| [1] |
BRAY F, FERLAY J, SOERJOMATARAM I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2018, 68(6): 394-424. DOI: 10.3322/caac.21492. |
| [2] |
|
| [3] |
RUMGAY H, ARNOLD M, FERLAY J, et al. Global burden of primary liver cancer in 2020 and predictions to 2040[J]. J Hepatol, 2022, 77(6): 1598-1606. DOI: 10.1016/j.jhep.2022.08.021. |
| [4] |
European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma[J]. J Hepatol, 2018, 69(1): 182-236. DOI: 10.1016/j.jhep.2018.03.019. |
| [5] |
WILHELM SM, CARTER C, TANG L, et al. BAY 43-9006 exhibits broad spectrum oral antitumor activity and targets the RAF/MEK/ERK pathway and receptor tyrosine kinases involved in tumor progression and angiogenesis[J]. Cancer Res, 2004, 64(19): 7099-7109. DOI: 10.1158/0008-5472.CAN-04-1443. |
| [6] |
GAO R, KALATHUR R, COTO-LLERENA M, et al. YAP/TAZ and ATF4 drive resistance to Sorafenib in hepatocellular carcinoma by preventing ferroptosis[J]. EMBO Mol Med, 2021, 13(12): e14351. DOI: 10.15252/emmm.202114351. |
| [7] |
ZHOU YY, ZHAO XX, CHEN YY, et al. Tumor immune checkpoint inhibitor and its combination research progress of combination therapy[J]. China pharmacy, 2020, 31(7): 890-896. DOI: 10.6039/j.issn.1001-0408.2020.07.24. |
| [8] |
QIN S, REN Z, MENG Z, et al. Camrelizumab in patients with previously treated advanced hepatocellular carcinoma: a multicentre, open-label, parallel-group, randomised, phase 2 trial[J]. Lancet Oncol, 2020, 21(4): 571-580. DOI: 10.1016/S1470-2045(20)30011-5. |
| [9] |
KUDO M. Immune checkpoint blockade in hepatocellular carcinoma: 2017 update[J]. Liver Cancer, 2016, 6(1): 1-12. DOI: 10.1159/000449342. |
| [10] |
ZHAO X, CHEN Q, LIU W, et al. Codelivery of doxorubicin and curcumin with lipid nanoparticles results in improved efficacy of chemotherapy in liver cancer[J]. Int J Nanomedicine, 2014, 10: 257-270. DOI: 10.2147/IJN.S73322. |
| [11] |
GAO YX, YANG TW, YIN JM, et al. Progress and prospects of biomarkers in primary liver cancer (Review)[J]. Int J Oncol, 2020, 57(1): 54-66. DOI: 10.3892/ijo.2020.5035. |
| [12] |
HAO X, FAN R, HOU JL. Early warning and accurate screening for the high-risk population of hepatocellular carcinoma[J]. J Clin Hepatol, 2022, 38(3): 499-504. DOI: 10.3969/j.issn.1001-5256.2022.03.002. |
| [13] |
LENCIONI R, LLOVET JM. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma[J]. Semin Liver Dis, 2010, 30(1): 52-60. DOI: 10.1055/s-0030-1247132. |
| [14] |
SHIBA S, OKUSAKA T, IKEDA M, et al. Characteristics of 18 patients with hepatocellular carcinoma who obtained a complete response after treatment with sorafenib[J]. Hepatol Res, 2014, 44(13): 1268-1276. DOI: 10.1111/hepr.12297. |
| [15] |
LIU YC, CHENG TC, BIAN ZL. Progress and challenges of combined immune checkpoint inhibitors in the treatment of hepatocellular carcinoma[J/OL]. Chin J Immunol, 2023. [Online ahead of print]
刘一村, 程苕莼, 卞兆连. 联合免疫检查点抑制剂治疗肝细胞癌的进展与挑战[J/OL]. 中国免疫学杂志, 2023. [网络首发]
|
| [16] |
CHANG YS, ADNANE J, TRAIL PA, et al. Sorafenib (BAY 43-9006) inhibits tumor growth and vascularization and induces tumor apoptosis and hypoxia in RCC xenograft models[J]. Cancer Chemother Pharmacol, 2007, 59(5): 561-574. DOI: 10.1007/s00280-006-0393-4. |
| [17] |
FU Y, WEI X, LIN L, et al. Adverse reactions of sorafenib, sunitinib, and imatinib in treating digestive system tumors[J]. Thorac Cancer, 2018, 9(5): 542-547. DOI: 10.1111/1759-7714.12608. |
| [18] |
European Association for the Study of the Liver, European Organisation for Research and Treatment of Cancer. EASL-EORTC clinical practice guidelines: management of hepatocellular carcinoma[J]. J Hepatol, 2012, 56(4): 908-943. DOI: 10.1016/j.jhep.2011.12.001. |
| [19] |
MOTZER RJ, AGARWAL N, BEARD C, et al. NCCN clinical practice guidelines in oncology: kidney cancer[J]. J Natl Compr Canc Netw, 2009, 7(6): 618-630. DOI: 10.6004/jnccn.2009.0043. |
| [20] |
FINN RS, IKEDA M, ZHU AX, et al. Phase Ib study of lenvatinib plus pembrolizumab in patients with unresectable hepatocellular carcinoma[J]. J Clin Oncol, 2020, 38(26): 2960-2970. DOI: 10.1200/JCO.20.00808. |
| [21] |
FINN RS, QIN S, IKEDA M, et al. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma[J]. N Engl J Med, 2020, 382(20): 1894-1905. DOI: 10.1056/NEJMoa1915745. |
| [22] |
KAO WY, CHIOU YY, HUNG HH, et al. Serum alpha-fetoprotein response can predict prognosis in hepatocellular carcinoma patients undergoing radiofrequency ablation therapy[J]. Clin Radiol, 2012, 67(5): 429-436. DOI: 10.1016/j.crad.2011.10.009. |
| [23] |
SHAO YY, LIN ZZ, HSU C, et al. Early alpha-fetoprotein response predicts treatment efficacy of antiangiogenic systemic therapy in patients with advanced hepatocellular carcinoma[J]. Cancer, 2010, 116(19): 4590-4596. DOI: 10.1002/cncr.25257. |
| [24] |
SHAO YY, LIU TH, HSU C, et al. Early alpha-foetoprotein response associated with treatment efficacy of immune checkpoint inhibitors for advanced hepatocellular carcinoma[J]. Liver Int, 2019, 39(11): 2184-2189. DOI: 10.1111/liv.14210. |
| [25] |
PAUL SB, SAHU P, SREENIVAS V, et al. Prognostic role of serial alpha-fetoprotein levels in hepatocellular carcinoma treated with locoregional therapy[J]. Scand J Gastroenterol, 2019, 54(9): 1132-1137. DOI: 10.1080/00365521.2019.1660403. |
| [26] |
SÁNCHEZ A, ROCES LV, GARCÍA IZ, et al. Value of α-fetoprotein as an early biomarker for treatment response to sorafenib therapy in advanced hepatocellular carcinoma[J]. Oncol Lett, 2018, 15(6): 8863-8870. DOI: 10.3892/ol.2018.8400. |
| [27] |
PARK JG. Long-term outcomes of patients with advanced hepatocellular carcinoma who achieved complete remission after sorafenib therapy[J]. Clin Mol Hepatol, 2015, 21(3): 287-294. DOI: 10.3350/cmh.2015.21.3.287. |
| [28] |
LLOVET JM, RICCI S, MAZZAFERRO V, et al. Sorafenib in advanced hepatocellular carcinoma[J]. N Engl J Med, 2008, 359(4): 378-390. DOI: 10.1056/NEJMoa0708857. |
| [29] |
CHEN LT, LIU TW, CHAO Y, et al. alpha-fetoprotein response predicts survival benefits of thalidomide in advanced hepatocellular carcinoma[J]. Aliment Pharmacol Ther, 2005, 22(3): 217-226. DOI: 10.1111/j.1365-2036.2005.02547.x. |



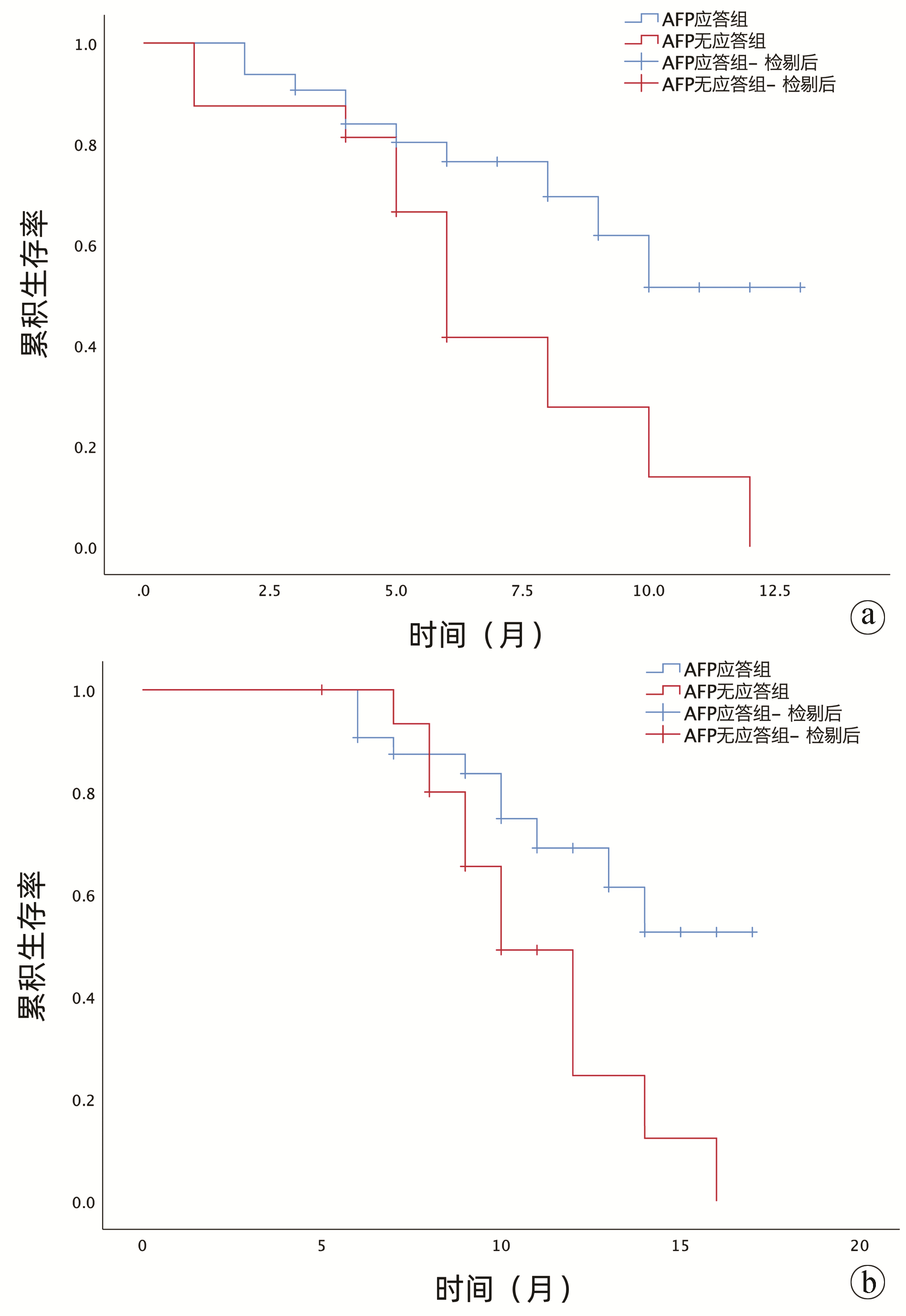




 DownLoad:
DownLoad: