Tribbles同源蛋白1 rs2954029单核苷酸多态性与非酒精性脂肪性肝病及冠状动脉粥样硬化性心脏病的相关性分析
DOI: 10.3969/j.issn.1001-5256.2019.06.030
Association of TRIB1 rs2954029 single nucleotide polymorphism with the risk of nonalcoholic fatty liver disease and coronary heart disease
-
摘要:
目的在青岛地区汉族人群中,探讨Tribbles同源蛋白1(TRIB1) rs2954029单核苷酸多态性与非酒精性脂肪性肝病(NAFLD)及冠状动脉粥样硬化性心脏病(CHD)的相关性,并探讨该基因多态性对血脂水平的影响。方法随机选取2017年1月-2017年11月收入青岛市市立医院的146例NAFLD患者为NAFLD组,155例CHD患者为CHD组,156例NAFLD合并CHD患者为合并组,175例健康对照人群为对照组。采集空腹静脉血进行生化检测和TRIB1 rs2954029基因型测定。应用χ2检验分析样本是否符合Hardy-Weinberg平衡;计数资料组间比较采用χ2检验;计量资料组间比较采用t检验或Wilcoxon秩和检验;比值比(OR)及其95%可信区间(95%CI)应用非条件logistic回归模型计算。结果 CHD组与对照组相比,合并组与NAFLD组相比,rs2954029位点基因型频率及等位基因频率分布差异均无统计学意义(P值均> 0. 05)。CHD组与对照组中,rs2954029 AT+AA携带者与TT携带者相比,发病风险未增加(OR=1. 496,95%CI...
Abstract:Objective To investigate the association of TRIB1 rs2954029 single nucleotide polymorphism with nonalcoholic fatty liver disease (NAFLD) and coronary heart disease (CHD) and its influence on blood lipids in the Chinese Han population in Qingdao, China.Methods A total of 146 patients with NAFLD, 155 patients with CHD, and 156 patients with NAFLD and CHD, who were admitted to Qingdao Municipal Hospital from January to November, 2017, were enrolled as NAFLD group, CHD group, and NAFLD + CHD group, respectively, and 175 healthy individuals were enrolled as control group. Fasting venous blood samples were collected for biochemical analysis and TRIB1 rs2954029 genotyping. The chi-square test was used to analyze whether the distribution of genotypes was in accordance with the Hardy-Weinberg equilibrium. The chi-square test was used for comparison of categorical data between groups; the t-test and the Wilcoxon rank-sum test were used for comparison of continuous data between groups. The unconditioned logistic regression model was used to calculate odds ratio (OR) and its 95% confidence interval (CI) . Results There were no significant differences in rs2954029 genotype/allele frequency and distribution between the CHD group and the control group, as well as between the NAFLD + CHD group and the NAFLD group (all P > 0. 05) . In the CHD group and the control group, rs2954029 AT + AA carriers did not have an increased risk of CHD compared with rs2954029 TT carriers (OR = 1. 496, 95% CI: 0. 947-2. 363, P = 0. 084) ; in the NAFLD + CHD group and the NAFLD group, rs2954029 AT + AA carriers did not have an increased risk of CHD compared with rs2954029 TT carriers (OR = 1. 361, 95% CI: 0. 832-2. 227, P =0. 219) ; there were still no significant differences after adjustment for sex, age, and body mass index. In the whole population and the NAFLD group, there were no significant differences in biochemical parameters between the individuals carrying A allele and those not carrying this allele (P > 0. 05) ; in the CHD group and the NAFLD + CHD group, the individuals carrying A allele had a significantly higher level of triglyceride than those not carrying this allele (Z =-1. 986, -2. 521, all P < 0. 05) ; in the control group, the individuals carrying A allele had a significantly higher level of total cholesterol than those not carrying this allele (Z =-2. 653, P < 0. 05) . Conclusion In the Chinese Han population in Qingdao, TRIB1 rs2954029 single nucleotide polymorphism does not increase the risk of CHD in NAFLD patients.
-
针对肝硬化上消化道出血,无论是传统的外科手术,还是内镜下治疗或经颈静脉肝内门体分流术的微创治疗,治疗时机是非常重要的,因而成功的预测出血风险则显得极为重要[1-3]。因为乙型肝炎肝硬化的肝脏及脾脏在密度及血流灌注上存在特异性表现,因而ΔCT值具有特殊变化规律[4]。笔者拟基于现有数据,寻找乙型肝炎肝硬化患者CT上的特征性表现,以建立肝硬化患者上消化道出血的预测模型,旨在预测出血风险,为治疗方案的制订提供理论依据。
1. 资料与方法
1.1 研究对象
回顾性分析2015年1月—2021年6月天津市第一中心医院101例乙型肝炎肝硬化患者的临床资料。年龄26~70周岁,平均年龄50岁,其中男85例,女16例。所有患者后期均接受肝脏移植手术,病理证实符合我国慢性乙型肝炎肝硬化诊断标准[5]。纳入标准:年龄为18~70周岁乙型肝炎肝硬化患者。排除标准:合并其他类型肝病;长期大量饮酒史;合并肝癌;肝内存在动静脉瘘;干细胞治疗史;脾切除史;肝部分切除史;门奇静脉曲张断流手术史;门腔静脉分流手术史。根据是否出血,分为出血组(n=58)和非出血组(n=43)。
1.2 观察指标
出血组记录出血即刻的血清TBil、血清肌酐(Cr)、凝血时间、国际标准化比值(INR)、WBC、PLT等指标。并记录同时期强化CT检查测得的肝脏及脾脏平扫期(Plain)、动脉期(A)、门脉期(P)以及静脉期(V)的CT值,并计算各期间CT值的变化(ΔCT),包括Δ动脉期-平扫期(ΔA-Plain)、Δ门脉期-平扫期(ΔP-Plain)、Δ静脉期-平扫期(ΔV-Plain)、Δ门脉期-动脉期(ΔP-A)、Δ静脉期-动脉期(ΔV-A)以及Δ静脉期-门脉期(ΔV-P)。非出血组记录肝移植评估时的实验室及影像学数据。
1.3 CT检查方法
所有患者均使用SOMATOM Definition Flash (Siemens Healthineers Co., Ltd,Germany) 128层螺旋CT进行扫描。参数设置为:管球电压120 kV,自动管电流200-420 mAs,扫描层厚5 mm,重建层厚1.2 mm,螺距0.8。使用双筒高压注射器自右肘正中静脉位置团注非离子型碘对比剂碘海醇注射液80 mL(350 mgI/mL)与生理盐水40 mL,团注速率为3.5 mL/s。动脉期采用团注智能跟踪启动扫描,对比剂团注后45~55 s进行门脉期扫描,70~75 s后行静脉期扫描。扫描后图像数据传输至工作站进行后处理。由同一名医生对于每例观察对象,于各个期像分别选取同一层面做为观察点进行CT值测量。为减少误差,观察点的选取需避开血管、胆管及韧带,同时为了减少观察点的差异,选取了该层面全部肝、脾组织的数据。
1.4 统计学方法
使用IBM SPSS Statistics 26.0进行统计分析。符合正态分布的计量资料以x±s表示,两组间的比较使用t检验;偏态分布的计量资料以M(P25~P75)表示,两组间的比较使用Mann-Whitney U检验。使用logistic回归分析方法,预测相关危险因素。通过计算受试者工作特征曲线下的面积(AUC,或c-统计量)来评估模型辨别力,而模型校准则通过Hosmer-Lemeshow确定。在多变量logistic回归分析结果的基础上,使用Rstudio4.1.2软件的R包(rms)构建预测的列线图模型,并绘制相应的ROC曲线、校准曲线以及临床决策曲线。用Bootstrap重抽样(1000次迭代)进行内部验证。临床决策曲线可以描述列线图给出的净效益,有助于确定在该模型上做出临床决策是否利大于弊。P<0.05为差异有统计学意义。
2. 结果
2.1 出血组与无出血组实验室指标比较
无出血组血清TBil、WBC、PLT与出血组比较,差异均有统计学意义(P值均<0.05);而两组Cr、INR比较,差异均无统计学意义(P值均>0.05)(表 1)。
表 1 合并上消化道出血与无上消化道出血两组间各项实验室检查指标Table 1. The difference of laboratory examination between the two groups with and without upper gastrointestinal bleeding项目 非出血组(n=43) 出血组(n=58) Z值 P值 TBil(μmol/L) 43.20(21.93~109.54) 27.74(18.88~56.63) -2.284 0.022 Cr(μmol/L) 62.00(52.00~86.00) 64.96(58.57~75.25) -0.289 0.773 INR 1.54(1.36~2.06) 1.49(1.28~1.78) -1.202 0.229 WBC(×109/L) 3.93(2.68~5.57) 2.80(1.95~3.60) -3.197 0.001 PLT(×109/L) 70.00(51.00~92.00) 52.50(37.00~77.00) -2.865 0.004 2.2 出血组与非出血组间肝脏及脾脏ΔCT的变化
两组在肝-Plain、脾-P-Plain、脾-P-A ΔCT值存在统计学差异(P值均<0.05),而肝脏在其他期像的CT值和各期间的ΔCT以及脾脏所有期像与ΔCT均无统计学差异(P值均>0.05)(表 2)。
表 2 出血组与非出血组间肝脏及脾脏ΔCT的变化情况Table 2. The difference of ΔCT of liver and spleen between the two groups with and without upper gastrointestinal bleeding项目 非出血组(n=43) 出血组(n=58) 统计值 P值 肝-Plain 51.70±6.77 56.28±5.13 t=-3.870 <0.001 肝-A 62.28±6.84 64.88±6.34 t=-1.971 0.052 肝-P 85.67±13.83 86.48±11.23 t=-0.324 0.747 肝-V 92.88±12.19 94.19±12.19 t=-0.532 0.596 肝-A-Plain 9.00(8.00~15.00) 9.00(4.00~12.00) Z=-1.848 0.065 肝-P-Plain 30.00(27.00~41.00) 30.00(21.75~37.25) Z=-1.395 0.163 肝-V-Plain 41.19±11.37 37.91±10.64 t=1.484 0.141 肝-P-A 20.00(16.00~30.00) 21.00(15.00~27.00) Z=-0.426 0.670 肝-V-A 30.60±12.20 29.31±10.82 t=0.563 0.575 肝-V-P 7.21±8.23 7.71±9.22 t=-0.281 0.780 脾-Plain 45.00±3.16 46.02±3.05 t=-1.634 0.105 脾-A 81.00(71.00~90.00) 82.50(73.00~93.25) Z=-0.722 0.470 脾-P 116.81±23.81 110.07±16.44 t=1.684 0.095 脾-V 105.81±17.73 103.16±14.02 t=0.836 0.405 脾-A-Plain 37.09±14.02 38.00±14.01 t=-0.322 0.748 脾-P-Plain 71.81±22.58 64.05±16.43 t=2.012 0.047 脾-V-Plain 60.81±16.80 57.14±14.15 t=1.192 0.236 脾-P-A 34.72±16.39 26.05±17.00 t=2.577 0.011 脾-V-A 23.72±13.16 19.14±17.50 t=1.441 0.153 脾-V-P -9.00(-17.00~-4.00) -8.00(-11.00~-2.75) Z=-1.836 0.066 2.3 乙型肝炎肝硬化上消化道出血的危险因素分析及预测模型的建立
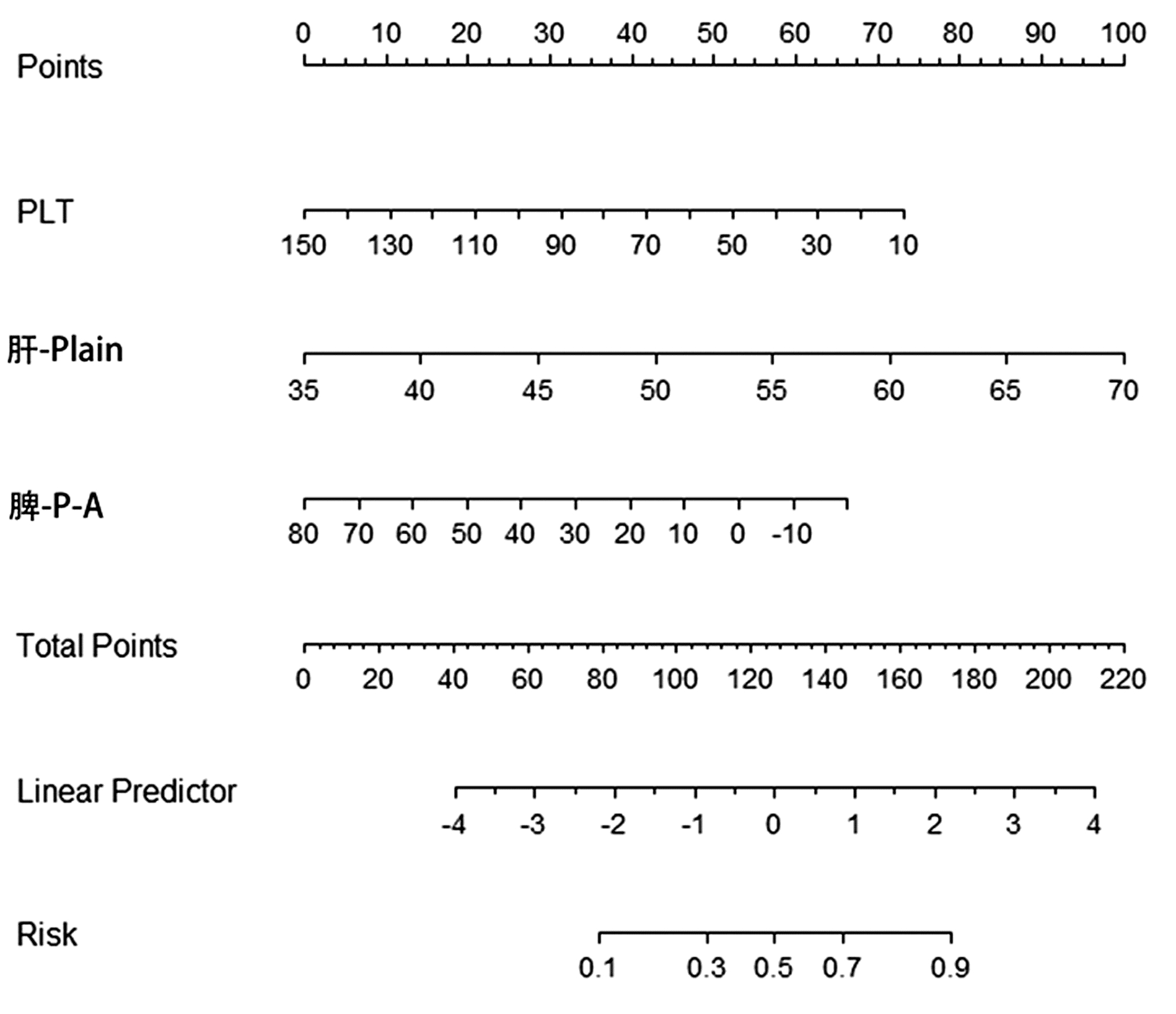
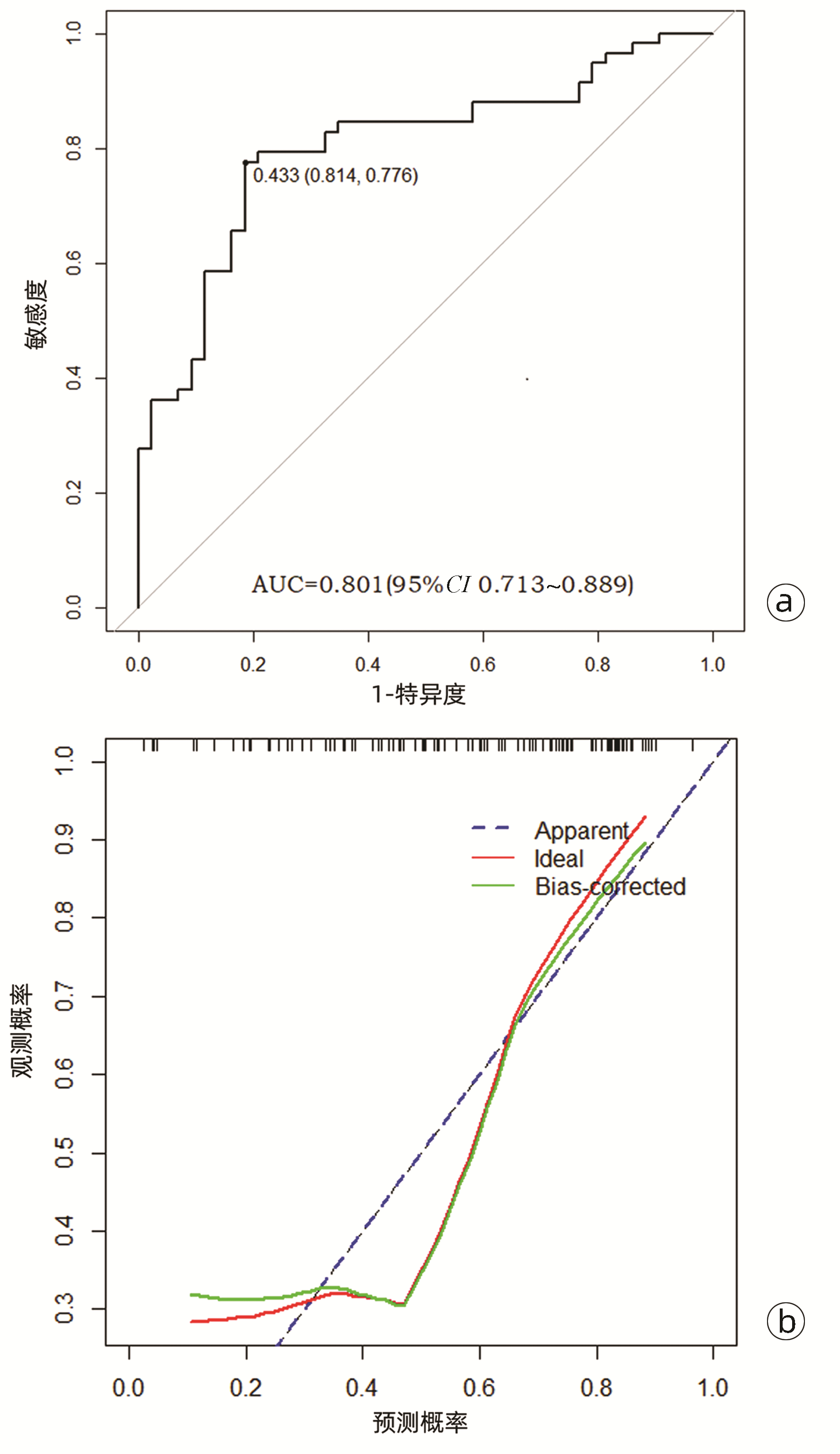
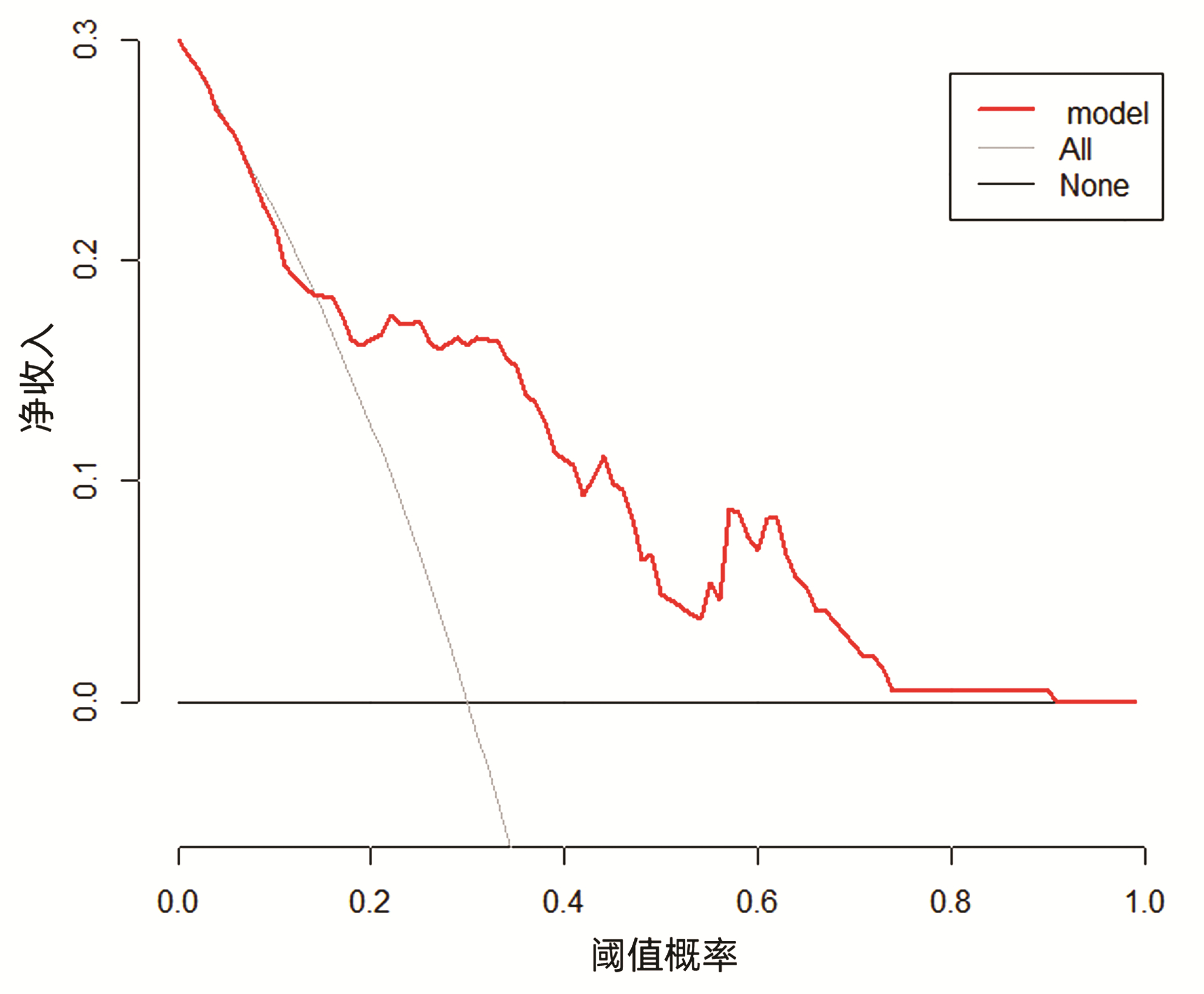
综合上述指标,发现乙型肝炎肝硬化患者WBC、PLT、肝-Plain、脾-P-Plain、脾-P-A ΔCT值在两组间具有统计学差异(P值均<0.05);应用多因素logistic分析结果显示PLT、肝-Plain、脾-P-A间ΔCT值为上消化道出血的独立危险因素(P值均<0.05) (表 3)。基于PLT、肝-Plain、脾-P-A间ΔCT值这3个危险因素,使用Rstudio4.1.2软件的R包(rms)构建预测乙型肝炎肝硬化上消化道出血的预测模型。该模型的AUC为0.801(95%CI:0.713~0.889),敏感度是0.814,特异度是0.776。模型的校准曲线贴近理想曲线(斜率为1.007)。临床决策曲线显示构建的列线图做出的临床决策给患者带来的净收益高于所有患者的净收益,表明患者可以从模型中获益(图 1~3)。
表 3 上消化道出血单因素和多因素的logistic分析结果Table 3. Univariate and multivariate logistic analysis of upper gastrointestinal bleeding参数 单因素分析 多因素分析 OR值 95%CI P值 OR值 95%CI P值 WBC 0.770 0.624~0.952 0.016 PLT 0.979 0.965~0.994 0.006 0.968 0.944~0.993 0.011 肝-Plain 1.142 1.058~1.233 0.001 1.148 1.047~1.259 0.003 脾-V-Plain 0.979 0.959~1.000 0.050 脾-P-A 0.968 0.944~0.994 0.015 0.951 0.908~0.995 0.030 3. 讨论
上消化道出血是肝硬化最常见的并发症之一,也是导致患者死亡的主要原因之一[6]。准确的判断出血风险是制订临床决策的关键因素。首选的检查方法是内镜检查,既可以直观的看到食道胃底静脉的曲张程度,也能发现胃黏膜病变以及溃疡的存在。但是内镜检查同样也有一定的局限性,比如患者拒绝有创检查;危重患者不具备检查条件等。因此需要更加简便且准确的方法。CT及核磁检查可以反映静脉曲张、脾肾分流等情况,可以对上消化道出血的风险预测起到帮助,但也存在影像学表现与临床不符以及无法量化的缺点。Yan等[7]研究发现肝脾体积比是门静脉高压以及上消化道出血的危险因素,并以此为基础构建了预测模型,该模型的敏感度为92.9%,特异度为87.0%。Yang等[8]研究发现肝脾体积比、脾脏扩大比以及AST是上消化道出血的危险因素,同样也建立了预测模型,该模型的敏感度为94.6%,特异度为84.9%。这些研究均对于肝硬化患者的上消化道出血风险进行了很好的预测。
本研究发现,上消化道出血的发生与PLT减少存在密切联系,而肝硬化时PLT下降的主要原因是由于脾大、脾功能亢进。而脾脏增大也是门静脉高压的直接表现,进一步证实了上消化道出血与门静脉高压之间的关系。
通过灌注CT的研究[9]证实,肝硬化时肝脏在动脉期及门静脉期呈现了高血流灌注的状态。本研究发现两组间脾脏动脉期CT值相近,而出血组脾脏的门脉期CT值则更低,造成脾脏门脉期-动脉期的ΔCT的统计学差异。这说明肝硬化合并上消化道出血患者的脾脏处于血流高循环的状态,增加了门静脉高压,进而更具有出血倾向。
本结果显示合并上消化道出血患者血清总胆红素、白细胞、血小板、肝脏平扫期、脾脏门脉期-平扫期、脾脏门脉期-动脉期间ΔCT值存在统计学差异。应用多因素logistic分析结果显示血小板、肝脏平扫期、脾脏门脉期-动脉期间ΔCT值为上消化道出血的独立危险因素。基于上述结果,使用Rstudio4.1.2软件的R包(rms)构建乙型肝炎肝硬化上消化道出血的预测模型。此模型预测上消化道出血的敏感度是81.4%,特异度是77.6%,对于上消化道出血具有良好的预测能力。将这一研究结果应用于临床,将有效的预测上消化道出血风险,为外科手术、内镜及经颈静脉肝内门体分流术等治疗的临床决策提供依据。
-
[1]KOCH LK, YEH MM.Nonalcoholic fatty liver disease (NAFLD) :Diagnosis, pitfalls, and staging[J].Ann Diagn Pathol, 2018, 37 (1) :83-90. [2]WONG VW.Current prevention and treatment options for NAFLD[J].Adv Exp Med Biol, 2018, 1061 (1) :149-157. [3]DAY CP, JAMES OF.Steatohepatitis:A tale of two"hits"?[J].Gastroenterology, 1998, 114 (4) :842-845. [4]TILG H, MOSCHEN AR.Evolution of inflammation in nonalcoholic fatty liver disease:The multiple parallel hits hypothesis[J].Hepatology, 2010, 52 (5) :1836-1846. [5]LUCAS C, LUCAS G, LUCAS N, et al.A systematic review of the present and future of non-alcoholic fatty liver disease[J].Clin Exp Hepatol, 2018, 4 (3) :165-174. [6]FRIEDMAN SL, NEUSCHWANDER-TETRI BA, RINELLA M, et al.Mechanisms of NAFLD development and therapeutic strategies[J].Nat Med, 2018, 24 (7) :908-922. [7]LI XL, SUI JQ, LU LL, et al.Gene polymorphisms associated with non-alcoholic fatty liver disease and coronary artery disease:A concise review[J].Lipids Health Dis, 2016, 15 (1) :53. [8]HSU SH, JANG MH, TORNG PL, et al.Positive association between small dense low-density lipoprotein cholesterol concentration and biomarkers of inflammation, thrombosis, and prediabetes in non-diabetic adults[J].J Atheroscler Thromb, 2018, 275 (1) :87-95. [9]ZHOU H, LIU JC, LUO GH, et al.Application of coronary artery disease-reporting and data system in risk factor analysis of patients with coronary artery disease[J].J Jilin Univ:Med Edit, 2017, 43 (3) :617-621. (in Chinese) 周宏, 刘进才, 罗光华, 等.冠状动脉疾病报告与数据系统在冠心病患者危险因素分析中的应用[J].吉林大学学报:医学版, 2017, 43 (3) :617-621. [10]KATSIKI N, AL-RASADI K, MIKHAILIDIS DP.Lipoprotein (a) and cardiovascular risk:The show must go on[J].Curr Med Chem, 2017, 24 (10) :989-1006. [11]MANTOVANI A, BALLESTRI S, LONARDO A, et al.Cardiovascular disease and myocardial abnormalities in nonalcoholic fatty liver disease[J].Dig Dis Sci, 2016, 61 (5) :1246-1267. [12]ESLAM M, VALENTI L, ROMEO S.Genetics and epigenetics of NAFLD and NASH:Clinical impact[J].J Hepatol, 2018, 68 (2) :268-279. [13]SOOKOIAN S, PIROLA CJ.Genetic predisposition in nonalcoholic fatty liver disease[J].Clin Mol Hepatol, 2017, 23 (1) :1-12. [14]BURKHARDT R, TOH SA, LAGOR WR, et al.Trib1 is a lipidand myocardial infarction-associated gene that regulates hepatic lipogenesis and VLDL production in mice[J].J Clin Invest, 2010, 120 (12) :4410-4414. [15]MAKISHIMA S, BOONVISUT S, ISHIZUKA Y, et al.Sin3A-associated protein, 18 kDa, a novel binding partner of TRIB1, regulates MTTP expression[J].J Lipid Res, 2015, 56 (6) :1145-1152. [16]IWAMOTO S, BOONVISUT S, MAKISHIMA S, et al.The role of TRIB1 in lipid metabolism;from genetics to pathways[J].Biochem Soc Trans, 2015, 43 (5) :1063-1068. [17]OLLILA HM, UTGE S, KRONHOLM E, et al.TRIB1 constitutes a molecular link between regulation of sleep and lipid metabolism in humans[J].Transl Psychiatry, 2012, 2 (1) :97. [18]AUNG LH, YIN RX, WU DF, et al.Association of the TRIB1tribbles homolog 1 gene rs17321515 A>G polymorphism and serum lipid levels in the Mulao and Han populations[J].Lipids Health Dis, 2011, 10 (1) :230. [19]DOUVRIS A, SOUBEYRAND S, NAING T, et al.Functional analysis of the TRIB1 associated locus linked to plasma triglycerides and coronary artery disease[J].J Am Heart Assoc, 2014, 3 (3) :884. [20]WANG L, JING J, FU Q, et al.Association study of genetic variants at newly identified lipid gene TRIB1 with coronary heart disease in Chinese Han population[J].Lipids Health Dis, 2015, 14 (1) :46. [21]KITAMOTO A, KITAMOTO T, NAKAMURA T, et al.Association of polymorphisms in GCKR and TRIB1 with nonalcoholic fatty liver disease and metabolic syndrome traits[J].Endocr J, 2014, 61 (7) :683-689. [22]SPELIOTES EK, YERGES-ARMSTRONG LM, WU J, et al.Genome-wide association analysis identifies variants associated with nonalcoholic fatty liver disease that have distinct effects on metabolic traits[J].PLo S Genet, 2011, 7 (3) :1324. [23]National Workshop on Fatty Liver and Alcoholic Liver Disease, Chinese Society of Hepatology, Chinese Medical Association;Fatty Liver Expert Committee, Chinese Medical Doctor Association.Guidelines of prevention and treatment for nonalcoholic fatty liver disease:A 2018 update[J].J Clin Hepatol, 2018, 34 (5) :947-957. (in Chinese) 中华医学会肝病学分会脂肪肝和酒精性肝病学组, 中国医师协会脂肪性肝病专家委员会.非酒精性脂肪性肝病防治指南 (2018年更新版) [J].临床肝胆病杂志, 2018, 34 (5) :947-957. [24]BALLESTRI S, LONARDO A, BONAPACE S, et al.Risk of cardiovascular, cardiac and arrhythmic complications in patients with non-alcoholic fatty liver disease[J].World J Gastroenterol, 2014, 20 (7) :1724-1745. [25]MO XQ, HUANG C, SONG QQ, et al.Correlation study of cystatin C and insulin resistance in type 2 diabetes mellitus patients with non-alcoholic fatty liver[J].China Med Herald, 2017, 14 (24) :78-80. (in Chinese) 莫小庆, 黄春, 宋青青, 等.2型糖尿病伴非酒精性脂肪肝患者胱抑素C与胰岛素抵抗的相关性研究[J].中国医药导报, 2017, 14 (24) :78-80. [26]PARK MH, KIM N, LEE JY, et al.Genetic loci associated with lipid concentrations and cardiovascular risk factors in the Korean population[J].J Med Genet, 2011, 48 (1) :10-15. [27]VARBO A, BENN M, TYBJAERG-HANSEN A, et al.TRIB1and GCKR polymorphisms, lipid levels, and risk of ischemic heart disease in the general population[J].Arterioscler Thromb Vasc Biol, 2011, 31 (2) :451-457. [28]SUMEGI K, JAROMI L, MAGYARI L, et al.Functional variants of lipid level modifier MLXIPL, GCKR, GALNT2, CILP2, AN-GPTL3 and TRIB1 genes in healthy Roma and Hungarian populations[J].Pathol Oncol Res, 2015, 21 (3) :743-749. [29]JAROMI L, CSONGEI V, POLGAR N, et al.Triglyceride level-influencing functional variants of the ANGPTL3, CILP2, and TRIB1 loci in ischemic stroke[J].Neuromolecular Med, 2011, 13 (3) :179-186. [30]GARCIA-RIOS A, PEREZ-MARTINEZ P, MATA P, et al.Polymorphism at the TRIB1 gene modulates plasma lipid levels:Insight from the Spanish familial hypercholesterolemia cohort study[J].Nutr Metab Cardiovasc Dis, 2011, 21 (12) :957-963. 期刊类型引用(8)
1. 王娜. 肝功能检验对肝炎后肝硬化疾病诊断及评估. 中国城乡企业卫生. 2023(05): 81-83 .  百度学术
百度学术2. 王华丽,颜文娟,严继诗. 细节干预对肝硬化合并上消化道出血患者的效果及生存质量的影响. 中外医疗. 2023(08): 170-173+198 .  百度学术
百度学术3. 张莉,吴凤丽. 裸花紫珠胶囊结合生长抑素治疗老年肝硬化上消化道出血患者的临床效果. 河南医学研究. 2023(16): 3023-3026 .  百度学术
百度学术4. 赵润玲. 乙型肝炎肝硬化上消化道出血患者血清TPO、YKL-40水平变化及对DIC发生风险的指导意义. 国际检验医学杂志. 2023(20): 2521-2525 .  百度学术
百度学术5. 孔敏,靳欣,赵琴. 微信群为基础的短视频远程干预联合IMB模型在肝硬化患者居家护理效果指导. 国际医药卫生导报. 2023(18): 2649-2654 .  百度学术
百度学术6. 阚丽娜,杨昊龙,刘微,孙小媚,贾春霞,丁秀婷. 不同剂量泮托拉唑联合奥曲肽治疗肝硬化上消化道出血患者止血效果比较及其对血清一氧化氮、皮质醇影响. 临床军医杂志. 2023(12): 1317-1320 .  百度学术
百度学术7. 丁敏,刘庭玮,张立波,陈鸿鑫,祁兴顺. 常见肝良性疾病造成胆囊形态改变机制、影像学评估及意义. 临床军医杂志. 2022(09): 887-889+893 .  百度学术
百度学术8. 孙海霞. 探讨多层螺旋CT增强扫描用于肝血管瘤与肝转移癌鉴别诊断的临床价值. 影像研究与医学应用. 2022(24): 70-72 .  百度学术
百度学术其他类型引用(4)
-




 PDF下载 ( 284 KB)
PDF下载 ( 284 KB)

 下载:
下载:



 百度学术
百度学术
 下载:
下载: