| [1] |
LI Q, GUAN X, WU P, et al. Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia[J]. N Engl J Med, 2020, 382( 13): 1199- 1207. DOI: 10.1056/NEJMoa2001316. |
| [2] |
GARG S, KIM L, WHITAKER M, et al. Hospitalization rates and characteristics of patients hospitalized with laboratory-confirmed coronavirus disease 2019- COVID-NET, 14 States, March 1-30, 2020[J]. MMWR Morb Mortal Wkly Rep, 2020, 69( 15): 458- 464. DOI: 10.15585/mmwr.mm6915e3. |
| [3] |
VERITY R, OKELL LC, DORIGATTI I, et al. Estimates of the severity of coronavirus disease 2019: a model-based analysis[J]. Lancet Infect Dis, 2020, 20( 6): 669- 677. DOI: 10.1016/S1473-3099(20)30243-7. |
| [4] |
YANG X, YU Y, XU J, et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study[J]. Lancet Respir Med, 2020, 8( 5): 475- 481. DOI: 10.1016/S2213-2600(20)30079-5. |
| [5] |
|
| [6] |
LI G, de CLERCQ E. Therapeutic options for the 2019 novel coronavirus(2019-nCoV)[J]. Nat Rev Drug Discov, 2020, 19( 3): 149- 150. DOI: 10.1038/d41573-020-00016-0. |
| [7] |
CHEN YC, HUANG WX. Expert consensus on the application of azvudine in the treatment of SARS-CoV-2 infection[J]. China Pharm, 2023, 32( 3): 1- 6. DOI: 10.3969/j.issn.1006-4931.2023.03.001. |
| [8] |
BREVINI T, MAES M, WEBB GJ, et al. FXR inhibition may protect from SARS-CoV-2 infection by reducing ACE2[J]. Nature, 2023, 615( 7950): 134- 142. DOI: 10.1038/s41586-022-05594-0. |
| [9] |
YE H, DENG SW. Clinical evaluation of taurine ursodeoxycholic acid in the treatment of chronic hepatitis B with overlapping hepatitis E infection[J]. Chin Foreign Med Res, 2019, 17( 8): 47- 48. DOI: 10.14033/j.cnki.cfmr.2019.08.022. |
| [10] |
van den BOSSCHE L, HINDRYCKX P, DEVISSCHER L, et al. Ursodeoxycholic acid and its taurine- or glycine-conjugated species reduce colitogenic dysbiosis and equally suppress experimental colitis in mice[J]. Appl Environ Microbiol, 2017, 83( 7): e02766-16. DOI: 10.1128/AEM.02766-16. |
| [11] |
SUN XL, HU X, ZHANG YT. Clinical application of ursodeoxycholic acid[J]. Chin J Pharmacov, 2022, 19( 10): 1149- 1153. DOI: 10.19803/j.1672-8629.20210604. |
| [12] |
LI YG, ZHU ZH. Efficacy of tenofovir combined with ursodeoxycholic acid in outpatient treatment of chronic hepatitis B[J]. Health Med Res Prac, 2022, 19( 4): 22- 26, 37. DOI: 10.11986/j.issn.1673-873X.2022.04.006. |
| [13] |
REIFFEL JA. Propensity score matching: The‘devil is in the details’ where more may be hidden than you know[J]. Am J Med, 2020, 133( 2): 178- 181. DOI: 10.1016/j.amjmed.2019.08.055. |
| [14] |
Chinese Society of Hepatology and Chinese Society of Infectious Diseases, Chinese Medical Association. Guidelines for the prevention and treatment of chronic hepatitis B(version 2022)[J]. Infect Dis Info, 2023, 36( 1): 1- 17. DOI: 10.3969/j.issn.1007-8134.2023.01.01. |
| [15] |
National Health Commission of the People’s Republic of China. Diagnosis and Treatment of COVID-19(Trial Version 10)[J]. Chin J Rational Drug Use, 2023, 20( 1): 1- 11. DOI: 10.3969/j.issn.2096-3327.2023.01.001. |
| [16] |
ZAMPINO R, MELE F, FLORIO LL, et al. Liver injury in remdesivir-treated COVID-19 patients[J]. Hepatol Int, 2020, 14( 5): 881- 883. DOI: 10.1007/s12072-020-10077-3. |
| [17] |
WANG Q, GUO Y, IKETANI S, et al. Antibody evasion by SARS-CoV-2 Omicron subvariants BA.2.12.1, BA.4 and BA.5[J]. Nature, 2022, 608( 7923): 603- 608. DOI: 10.1038/s41586-022-05053-w. |
| [18] |
|
| [19] |
GAZIANO L, GIAMBARTOLOMEI C, PEREIRA AC, et al. Actionable druggable genome-wide Mendelian randomization identifies repurposing opportunities for COVID-19[J]. Nat Med, 2021, 27( 4): 668- 676. DOI: 10.1038/s41591-021-01310-z. |
| [20] |
FUCHS CD, TRAUNER M. Role of bile acids and their receptors in gastrointestinal and hepatic pathophysiology[J]. Nat Rev Gastroenterol Hepatol, 2022, 19( 7): 432- 450. DOI: 10.1038/s41575-021-00566-7. |
| [21] |
WANG CH, YAO XW, WANG R, et al. The latest research progress of novel coronavirus“Omicron sub-variant BA.5”[J]. J Hanan Med Coll, 2022, 28( 20): 1521- 1525. DOI: 10.13210/j.cnki.jhmu.20220909.002. |
| [22] |
LEVIN EG, LUSTIG Y, COHEN C, et al. Waning immune humoral response to BNT162b2 Covid-19 vaccine over 6 months[J]. N Engl J Med, 2021, 385( 24): e84. DOI: 10.1056/NEJMoa2114583. |
| [23] |
BERGWERK M, GONEN T, LUSTIG Y, et al. Covid-19 breakthrough infections in vaccinated health care workers[J]. N Engl J Med, 2021, 385( 16): 1474- 1484. DOI: 10.1056/NEJMoa2109072. |
| [24] |
|
| [25] |
CHEN C. Zhang Xuqing: Can patients with liver disease be vaccinated with COVID-19 vaccine?[J]. Liver Doctor, 2021, 3: 8- 9.
陈词. 张绪清: 肝病患者能否接种新冠肺炎疫苗?[J]. 肝博士, 2021, 3: 8- 9.
|
| [26] |
|
| [27] |
BEUERS U, TRAUNER M, JANSEN P, et al. New paradigms in the treatment of hepatic cholestasis: From UDCA to FXR, PXR and beyond[J]. J Hepatol, 2015, 62( 1 Suppl): S25- S37. DOI: 10.1016/j.jhep.2015.02.023. |
| [28] |
LIU Y, FENG Y, REN J, et al. The influencing factors of abnormal liver function in patients with COVID-19 and its dynamic changes in different drug treatments[J]. J Capit Med Univ, 2022, 43( 3): 433- 439. DOI: 10.3969/j.issn.1006-7795.2022.03.017. |
| [29] |
DAVIES MA, MORDEN E, ROSSEAU P, et al. Outcomes of laboratory-confirmed SARS-CoV-2 infection during resurgence driven by Omicron lineages BA.4 and BA.5 compared with previous waves in the Western Cape Province, South Africa[J]. Int J Infect Dis, 2023, 127: 63- 68. DOI: 10.1016/j.ijid.2022.11.024. |
| [30] |
LEI SY, PENG H, LUO XH. Pathogenic mechanism of liver injury caused by coronavirus disease 2019 and protective strategies for patients with viral hepatitis cirrhosis[J]. J Clin Hepatol, 2020, 36( 7): 1619- 1622. DOI: 10.3969/j.issn.1001-5256.2020.07.037. |



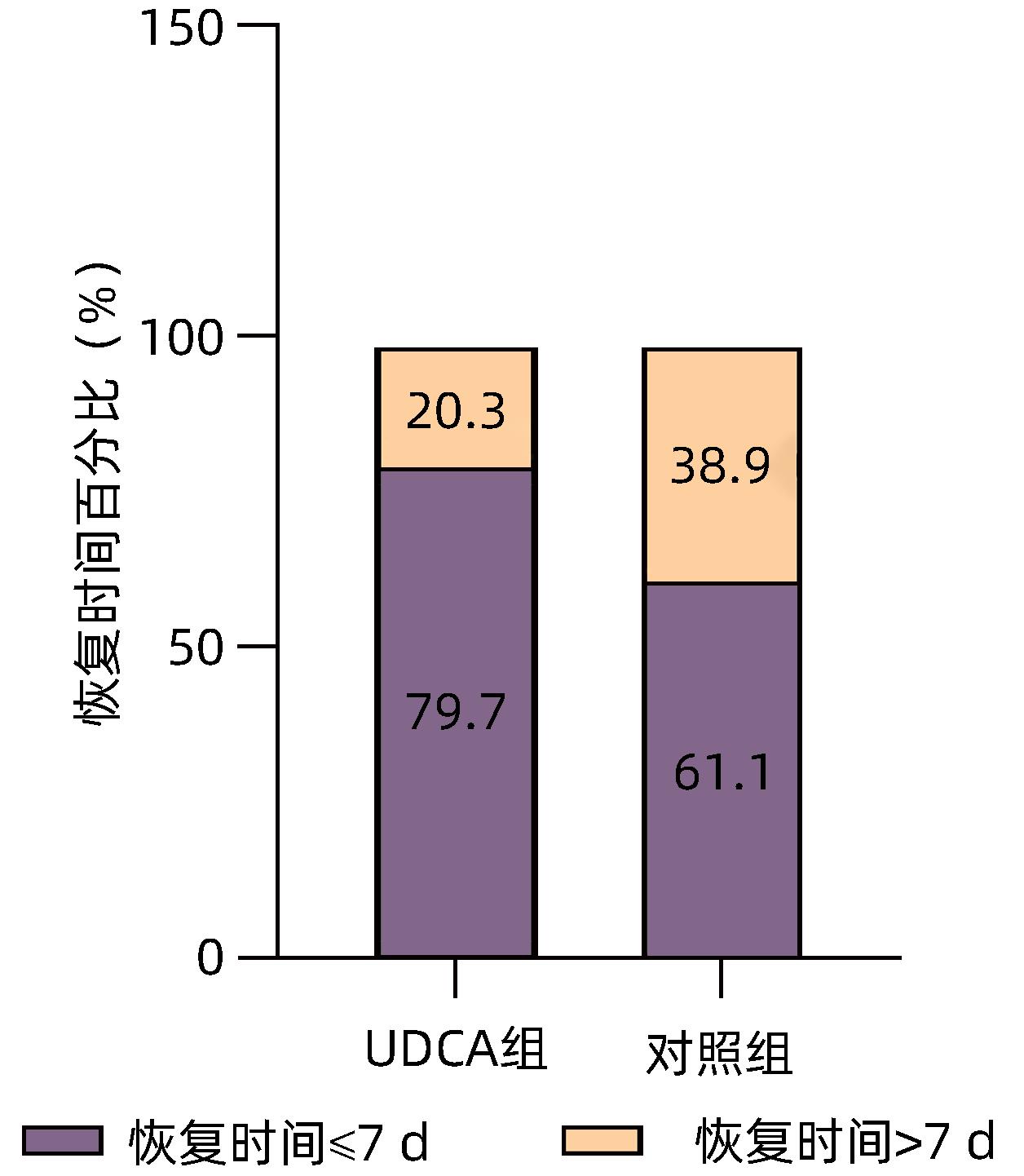




 DownLoad:
DownLoad: