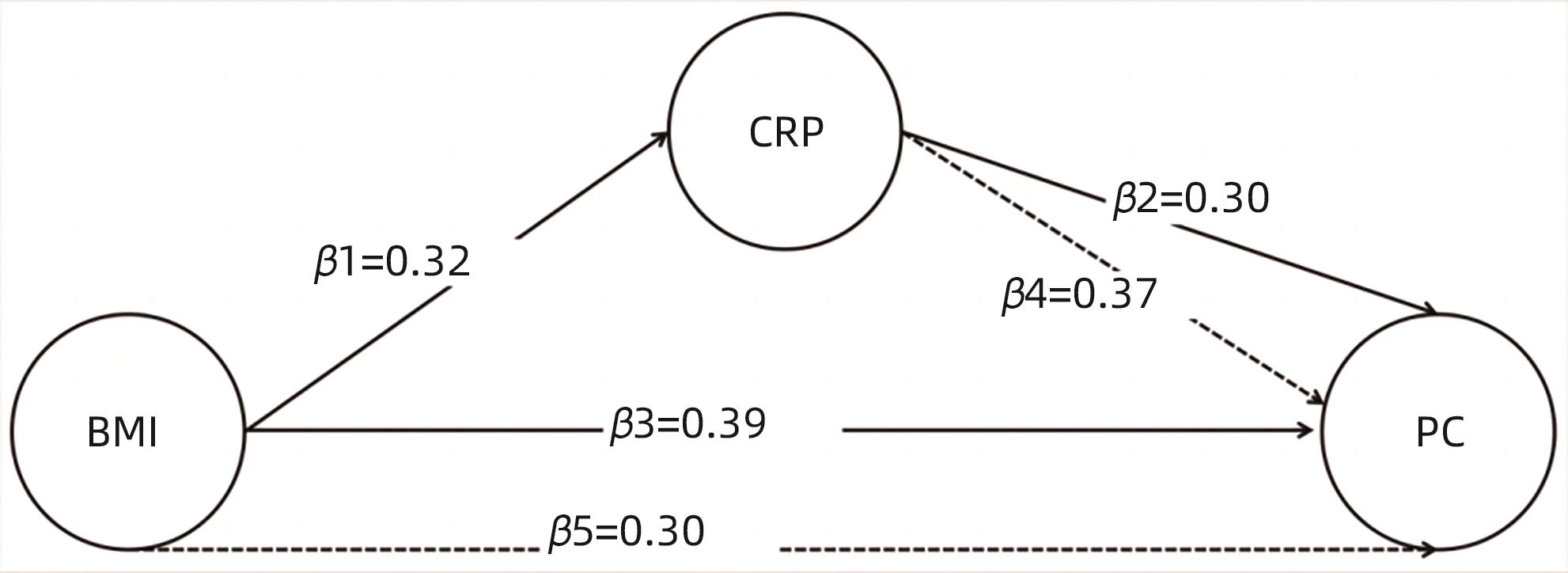
| [1] |
ARNOLD M, ABNET CC, NEALE RE, et al. Global burden of 5 major types of gastrointestinal cancer[J]. Gastroenterology, 2020, 159( 1): 335- 349. e 15. DOI: 10.1053/j.gastro.2020.02.068. |
| [2] |
ILIC M, ILIC I. Epidemiology of pancreatic cancer[J]. World J Gastroenterol, 2016, 22( 44): 9694- 9705. DOI: 10.3748/wjg.v22.i44.9694. |
| [3] |
SUNG H, FERLAY J, SIEGEL RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2021, 71( 3): 209- 249. DOI: 10.3322/caac.21660. |
| [4] |
FERLAY J, SOERJOMATARAM I, DIKSHIT R, et al. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012[J]. Int J Cancer, 2015, 136( 5): E359- E386. DOI: 10.1002/ijc.29210. |
| [5] |
HIDALGO M, CASCINU S, KLEEFF J, et al. Addressing the challenges of pancreatic cancer: Future directions for improving outcomes[J]. Pancreatology, 2015, 15( 1): 8- 18. DOI: 10.1016/j.pan.2014.10.001. |
| [6] |
General Office of National Health Commission. Standard for diagnosis and treatment of pancreatic cancer(2022 edition)[J]. J Clin Hepatol, 2022, 38( 5): 1006- 1015. DOI: 10.3969/j.issn.1001-5256.2022.05.007. |
| [7] |
MILLIKAN KW, DEZIEL DJ, SILVERSTEIN JC, et al. Prognostic factors associated with resectable adenocarcinoma of the head of the pancreas[J]. Am Surg, 1999, 65( 7): 618-623; discussion 623-624.
|
| [8] |
KOLBEINSSON HM, CHANDANA S, WRIGHT GP, et al. Pancreatic cancer: A review of current treatment and novel therapies[J]. J Invest Surg, 2023, 36( 1): 2129884. DOI: 10.1080/08941939.2022.2129884. |
| [9] |
PARKIN DM, BOYD L, WALKER LC. 16. The fraction of cancer attributable to lifestyle and environmental factors in the UK in 2010[J]. Br J Cancer, 2011, 105( Suppl 2): S77- S81. DOI: 10.1038/bjc.2011.489. |
| [10] |
|
| [11] |
BOSETTI C, BERTUCCIO P, NEGRI E, et al. Pancreatic cancer: Overview of descriptive epidemiology[J]. Mol Carcinog, 2012, 51( 1): 3- 13. DOI: 10.1002/mc.20785. |
| [12] |
ANAND P, KUNNUMAKKARA AB, SUNDARAM C, et al. Cancer is a preventable disease that requires major lifestyle changes[J]. Pharm Res, 2008, 25( 9): 2097- 2116. DOI: 10.1007/s11095-008-9661-9. |
| [13] |
|
| [14] |
DAVEY SMITH G, HEMANI G. Mendelian randomization: Genetic anchors for causal inference in epidemiological studies[J]. Hum Mol Genet, 2014, 23( R1): R89- R98. DOI: 10.1093/hmg/ddu328. |
| [15] |
ZHENG J, BAIRD D, BORGES MC, et al. Recent developments in Mendelian randomization studies[J]. Curr Epidemiol Rep, 2017, 4( 4): 330- 345. DOI: 10.1007/s40471-017-0128-6. |
| [16] |
LAWLOR DA, HARBORD RM, STERNE JA, et al. Mendelian randomization: Using genes as instruments for making causal inferences in epidemiology[J]. Stat Med, 2008, 27( 8): 1133- 1163. DOI: 10.1002/sim.3034. |
| [17] |
VERBANCK M, CHEN CY, NEALE B, et al. Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases[J]. Nat Genet, 2018, 50( 5): 693- 698. DOI: 10.1038/s41588-018-0099-7. |
| [18] |
BOWDEN J, DAVEY SMITH G, BURGESS S. Mendelian randomization with invalid instruments: Effect estimation and bias detection through Egger regression[J]. Int J Epidemiol, 2015, 44( 2): 512- 525. DOI: 10.1093/ije/dyv080. |
| [19] |
ZHAO J, MING JS, HU XH, et al. Bayesian weighted Mendelian randomization for causal inference based on summary statistics[J]. Bioinformatics, 2020, 36( 5): 1501- 1508. DOI: 10.1093/bioinformatics/btz749. |
| [20] |
LAWLOR DA. Commentary: Two-sample Mendelian randomization: Opportunities and challenges[J]. Int J Epidemiol, 2016, 45( 3): 908- 915. DOI: 10.1093/ije/dyw127. |
| [21] |
BURGESS S, BUTTERWORTH A, THOMPSON SG. Mendelian randomization analysis with multiple genetic variants using summarized data[J]. Genet Epidemiol, 2013, 37( 7): 658- 665. DOI: 10.1002/gepi.21758. |
| [22] |
YU TQ, XU WT, SU YN, et al. Basic principles, methods and limitations of Mendelian randomization studies[J]. Chin J Evid-Based Med, 2021, 21( 10): 1227- 1234. DOI: 10.7507/1672-2531.202107008. |
| [23] |
BOWDEN J, DAVEY SMITH G, HAYCOCK PC, et al. Consistent estimation in Mendelian randomization with some invalid instruments using a weighted Median estimator[J]. Genet Epidemiol, 2016, 40( 4): 304- 314. DOI: 10.1002/gepi.21965. |
| [24] |
BOWDEN J, DEL GRECO MF, MINELLI C, et al. Assessing the suitability of summary data for two-sample Mendelian randomization analyses using MR-Egger regression: The role of the I 2 statistic[J]. Int J Epidemiol, 2016, 45( 6): 1961- 1974. DOI: 10.1093/ije/dyw220. |
| [25] |
JIANG ZC, MOU YP, WANG HJ, et al. Causal effect between gut microbiota and pancreatic cancer: A two-sample Mendelian randomization study[J]. BMC Cancer, 2023, 23( 1): 1091. DOI: 10.1186/s12885-023-11493-y. |
| [26] |
ZHU SJ, DING Z. Association between gut microbiota and seven gastrointestinal diseases: A Mendelian randomized study[J]. J Gene Med, 2024, 26( 1): e3623. DOI: 10.1002/jgm.3623. |
| [27] |
ZHANG XN, ZHAO H, MAN JY, et al. Investigating causal associations of diet-derived circulating antioxidants with the risk of digestive system cancers: A Mendelian randomization study[J]. Nutrients, 2022, 14( 15): 3237. DOI: 10.3390/nu14153237. |
| [28] |
MIN Y, LIU ZR, LI RD, et al. Association between inflammatory bowel disease and pancreatic cancer: Results from the two-sample Mendelian randomization study[J]. Front Oncol, 2023, 13: 1155123. DOI: 10.3389/fonc.2023.1155123. |
| [29] |
PIERCE BL, AHSAN H, VANDERWEELE TJ. Power and instrument strength requirements for Mendelian randomization studies using multiple genetic variants[J]. Int J Epidemiol, 2011, 40( 3): 740- 752. DOI: 10.1093/ije/dyq151. |
| [30] |
ADAK A, KHAN MR. An insight into gut microbiota and its functionalities[J]. Cell Mol Life Sci, 2019, 76( 3): 473- 493. DOI: 10.1007/s00018-018-2943-4. |
| [31] |
de VOS WM, TILG H, van HUL M, et al. Gut microbiome and health: Mechanistic insights[J]. Gut, 2022, 71( 5): 1020- 1032. DOI: 10.1136/gutjnl-2021-326789. |
| [32] |
LIU MY. Arid1a: A gatekeeper in the development of pancreatic cancer from a rare precursor lesion[J]. Gastroenterology, 2022, 163( 2): 371- 373. DOI: 10.1053/j.gastro.2022.05.046. |
| [33] |
CAI J, CHEN HD, LU M, et al. Advances in the epidemiology of pancreatic cancer: Trends, risk factors, screening, and prognosis[J]. Cancer Lett, 2021, 520: 1- 11. DOI: 10.1016/j.canlet.2021.06.027. |
| [34] |
TONG Y, GAO HR, QI QC, et al. High fat diet, gut microbiome and gastrointestinal cancer[J]. Theranostics, 2021, 11( 12): 5889- 5910. DOI: 10.7150/thno.56157. |
| [35] |
COHEN LJ, CHO JH, GEVERS D, et al. Genetic factors and the intestinal microbiome guide development of microbe-based therapies for inflammatory bowel diseases[J]. Gastroenterology, 2019, 156( 8): 2174- 2189. DOI: 10.1053/j.gastro.2019.03.017. |
| [36] |
WONG SH, YU J. Gut microbiota in colorectal cancer: Mechanisms of action and clinical applications[J]. Nat Rev Gastroenterol Hepatol, 2019, 16( 11): 690- 704. DOI: 10.1038/s41575-019-0209-8. |
| [37] |
MA C, HAN MJ, HEINRICH B, et al. Gut microbiome-mediated bile acid metabolism regulates liver cancer via NKT cells[J]. Science, 2018, 360( 6391): eaan5931. DOI: 10.1126/science.aan5931. |
| [38] |
ERTZ-ARCHAMBAULT N, KEIM P, von HOFF D. Microbiome and pancreatic cancer: A comprehensive topic review of literature[J]. World J Gastroenterol, 2017, 23( 10): 1899- 1908. DOI: 10.3748/wjg.v23.i10.1899. |
| [39] |
YANG QY, ZHANG JH, ZHU Y. Potential roles of the gut microbiota in pancreatic carcinogenesis and therapeutics[J]. Front Cell Infect Microbiol, 2022, 12: 872019. DOI: 10.3389/fcimb.2022.872019. |
| [40] |
PUSHALKAR S, HUNDEYIN M, DALEY D, et al. The pancreatic cancer microbiome promotes oncogenesis by induction of innate and adaptive immune suppression[J]. Cancer Discov, 2018, 8( 4): 403- 416. DOI: 10.1158/2159-8290.CD-17-1134. |
| [41] |
SETHI V, KURTOM S, TARIQUE M, et al. Gut microbiota promotes tumor growth in mice by modulating immune response[J]. Gastroenterology, 2018, 155( 1): 33- 37. e 6. DOI: 10.1053/j.gastro.2018.04.001. |
| [42] |
YU Q, NEWSOME RC, BEVERIDGE M, et al. Intestinal microbiota modulates pancreatic carcinogenesis through intratumoral natural killer cells[J]. Gut Microbes, 2022, 14( 1): 2112881. DOI: 10.1080/19490976.2022.2112881. |
| [43] |
CARINI F, MAZZOLA M, RAPPA F, et al. Colorectal carcinogenesis: Role of oxidative stress and antioxidants[J]. Anticancer Res, 2017, 37( 9): 4759- 4766. DOI: 10.21873/anticanres.11882. |
| [44] |
SHIMIZU Y, TAMURA T, KEMMOCHI A, et al. Oxidative stress and Liver X Receptor agonist induce hepatocellular carcinoma in Non-alcoholic steatohepatitis model[J]. J Gastroenterol Hepatol, 2021, 36( 3): 800- 810. DOI: 10.1111/jgh.15239. |
| [45] |
AKBARI A, MAJD HM, RAHNAMA R, et al. Cross-talk between oxidative stress signaling and microRNA regulatory systems in carcinogenesis: Focused on gastrointestinal cancers[J]. Biomedecine Pharmacother, 2020, 131: 110729. DOI: 10.1016/j.biopha.2020.110729. |
| [46] |
|
| [47] |
YIN LL, YAN HH, CHEN KD, et al. Diet-derived circulating antioxidants and risk of digestive system tumors: A Mendelian randomization study[J]. Nutrients, 2022, 14( 16): 3274. DOI: 10.3390/nu14163274. |
| [48] |
SEMPERE-RUBIO N, AGUAS M, FAUBEL R. Association between chronotype, physical activity and sedentary behaviour: A systematic review[J]. Int J Environ Res Public Health, 2022, 19( 15): 9646. DOI: 10.3390/ijerph19159646. |
| [49] |
MAZRI FH, MANAF ZA, SHAHAR S, et al. The association between chronotype and dietary pattern among adults: A scoping review[J]. Int J Environ Res Public Health, 2019, 17( 1): 68. DOI: 10.3390/ijerph17010068. |
| [50] |
SROUR B, PLANCOULAINE S, ANDREEVA VA, et al. Circadian nutritional behaviours and cancer risk: New insights from the NutriNet-santé prospective cohort study: Disclaimers[J]. Int J Cancer, 2018, 143( 10): 2369- 2379. DOI: 10.1002/ijc.31584. |
| [51] |
CARASSO S, FISHMAN B, LASK LS, et al. Metagenomic analysis reveals the signature of gut microbiota associated with human chronotypes[J]. FASEB J, 2021, 35( 11): e22011. DOI: 10.1096/fj.202100857RR. |
| [52] |
MENG CT, BAI CM, BROWN TD, et al. Human gut microbiota and gastrointestinal cancer[J]. Genomics Proteomics Bioinformatics, 2018, 16( 1): 33- 49. DOI: 10.1016/j.gpb.2017.06.002. |
| [53] |
FENG Q, LIANG SS, JIA HJ, et al. Gut microbiome development along the colorectal adenoma-carcinoma sequence[J]. Nat Commun, 2015, 6: 6528. DOI: 10.1038/ncomms7528. |
| [54] |
YUAN S, MASON AM, TITOVA OE, et al. Morning chronotype and digestive tract cancers: Mendelian randomization study[J]. Int J Cancer, 2023, 152( 4): 697- 704. DOI: 10.1002/ijc.34284. |
| [55] |
RISK FACTOR COLLABORATION NCD-RISC) NCD. Worldwide trends in diabetes since 1980: A pooled analysis of 751 population-based studies with 4.4 million participants[J]. Lancet, 2016, 387( 10027): 1513- 1530. DOI: 10.1016/S0140-6736(16)00618-8. |
| [56] |
TSILIDIS KK, KASIMIS JC, LOPEZ DS, et al. Type 2 diabetes and cancer: Umbrella review of meta-analyses of observational studies[J]. BMJ, 2015, 350: g7607. DOI: 10.1136/bmj.g7607. |
| [57] |
PEARSON-STUTTARD J, PAPADIMITRIOU N, MARKOZANNES G, et al. Type 2 diabetes and cancer: An umbrella review of observational and Mendelian randomization studies[J]. Cancer Epidemiol Biomarkers Prev, 2021, 30( 6): 1218- 1228. DOI: 10.1158/1055-9965.EPI-20-1245. |
| [58] |
SHEN BY, LI YY, SHENG CS, et al. Association between age at diabetes onset or diabetes duration and subsequent risk of pancreatic cancer: Results from a longitudinal cohort and Mendelian randomization study[J]. Lancet Reg Health West Pac, 2023, 30: 100596. DOI: 10.1016/j.lanwpc.2022.100596. |
| [59] |
INCIO J, LIU H, SUBOJ P, et al. Obesity-induced inflammation and desmoplasia promote pancreatic cancer progression and resistance to chemotherapy[J]. Cancer Discov, 2016, 6( 8): 852- 869. DOI: 10.1158/2159-8290.CD-15-1177. |
| [60] |
PARK JH, AMERI AH, DEMPSEY KE, et al. Nuclear IL-33/SMAD signaling axis promotes cancer development in chronic inflammation[J]. EMBO J, 2021, 40( 7): e106151. DOI: 10.15252/embj.2020106151. |
| [61] |
YUDKIN JS, STEHOUWER CD, EMEIS JJ, et al. C-reactive protein in healthy subjects: Associations with obesity, insulin resistance, and endothelial dysfunction: A potential role for cytokines originating from adipose tissue?[J]. Arterioscler Thromb Vasc Biol, 1999, 19( 4): 972- 978. DOI: 10.1161/01.atv.19.4.972. |
| [62] |
XU HY, BARNES GT, YANG Q, et al. Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance[J]. J Clin Invest, 2003, 112( 12): 1821- 1830. DOI: 10.1172/JCI19451. |
| [63] |
SINGH A, MAYENGBAM SS, YADUVANSHI H, et al. Obesity programs macrophages to support cancer progression[J]. Cancer Res, 2022, 82( 23): 4303- 4312. DOI: 10.1158/0008-5472.CAN-22-1257. |
| [64] |
XIA B, HE QS, PAN YH, et al. Metabolic syndrome and risk of pancreatic cancer: A population-based prospective cohort study[J]. Int J Cancer, 2020, 147( 12): 3384- 3393. DOI: 10.1002/ijc.33172. |
| [65] |
LI ZQ, JIN LQ, XIA L, et al. Body mass index, C-reactive protein, and pancreatic cancer: A Mendelian randomization analysis to investigate causal pathways[J]. Front Oncol, 2023, 13: 1042567. DOI: 10.3389/fonc.2023.1042567. |
| [66] |
SON J, LYSSIOTIS CA, YING HQ, et al. Glutamine supports pancreatic cancer growth through a KRAS-regulated metabolic pathway[J]. Nature, 2013, 496( 7443): 101- 105. DOI: 10.1038/nature12040. |
| [67] |
CHEUNG PK, MA MH, TSE HF, et al. The applications of metabolomics in the molecular diagnostics of cancer[J]. Expert Rev Mol Diagn, 2019, 19( 9): 785- 793. DOI: 10.1080/14737159.2019.1656530. |
| [68] |
XIE GX, LU LG, QIU YP, et al. Plasma metabolite biomarkers for the detection of pancreatic cancer[J]. J Proteome Res, 2015, 14( 2): 1195- 1202. DOI: 10.1021/pr501135f. |
| [69] |
LI QX, JIN M, LIU YH, et al. Gut microbiota: Its potential roles in pancreatic cancer[J]. Front Cell Infect Microbiol, 2020, 10: 572492. DOI: 10.3389/fcimb.2020.572492. |
| [70] |
ZHONG H, LIU S, ZHU JJ, et al. Elucidating the role of blood metabolites on pancreatic cancer risk using two-sample Mendelian randomization analysis[J]. Int J Cancer, 2024, 154( 5): 852- 862. DOI: 10.1002/ijc.34771. |
| [71] |
NADEEM MS, KUMAR V, AL-ABBASI FA, et al. Risk of colorectal cancer in inflammatory bowel diseases[J]. Semin Cancer Biol, 2020, 64: 51- 60. DOI: 10.1016/j.semcancer.2019.05.001. |
| [72] |
ZHANG HM, ZHANG MM, CHEN XF, et al. Risk of malignancy in patients with inflammatory bowel disease: A population-based cohort study from China[J]. Int J Cancer, 2022, 150( 11): 1770- 1778. DOI: 10.1002/ijc.33932. |
| [73] |
JUNG YS, HAN M, PARK S, et al. Cancer risk in the early stages of inflammatory bowel disease in Korean patients: A nationwide population-based study[J]. J Crohns Colitis, 2017, 11( 8): 954- 962. DOI: 10.1093/ecco-jcc/jjx040. |
| [74] |
ÅH EVERHOV, ERICHSEN R, SACHS MC, et al. Inflammatory bowel disease and pancreatic cancer: A Scandinavian register-based cohort study 1969-2017[J]. Aliment Pharmacol Ther, 2020, 52( 1): 143- 154. DOI: 10.1111/apt.15785. |
| [75] |
LO B, ZHAO M, VIND I, et al. The risk of extraintestinal cancer in inflammatory bowel disease: A systematic review and meta-analysis of population-based cohort studies[J]. Clin Gastroenterol Hepatol, 2021, 19( 6): 1117- 1138. e 19. DOI: 10.1016/j.cgh.2020.08.015. |
| [76] |
DENHAM W, YANG J, FINK G, et al. Gene targeting demonstrates additive detrimental effects of interleukin 1 and tumor necrosis factor during pancreatitis[J]. Gastroenterology, 1997, 113( 5): 1741- 1746. DOI: 10.1053/gast.1997.v113.pm9352880. |
| [77] |
MARRACHE F, TU SP, BHAGAT G, et al. Overexpression of interleukin-1beta in the murine pancreas results in chronic pancreatitis[J]. Gastroenterology, 2008, 135( 4): 1277- 1287. DOI: 10.1053/j.gastro.2008.06.078. |
| [78] |
TAKAHASHI R, MACCHINI M, SUNAGAWA M, et al. Interleukin-1β- induced pancreatitis promotes pancreatic ductal adenocarcinoma via B lymphocyte-mediated immune suppression[J]. Gut, 2021, 70( 2): 330- 341. DOI: 10.1136/gutjnl-2019-319912. |
| [79] |
ZHANG Y, CHEN XY, WANG HM, et al. Innate immune mediator, Interleukin-1 receptor accessory protein(IL1RAP), is expressed and pro-tumorigenic in pancreatic cancer[J]. J Hematol Oncol, 2022, 15( 1): 70. DOI: 10.1186/s13045-022-01286-4. |
| [80] |
TJOMSLAND V, SANDNES D, POMIANOWSKA E, et al. The TGFβ- SMAD3 pathway inhibits IL-1α induced interactions between human pancreatic stellate cells and pancreatic carcinoma cells and restricts cancer cell migration[J]. J Exp Clin Cancer Res, 2016, 35( 1): 122. DOI: 10.1186/s13046-016-0400-5. |
| [81] |
MAKER AV, KATABI N, QIN LX, et al. Cyst fluid interleukin-1beta(IL1beta) levels predict the risk of carcinoma in intraductal papillary mucinous neoplasms of the pancreas[J]. Clin Cancer Res, 2011, 17( 6): 1502- 1508. DOI: 10.1158/1078-0432.CCR-10-1561. |
| [82] |
YUAN S, MIAO YY, RUAN XX, et al. Therapeutic role of interleukin-1 receptor antagonist in pancreatic diseases: Mendelian randomization study[J]. Front Immunol, 2023, 14: 1240754. DOI: 10.3389/fimmu.2023.1240754. |
| [83] |
GRECO M FD, MINELLI C, SHEEHAN NA, et al. Detecting pleiotropy in Mendelian randomisation studies with summary data and a continuous outcome[J]. Stat Med, 2015, 34( 21): 2926- 2940. DOI: 10.1002/sim.6522. |
| [84] |
WANG X, GAO HY, ZENG YY, et al. A Mendelian analysis of the relationships between immune cells and breast cancer[J]. Front Oncol, 2024, 14: 1341292. DOI: 10.3389/fonc.2024.1341292. |
| [85] |
MORRISON J, KNOBLAUCH N, MARCUS JH, et al. Mendelian randomization accounting for correlated and uncorrelated pleiotropic effects using genome-wide summary statistics[J]. Nat Genet, 2020, 52( 7): 740- 747. DOI: 10.1038/s41588-020-0631-4. |
| [86] |
BARANOVA A, CAO HB, ZHANG FQ. Causal effect of COVID-19 on Alzheimer’s disease: A Mendelian randomization study[J]. J Med Virol, 2023, 95( 1): e28107. DOI: 10.1002/jmv.28107. |
| [87] |
MICHAËLSSON M, YUAN S, MELHUS H, et al. The impact and causal directions for the associations between diagnosis of ADHD, socioeconomic status, and intelligence by use of a bi-directional two-sample Mendelian randomization design[J]. BMC Med, 2022, 20( 1): 106. DOI: 10.1186/s12916-022-02314-3. |
| [88] |
SANDERSON E. Multivariable Mendelian randomization and mediation[J]. Cold Spring Harb Perspect Med, 2021, 11( 2): a038984. DOI: 10.1101/cshperspect.a038984. |
| [89] |
WILLER CJ, SCHMIDT EM, SENGUPTA S, et al. Discovery and refinement of loci associated with lipid levels[J]. Nat Genet, 2013, 45( 11): 1274- 1283. DOI: 10.1038/ng.2797. |
| [90] |
WÜRTZ P, KANGAS AJ, SOININEN P, et al. Lipoprotein subclass profiling reveals pleiotropy in the genetic variants of lipid risk factors for coronary heart disease: A note on Mendelian randomization studies[J]. J Am Coll Cardiol, 2013, 62( 20): 1906- 1908. DOI: 10.1016/j.jacc.2013.07.085. |
| [91] |
BURGESS S, THOMPSON SG. Multivariable Mendelian randomization: The use of pleiotropic genetic variants to estimate causal effects[J]. Am J Epidemiol, 2015, 181( 4): 251- 260. DOI: 10.1093/aje/kwu283. |
| [92] |
BURGESS S, DUDBRIDGE F, THOMPSON SG. Re:“Multivariable Mendelian randomization: The use of pleiotropic genetic variants to estimate causal effects”[J]. Am J Epidemiol, 2015, 181( 4): 290- 291. DOI: 10.1093/aje/kwv017. |
| [93] |
KEMP JP, SAYERS A, SMITH GD, et al. Using Mendelian randomization to investigate a possible causal relationship between adiposity and increased bone mineral density at different skeletal sites in children[J]. Int J Epidemiol, 2016, 45( 5): 1560- 1572. DOI: 10.1093/ije/dyw079. |



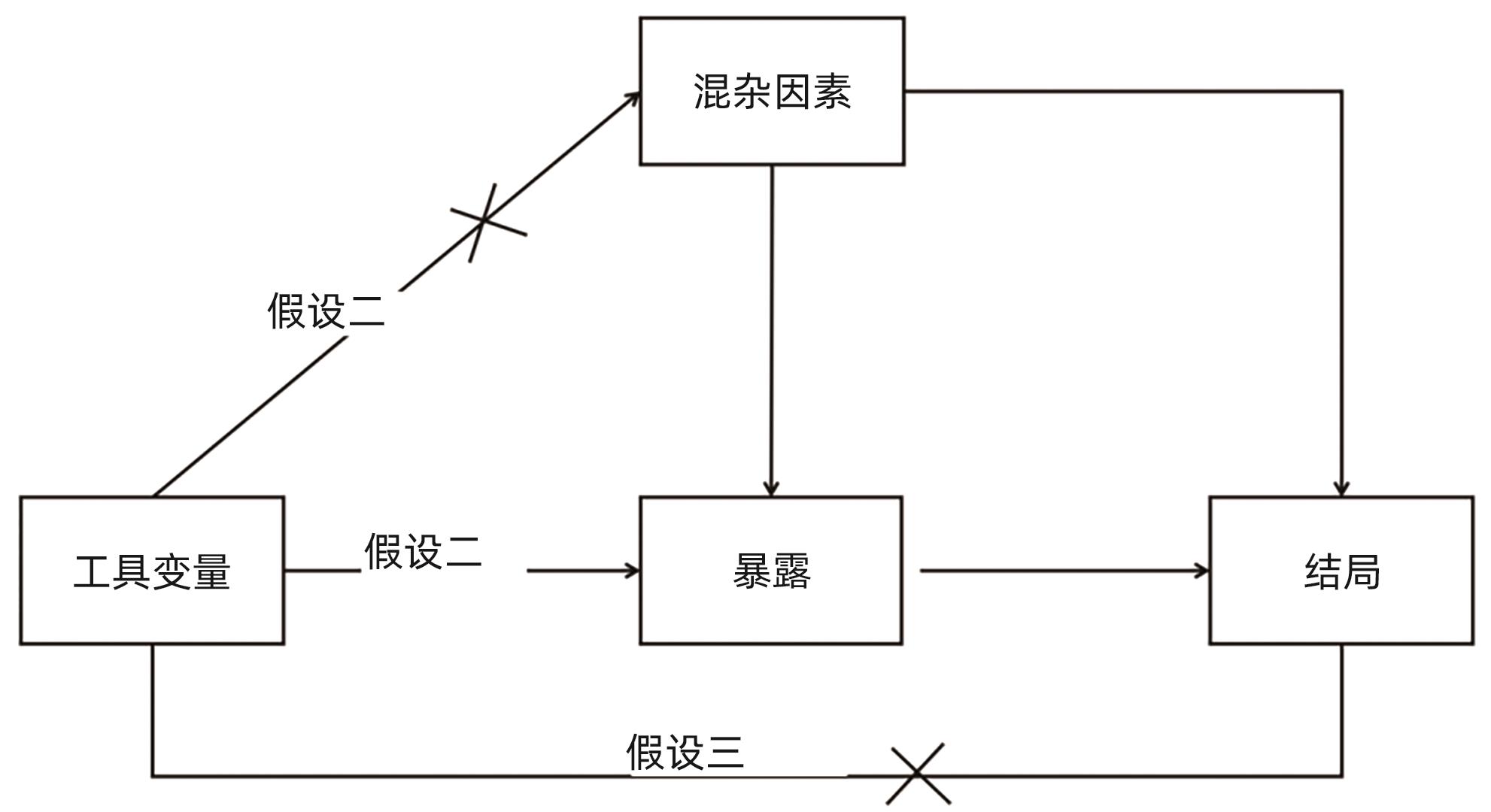




 DownLoad:
DownLoad: