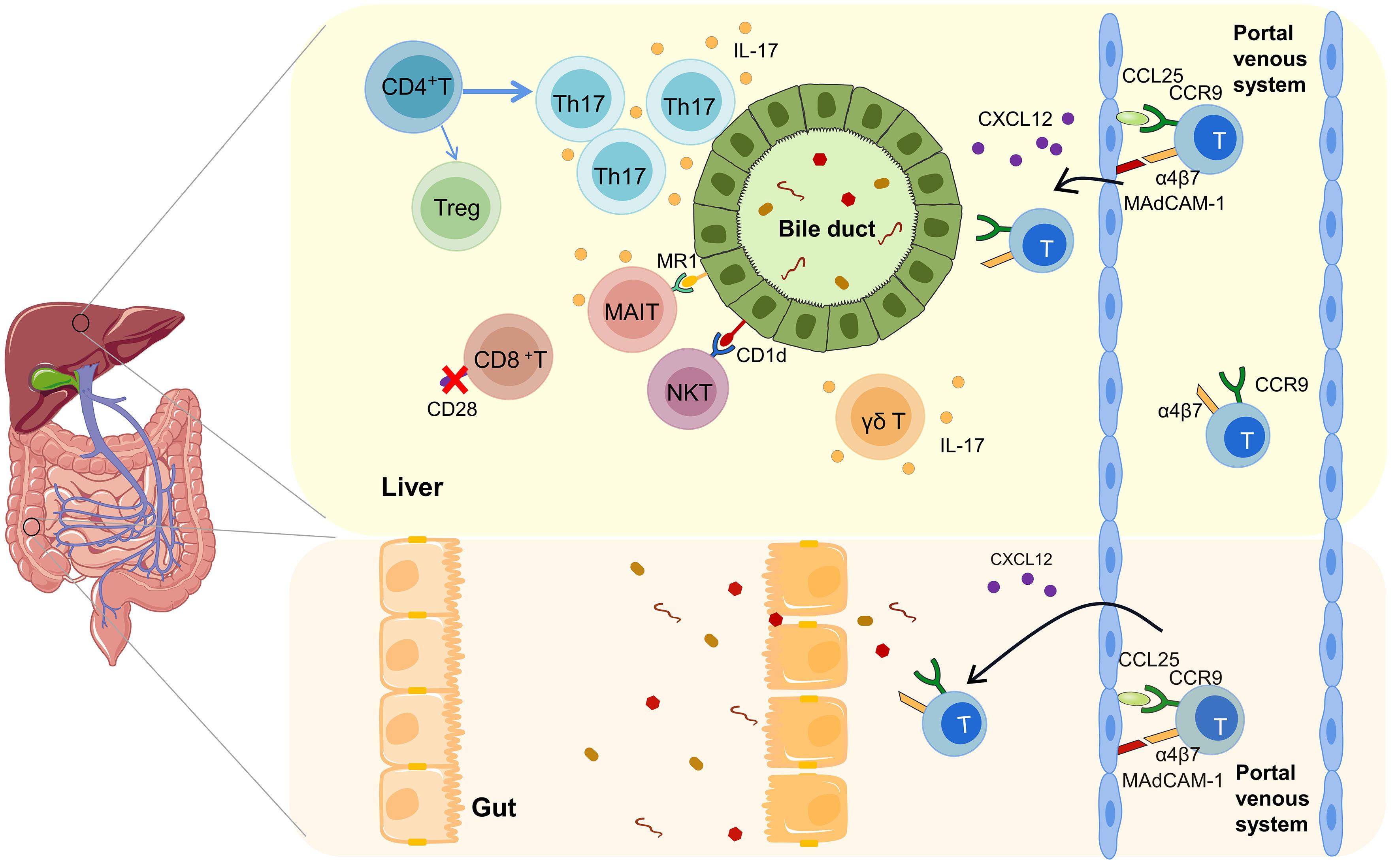
摘要: 原发性硬化性胆管炎(PSC)是一种免疫介导的慢性胆汁淤积性肝病,可进展为肝硬化和肝衰竭等终末期肝病,目前尚无有效的治疗方法。研究发现,T淋巴细胞与PSC的发生发展密切相关。现对T淋巴细胞在PSC中的作用进行综述,以期为PSC发病机制的研究及临床诊疗提供新的思路。Abstract: Primary sclerosing cholangitis (PSC) is an immune-mediated chronic cholestatic liver disease and can progress to end-stage liver diseases such as liver cirrhosis and liver failure, and there are still no effective treatment methods at present. Studies have found that T lymphocytes are closely associated with the development and progression of PSC. This article reviews the role of T lymphocytes in PSC, so as to provide new ideas for research on the pathogenesis of PSC and the clinical diagnosis and treatment of PSC.
-
Key words:
- T lymphocytes /
- Primary Sclerosing Cholangitis /
- Pathogenesis
-
[1] Chinese Society of Hepatology. Guidelines on the diagnosis and management of primary sclerosing cholangitis(2021)[J]. J Clin Hepatol, 2022, 38( 1): 50- 61. DOI: 10.3760/cma. j. cnl12138-20211109-0078.中华医学会肝病学分会. 原发性硬化性胆管炎诊断及治疗指南(2021)[J]. 临床肝胆病杂志, 2022, 38( 1): 50- 61. DOI: 10.3760/ cma. j. cnl12138-20211109-00786. [2] POCH T, KRAUSE J, CASAR C, et al. Single-cell atlas of hepatic T cells reveals expansion of liver-resident naive-like CD4+ T cells in primary sclerosing cholangitis[J]. J Hepatol, 2021, 75( 2): 414- 423. DOI: 10.1016/j.jhep.2021.03.016. [3] CHEN Y, TIAN Z. Innate lymphocytes: pathogenesis and therapeutic targets of liver diseases and cancer[J]. Cell Mol Immunol, 2021, 18( 1): 57- 72. DOI: 10.1038/s41423-020-00561-z. [4] BERINGER A, MIOSSEC P. IL-17 and IL-17-producing cells and liver diseases, with focus on autoimmune liver diseases[J]. Autoimmun Rev, 2018, 17( 12): 1176- 1185. DOI: 10.1016/j.autrev.2018.06.008. [5] KUNZMANN LK, SCHOKNECHT T, POCH T, et al. Monocytes as potential mediators of pathogen-induced T-helper 17 differentiation in patients with primary sclerosing cholangitis(PSC)[J]. Hepatology, 2020, 72( 4): 1310- 1326. DOI: 10.1002/hep.31140. [6] JEFFERY HC, HUNTER S, HUMPHREYS EH, et al. Bidirectional cross-talk between biliary epithelium and Th17 cells promotes local Th17 expansion and bile duct proliferation in biliary liver diseases[J]. J Immunol, 2019, 203( 5): 1151- 1159. DOI: 10.4049/jimmunol.1800455. [7] NAKAMOTO N, SASAKI N, AOKI R, et al. Gut pathobionts underlie intestinal barrier dysfunction and liver T helper 17 cell immune response in primary sclerosing cholangitis[J]. Nat Microbiol, 2019, 4( 3): 492- 503. DOI: 10.1038/s41564-018-0333-1. [8] LONGHI MS, MIELI-VERGANI G, VERGANI D. Regulatory T cells in autoimmune hepatitis: an updated overview[J]. J Autoimmun, 2021, 119: 102619. DOI: 10.1016/j.jaut.2021.102619. [9] TAYLOR AE, CAREY AN, KUDIRA R, et al. Interleukin 2 promotes hepatic regulatory T cell responses and protects from biliary fibrosis in murine sclerosing cholangitis[J]. Hepatology, 2018, 68( 5): 1905- 1921. DOI: 10.1002/hep.30061. [10] SCHWINGE D, VON HAXTHAUSEN F, QUAAS A, et al. Dysfunction of hepatic regulatory T cells in experimental sclerosing cholangitis is related to IL-12 signaling[J]. J Hepatol, 2017, 66( 4): 798- 805. DOI: 10.1016/j.jhep.2016.12.001. [11] HELMIN KA, MORALES-NEBREDA L, TORRES ACOSTA MA, et al. Maintenance DNA methylation is essential for regulatory T cell development and stability of suppressive function[J]. J Clin Invest, 2020, 130( 12): 6571- 6587. DOI: 10.1172/JCI137712. [12] VOSKENS C, STOICA D, ROSENBERG M, et al. Autologous regulatory T-cell transfer in refractory ulcerative colitis with concomitant primary sclerosing cholangitis[J]. Gut, 2023, 72( 1): 49- 53. DOI: 10.1136/gutjnl-2022-327075. [13] JIANG T, ZHANG HW, WEN YP, et al. 5-Aza-2-deoxycytidine alleviates the progression of primary biliary cholangitis by suppressing the FoxP3 methylation and promoting the Treg/Th17 balance[J]. Int Immunopharmacol, 2021, 96: 107820. DOI: 10.1016/j.intimp.2021.107820. [14] ZIMMER CL, VON SETH E, BUGGERT M, et al. A biliary immune landscape map of primary sclerosing cholangitis reveals a dominant network of neutrophils and tissue-resident T cells[J]. Sci Transl Med, 2021, 13( 599): eabb3107. DOI: 10.1126/scitranslmed.abb3107. [15] RAVICHANDRAN G, NEUMANN K, BERKHOUT LK, et al. Interferon-γ-dependent immune responses contribute to the pathogenesis of sclerosing cholangitis in mice[J]. J Hepatol, 2019, 71( 4): 773- 782. DOI: 10.1016/j.jhep.2019.05.023. [16] LUO P, LIU L, HOU W, et al. Gene set enrichment analysis detected immune cell-related pathways associated with primary sclerosing cholangitis[J]. Biomed Res Int, 2022, 2022: 2371347. DOI: 10.1155/2022/2371347. [17] LIASKOU E, JEFFERY LE, TRIVEDI PJ, et al. Loss of CD28 expression by liver-infiltrating T cells contributes to pathogenesis of primary sclerosing cholangitis[J]. Gastroenterology, 2014, 147( 1): 221- 232.e7. DOI: 10.1053/j.gastro.2014.04.003. [18] TEDESCO D, THAPA M, CHIN CY, et al. Alterations in intestinal microbiota lead to production of interleukin 17 by Intrahepatic γδ T-cell receptor-positive cells and pathogenesis of cholestatic liver disease[J]. Gastroenterology, 2018, 154( 8): 2178- 2193. DOI: 10.1053/j.gastro.2018.02.019. [19] VALESTRAND L, BERNTSEN NL, ZHENG F, et al. Lipid antigens in bile from patients with chronic liver diseases activate natural killer T cells[J]. Clin Exp Immunol, 2021, 203( 2): 304- 314. DOI: 10.1111/cei.13541. [20] BERNTSEN NL, FOSBY B, TAN C, et al. Natural killer T cells mediate inflammation in the bile ducts[J]. Mucosal Immunol, 2018, 11( 6): 1582- 1590. DOI: 10.1038/s41385-018-0066-8. [21] VALESTRAND L, ZHENG F, HANSEN SH, et al. Bile from patients with primary sclerosing cholangitis contains mucosal-associated invariant T-cell antigens[J]. Am J Pathol, 2022, 192( 4): 629- 641. DOI: 10.1016/j.ajpath.2021.12.008. [22] VON SETH E, ZIMMER CL, REUTERWALL-HANSSON M, et al. Primary sclerosing cholangitis leads to dysfunction and loss of MAIT cells[J]. Eur J Immunol, 2018, 48( 12): 1997- 2004. DOI: 10.1002/eji.201847608. [23] BÖTTCHER K, ROMBOUTS K, SAFFIOTI F, et al. MAIT cells are chronically activated in patients with autoimmune liver disease and promote profibrogenic hepatic stellate cell activation[J]. Hepatology, 2018, 68( 1): 172- 186. DOI: 10.1002/hep.29782. [24] de KRIJGER M, WILDENBERG ME, de JONGE WJ, et al. Return to sender: Lymphocyte trafficking mechanisms as contributors to primary sclerosing cholangitis[J]. J Hepatol, 2019, 71( 3): 603- 615. DOI: 10.1016/j.jhep.2019.05.006. [25] GRAHAM JJ, MUKHERJEE S, YUKSEL M, et al. Aberrant hepatic trafficking of gut-derived T cells is not specific to primary sclerosing cholangitis[J]. Hepatology, 2022, 75( 3): 518- 530. DOI: 10.1002/hep.32193. [26] CHRISTENSEN B, MICIC D, GIBSON PR, et al. Vedolizumab in patients with concurrent primary sclerosing cholangitis and inflammatory bowel disease does not improve liver biochemistry but is safe and effective for the bowel disease[J]. Aliment Pharmacol Ther, 2018, 47( 6): 753- 762. DOI: 10.1111/apt.14525. [27] RAI RP, LIU Y, IYER SS, et al. Blocking integrin α4β7-mediated CD4 T cell recruitment to the intestine and liver protects mice from western diet-induced non-alcoholic steatohepatitis[J]. J Hepatol, 2020, 73( 5): 1013- 1022. DOI: 10.1016/j.jhep.2020.05.047. -



 PDF下载 ( 803 KB)
PDF下载 ( 803 KB)

 下载:
下载:


