| [1] |
HUBER W, PHILLIP V, SCHMID R, et al. Acute Pancreatitis: What is new?[J]. Dtsch Med Wochenschr, 2017, 142(7): 525-529. DOI: 10.1055/s-0042-100233. |
| [2] |
VEGE SS, GARDNER TB, CHARI ST, et al. Low mortality and high morbidity in severe acute pancreatitis without organ failure: A case for revising the Atlanta classification to include "moderately severe acute pancreatitis"[J]. Am J Gastroenterol, 2009, 104(3): 710-715. DOI: 10.1038/ajg.2008.77. |
| [3] |
GUDA NM, TRIKUDANATHAN G, FREEMAN ML. Idiopathic recurrent acute pancreatitis[J]. Lancet Gastroenterol Hepatol, 2018, 3(10): 720-728. DOI: 10.1016/S2468-1253(18)30211-5. |
| [4] |
WANG L, ZENG YB, CHEN JY, et al. A simple new scoring system for predicting the mortality of severe acute pancreatitis: A retrospective clinical study[J]. Medicine (Baltimore), 2020, 99(23): e20646. DOI: 10.1097/MD.0000000000020646. |
| [5] |
Pancreas Study Group, Chinese Society of Gastroenterology, Chinese Medical Association; Editorial Board of Chinese Journal of Pancreatology; Editorial Board of Chinese Journal of Digestion. Chinese guidelines for the management of acute pancreatitis (Shengyang, 2019)[J]. J Clin Hepatol, 2019, 35(12): 2706-2711. DOI: 10.3969/j.issn.1001-5256.2019.12.013. |
| [6] |
WU BU, JOHANNES RS, SUN X, et al. The early prediction of mortality in acute pancreatitis: A large population-based study[J]. Gut, 2008, 57(12): 1698-1703. DOI: 10.1136/gut.2008.152702. |
| [7] |
ALHAJERI A, ERWIN S. Acute pancreatitis: Value and impact of CT severity index[J]. Abdom Imaging, 2008, 33(1): 18-20. DOI: 10.1007/s00261-007-9315-0. |
| [8] |
|
| [9] |
CHEN HJ, WANG JJ, TSAY WI, et al. Epidemiology and outcome of acute pancreatitis in end-stage renal disease dialysis patients: A 10-year national cohort study[J]. Nephrol Dial Transplant, 2017, 32(10): 1731-1736. DOI: 10.1093/ndt/gfw400. |
| [10] |
GARG PK, SINGH VP. Organ failure due to systemic injury in acute pancreatitis[J]. Gastroenterology, 2019, 156(7): 2008-2023. DOI: 10.1053/j.gastro.2018.12.041. |
| [11] |
|
| [12] |
TAN Y, RAFI S, TYEBALLY FANG M, et al. Validation of the modified Ranson versus Glasgow score for pancreatitis in a Singaporean population[J]. ANZ J Surg, 2017, 87(9): 700-703. DOI: 10.1111/ans.13139. |
| [13] |
KNOEPFLI AS, KINKEL K, BERNEY T, et al. Prospective study of 310 patients: Can early CT predict the severity of acute pancreatitis?[J]. Abdom Imaging, 2007, 32(1): 111-115. DOI: 10.1007/s00261-006-9034-y. |
| [14] |
YE JF, ZHAO YX, JU J, et al. Building and verifying a severity prediction model of acute pancreatitis (AP) based on BISAP, MEWS and routine test indexes[J]. Clin Res Hepatol Gastroenterol, 2017, 41(5): 585-591. DOI: 10.1016/j.clinre.2016.11.013. |
| [15] |
SAHU B, ABBEY P, ANAND R, et al. Severity assessment of acute pancreatitis using CT severity index and modified CT severity index: Correlation with clinical outcomes and severity grading as per the Revised Atlanta Classification[J]. Indian J Radiol Imaging, 2017, 27(2): 152-160. DOI: 10.4103/ijri.IJRI_300_16. |
| [16] |
RIECHERS RG 2nd, RAMAGE A, BROWN W, et al. Physician knowledge of the Glasgow Coma Scale[J]. J Neurotrauma, 2005, 22(11): 1327-1334. DOI: 10.1089/neu.2005.22.1327. |
| [17] |
BLEDSOE BE, CASEY MJ, FELDMAN J, et al. Glasgow coma scale scoring is often inaccurate[J]. Prehosp Disaster Med, 2015, 30(1): 46-53. DOI: 10.1017/S1049023X14001289. |
| [18] |
HEIM C, SCHOETTKER P, GILLIARD N, et al. Knowledge of Glasgow coma scale by air-rescue physicians[J]. Scand J Trauma Resusc Emerg Med, 2009, 17: 39. DOI: 10.1186/1757-7241-17-39. |
| [19] |
YANG YX, LI L. Evaluating the ability of the bedside index for severity of acute pancreatitis score to predict severe acute pancreatitis: A meta-analysis[J]. Med Princ Pract, 2016, 25(2): 137-142. DOI: 10.1159/000441003. |
| [20] |
SENAPATI D, DEBATA PK, JENASAMANT SS, et al. A prospective study of the Bedside Index for Severity in Acute Pancreatitis (BISAP) score in acute pancreatitis: An Indian perspective[J]. Pancreatology, 2014, 14(5): 335-339. DOI: 10.1016/j.pan.2014.07.007. |
| [21] |
HAGJER S, KUMAR N. Evaluation of the BISAP scoring system in prognostication of acute pancreatitis - A prospective observational study[J]. Int J Surg, 2018, 54(Pt A): 76-81. DOI: 10.1016/j.ijsu.2018.04.026. |
| [22] |
HARSHIT KA, SINGH GM. A comparison of APACHE Ⅱ, BISAP, Ranson's score and modified CTSI in predicting the severity of acute pancreatitis based on the 2012 revised Atlanta Classification[J]. Gastroenterol Rep (Oxf), 2018, 6(2): 127-131. DOI: 10.1093/gastro/gox029. |
| [23] |
BANDAY IA, GATTOO I, KHAN AM, et al. Modified computed tomography severity index for evaluation of acute pancreatitis and its correlation with clinical outcome: A tertiary care hospital based observational study[J]. J Clin Diagn Res, 2015, 9(8): TC01-TC05. DOI: 10.7860/JCDR/2015/14824.6368. |
| [24] |
PETROV MS, SHANBHAG S, CHAKRABORTY M, et al. Organ failure and infection of pancreatic necrosis as determinants of mortality in patients with acute pancreatitis[J]. Gastroenterology, 2010, 139(3): 813-820. DOI: 10.1053/j.gastro.2010.06.010. |
| [25] |
MOFIDI R, SUTTIE SA, PATIL PV, et al. The value of procalcitonin at predicting the severity of acute pancreatitis and development of infected pancreatic necrosis: Systematic review[J]. Surgery, 2009, 146(1): 72-81. DOI: 10.1016/j.surg.2009.02.013. |










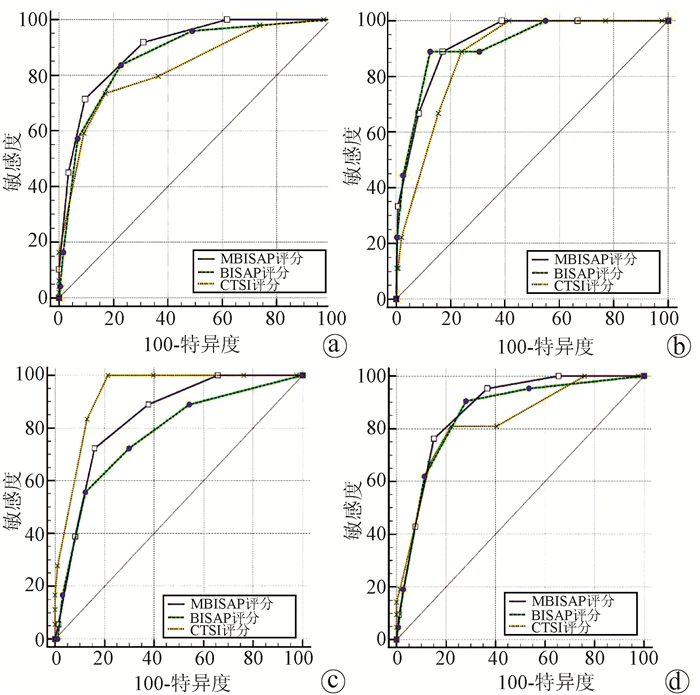




 DownLoad:
DownLoad: