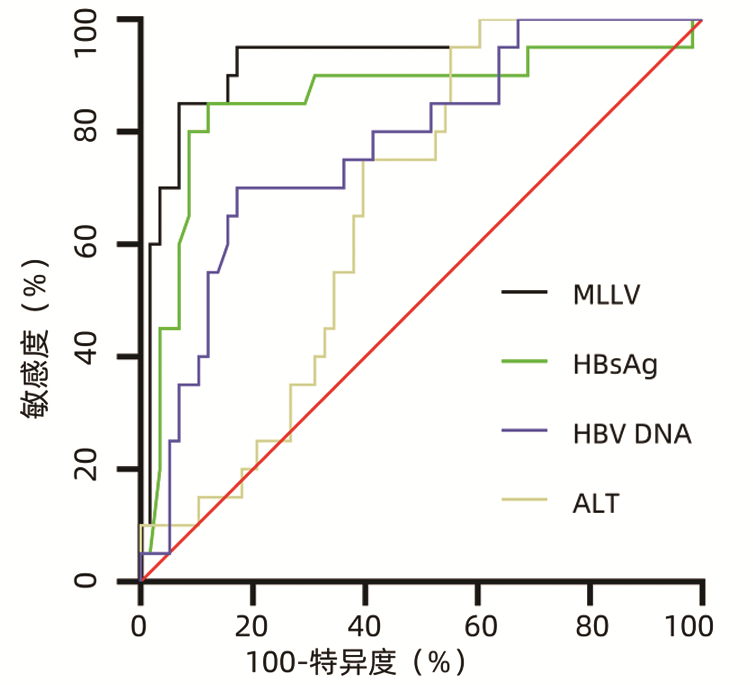
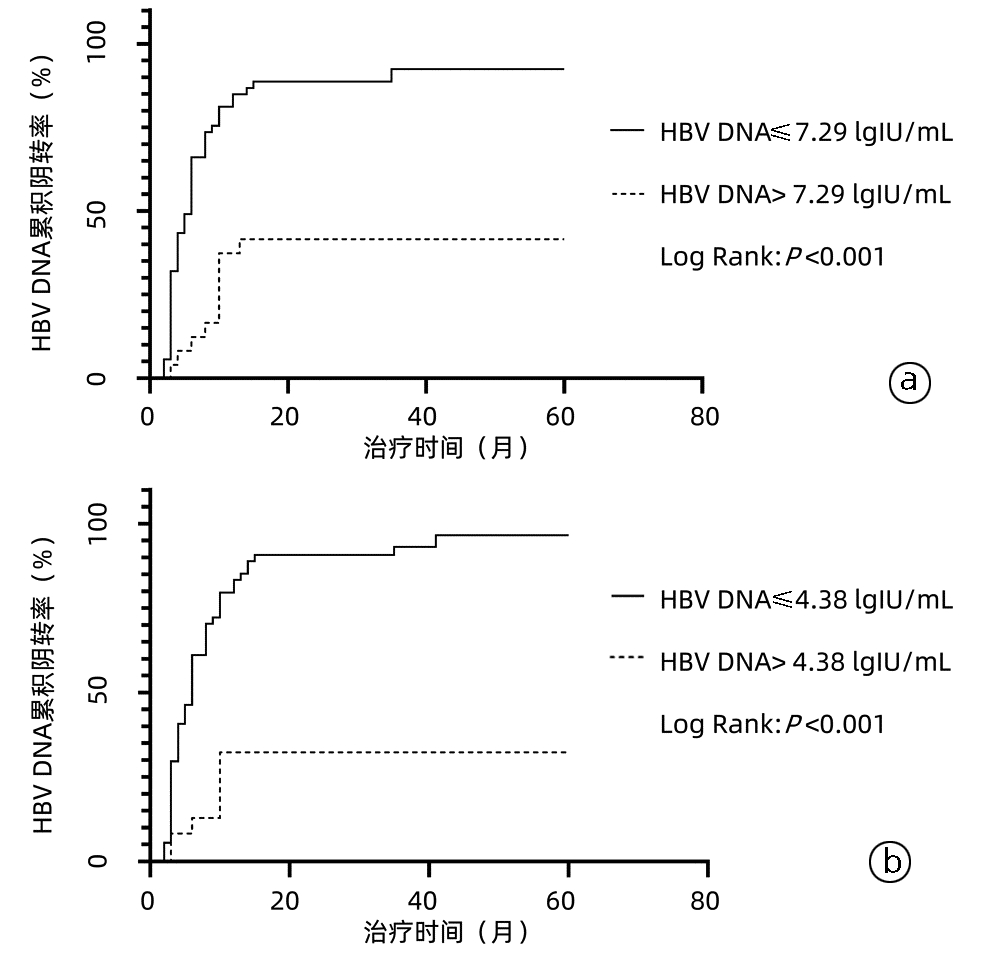
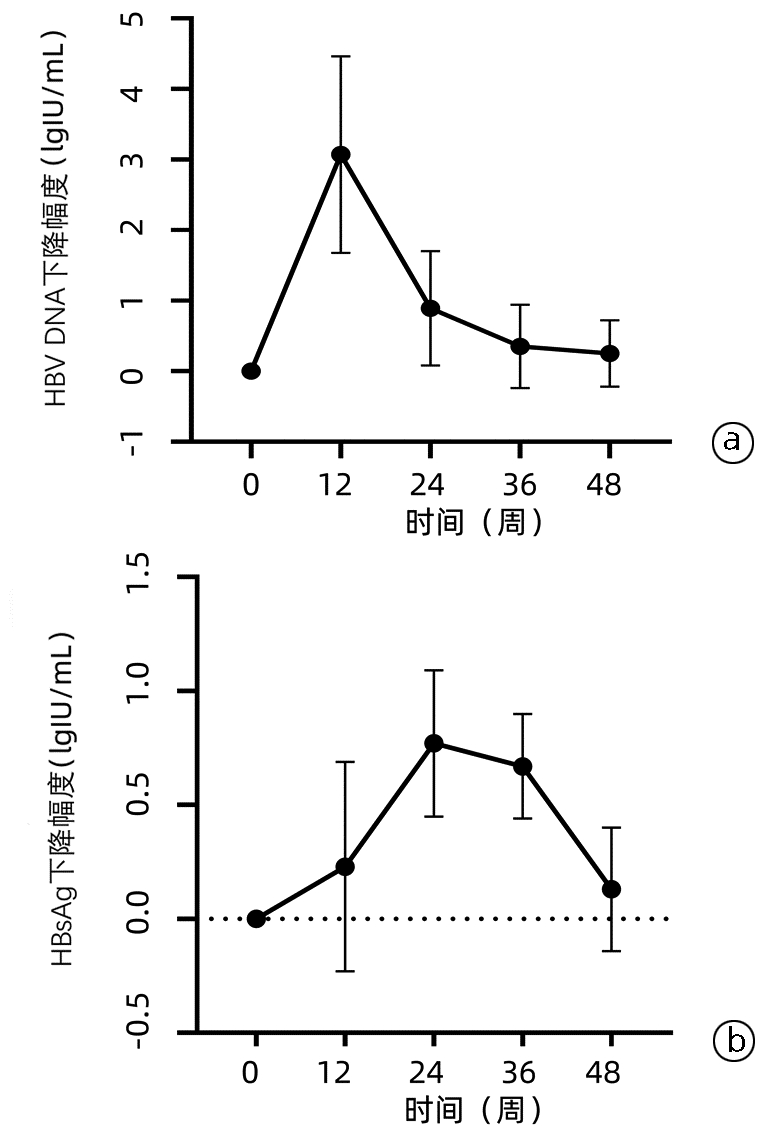
| [1] |
HUTIN Y, NASRULLAH M, EASTERBROOK P, et al. Access to treatment for hepatitis B virus infection-worldwide, 2016[J]. MMWR Morb Mortal Wkly Rep, 2018, 67(28): 773-777. DOI: 10.15585/mmwr.mm6728a2. |
| [2] |
Polaris Observatory Collaborators. Global prevalence, treatment, and prevention of hepatitis B virus infection in 2016: a modelling study[J]. Lancet Gastroenterol Hepatol, 2018, 3(6): 383-403. DOI: 10.1016/S2468-1253(18)30056-6. |
| [3] |
Chinese Society of Infectious Disease, Chinese Society of Hepatology, Chinese Medical Association. The expert consensus on clinical cure (functional cure) of chronic hepatitis B[J]. J Clin Hepatol, 2019, 35(8): 1693-1701. DOI: 10.3969/j.issn.1001-5256.2019.08.008. |
| [4] |
KIM HJ, CHO YK, JEON WK, et al. Clinical characteristics of patients with chronic hepatitis B who developed genotypic resistance to entecavir: Real-life experience[J]. Clin Mol Hepatol, 2017, 23(4): 323-330. DOI: 10.3350/cmh.2017.0005. |
| [5] |
SHIN SK, YIM HJ, KIM JH, et al. Partial virological response after 2 years of entecavir therapy increases the risk of hepatocellular carcinoma in patients with hepatitis B virus-associated cirrhosis[J]. Gut Liver, 2021, 15(3): 430-439. DOI: 10.5009/gnl20074. |
| [6] |
SUN Y, WU X, ZHOU J, et al. Persistent low level of hepatitis B virus promotes fibrosis progression during therapy[J]. Clin Gastroenterol Hepatol, 2020, 18(11): 2582-2591. e6. DOI: 10.1016/j.cgh.2020.03.001. |
| [7] |
MAK LY, HUANG Q, WONG DK, et al. Residual HBV DNA and pgRNA viraemia is associated with hepatocellular carcinoma in chronic hepatitis B patients on antiviral therapy[J]. J Gastroenterol, 2021, 56(5): 479-488. DOI: 10.1007/s00535-021-01780-5. |
| [8] |
Chinese Society of Infectious Diseases, Chinese Medical Association, Chinese Society of Hepatology, Chinese Medical Association. Guidelines for the prevention and treatment of chronic hepatitis B (version 2019)[J]. J Clin Hepatol, 2019, 35(12): 2648-2669. DOI: 10.3969/j.issn.1001-5256.2019.12.007. |
| [9] |
LU FM, FENG B, ZHENG SJ, et al. Current status of the research on low-level viremia in chronic hepatitis B patients receiving nucleos(t)ide analogues[J]. J Clin Hepatol, 2021, 37(6): 1268-1274. DOI: 10.3969/j.issn.1001-5256.2021.06.007. |
| [10] |
|
| [11] |
LI H, XU WT, DENG BC, et al. Research progress in the functional treatment of chronic hepatitis B with nucleoside (acid) analogues and pegylated interferon[J]. Clin J Med Offic, 2022, 50(9): 890-893. DOI: 10.16680/j.1671-3826.2022.09.04. |
| [12] |
OGAWA E, NOMURA H, NAKAMUTA M, et al. Tenofovir alafenamide after switching from entecavir or nucleos(t)ide combination therapy for patients with chronic hepatitis B[J]. Liver Int, 2020, 40(7): 1578-1589. DOI: 10.1111/liv.14482. |
| [13] |
AGARWAL K, BRUNETTO M, SETO WK, et al. 96 weeks treatment of tenofovir alafenamide vs. tenofovir disoproxil fumarate for hepatitis B virus infection[J]. J Hepatol, 2018, 68(4): 672-681. DOI: 10.1016/j.jhep.2017.11.039. |
| [14] |
LEE SB, JEONG J, PARK JH, et al. Low-level viremia and cirrhotic complications in patients with chronic hepatitis B according to adherence to entecavir[J]. Clin Mol Hepatol, 2020, 26(3): 364-375. DOI: 10.3350/cmh.2020.0012. |
| [15] |
KIM JH, SINN DH, KANG W, et al. Low-level viremia and the increased risk of hepatocellular carcinoma in patients receiving entecavir treatment[J]. Hepatology, 2017, 66(2): 335-343. DOI: 10.1002/hep.28916. |
| [16] |
REVILL PA, CHISARI FV, BLOCK JM, et al. A global scientific strategy to cure hepatitis B[J]. Lancet Gastroenterol Hepatol, 2019, 4(7): 545-558. DOI: 10.1016/S2468-1253(19)30119-0. |
| [17] |
WU IC, LAI CL, HAN SH, et al. Efficacy of entecavir in chronic hepatitis B patients with mildly elevated alanine aminotransferase and biopsy-proven histological damage[J]. Hepatology, 2010, 51(4): 1185-1189. DOI: 10.1002/hep.23424. |
| [18] |
ZHANG Q, PENG H, LIU X, et al. Chronic hepatitis B infection with low level viremia correlates with the progression of the liver disease[J]. J Clin Transl Hepatol, 2021, 9(6): 850-859. DOI: 10.14218/JCTH.2021.00046. |
| [19] |
BAO T, HU QG, YE J, et al. Value of HBsAg level in dynamic monitoring of disease progression in patients with chronic HBV infection[J]. J Clin Hepatol, 2017, 33(8): 1475-1478. DOI: 10.3969/j.issn.1001-5256.2017.08.012. |
| [20] |
SONG JC, MIN BY, KIM JW, et al. Pretreatment serum HBsAg-to-HBV DNA ratio predicts a virologic response to entecavir in chronic hepatitis B[J]. Korean J Hepatol, 2011, 17(4): 268-273. DOI: 10.3350/kjhep.2011.17.4.268. |
| [21] |
LEE JM, AHN SH, KIM HS, et al. Quantitative hepatitis B surface antigen and hepatitis B e antigen titers in prediction of treatment response to entecavir[J]. Hepatology, 2011, 53(5): 1486-1493. DOI: 10.1002/hep.24221. |
| [22] |
CHEN H, FU JJ, LI L, et al. Influencing factors for low-level viremia in chronic hepatitis B patients treated with long-term entecavir antiviral therapy[J]. J Clin Hepatol, 2021, 37(3): 556-559. DOI: 10.3969/j.issn.1001-5256.2021.03.011. |
| [23] |
LIM SG, PHYO WW, LING J, et al. Comparative biomarkers for HBsAg loss with antiviral therapy shows dominant influence of quantitative HBsAg (qHBsAg)[J]. Aliment Pharmacol Ther, 2021, 53(1): 172-182. DOI: 10.1111/apt.16149. |



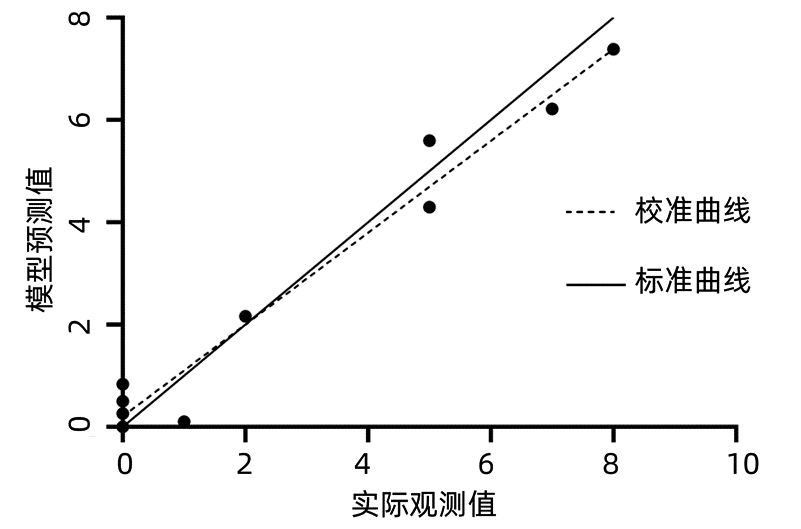




 DownLoad:
DownLoad: