| [1] |
YOUNOSSI ZM, KOENIG AB, ABDELATIF D, et al. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes[J]. Hepatology, 2016, 64(1): 73-84. DOI: 10.1002/hep.28431. |
| [2] |
CHALASANI N, YOUNOSSI Z, LAVINE JE, et al. The diagnosis and management of nonalcoholic fatty liver disease: Practice guidance from the American Association for the Study of Liver Diseases[J]. Hepatology, 2018, 67(1): 328-357. DOI: 10.1002/hep.29367. |
| [3] |
European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL-EASD-EASO clinical practice guidelines for the management of non-alcoholic fatty liver disease[J]. J Hepatol, 2016, 64(6): 1388-1402. DOI: 10.1016/j.jhep.2015.11.004. |
| [4] |
RINELLA ME, LOOMBA R, CALDWELL SH, et al. Controversies in the diagnosis and management of NAFLD and NASH[J]. Gastroenterol Hepatol (N Y), 2014, 10(4): 219-227.
|
| [5] |
SANYAL AJ, FRIEDMAN SL, McCULLOUGH AJ, et al. Challenges and opportunities in drug and biomarker development for nonalcoholic steatohepatitis: Findings and recommendations from an American Association for the Study of Liver Diseases-U.S. Food and Drug Administration Joint Workshop[J]. Hepatology, 2015, 61(4): 1392-1405. DOI: 10.1002/hep.27678. |
| [6] |
YOUNOSSI ZM, LOOMBA R, RINELLA ME, et al. Current and future therapeutic regimens for nonalcoholic fatty liver disease and nonalcoholic steatohepatitis[J]. Hepatology, 2018, 68(1): 361-371. DOI: 10.1002/hep.29724. |
| [7] |
KONERMAN MA, JONES JC, HARRISON SA. Pharmacotherapy for NASH: Current and emerging[J]. J Hepatol, 2018, 68(2): 362-375. DOI: 10.1016/j.jhep.2017.10.015. |
| [8] |
SIDDIQUI MS, IDOWU MO, PARMAR D, et al. A phase 2 double blinded, randomized controlled trial of saroglitazar in patients with nonalcoholic steatohepatitis[J]. Clin Gastroenterol Hepatol, 2020. DOI: 10.1016/j.cgh.2020.10.051.[Onlineaheadofprint] |
| [9] |
YOUNOSSI ZM, RATZIU V, LOOMBA R, et al. Obeticholic acid for the treatment of non-alcoholic steatohepatitis: Interim analysis from a multicentre, randomised, placebo-controlled phase 3 trial[J]. Lancet, 2019, 394(10215): 2184-2196. DOI: 10.1016/S0140-6736(19)33041-7. |
| [10] |
HARRISON SA, WONG VW, OKANOUE T, et al. Selonsertib for patients with bridging fibrosis or compensated cirrhosis due to NASH: Results from randomized phase Ⅲ STELLAR trials[J]. J Hepatol, 2020, 73(1): 26-39. DOI: 10.1016/j.jhep.2020.02.027. |
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
ANSTEE QM, NEUSCHWANDER-TETRI BA, WONG VW, et al. Cenicriviroc for the treatment of liver fibrosis in adults with nonalcoholic steatohepatitis: AURORA Phase 3 study design[J]. Contemp Clin Trials, 2020, 89: 105922. DOI: 10.1016/j.cct.2019.105922. |
| [15] |
|
| [16] |
HARRISON SA, BASHIR MR, GUY CD, et al. Resmetirom (MGL-3196) for the treatment of non-alcoholic steatohepatitis: A multicentre, randomised, double-blind, placebo-controlled, phase 2 trial[J]. Lancet, 2019, 394(10213): 2012-2024. DOI: 10.1016/S0140-6736(19)32517-6. |
| [17] |
|
| [18] |
CHIANELLI D, RUCKER PV, ROLAND J, et al. Nidufexor (LMB763), a novel FXR modulator for the treatment of nonalcoholic steatohepatitis[J]. J Med Chem, 2020, 63(8): 3868-3880. DOI: 10.1021/acs.jmedchem.9b01621. |
| [19] |
|
| [20] |
|
| [21] |
Guangdong Zhongsheng Pharmaceutical Co. Ltd. Tolerability, pharmacokinetics, and early pharmacodynamics of ZSP1601 tablets in NASH patients: A multicenter, randomized, double-blind, dose-escalation, and placebo-controlled phase Ib/Ⅱa clinical trial[EB/OL]. [2021-01-21]. http://www.chinadrugtrials.org.cn. |
| [22] |
YOUNES R, BUGIANESI E. NASH in lean individuals[J]. Semin Liver Dis, 2019, 39(1): 86-95. DOI: 10.1055/s-0038-1677517. |
| [23] |
SHI Y, WANG Q, SUN Y, et al. The prevalence of lean/nonobese nonalcoholic fatty liver disease: A systematic review and meta-analysis[J]. J Clin Gastroenterol, 2020, 54(4): 378-387. DOI: 10.1097/MCG.0000000000001270. |
| [24] |
HAGSTRÖM H, NASR P, EKSTEDT M, et al. Risk for development of severe liver disease in lean patients with nonalcoholic fatty liver disease: A long-term follow-up study[J]. Hepatol Commun, 2018, 2(1): 48-57. DOI: 10.1002/hep4.1124. |
| [25] |
WANG Q, YOU H, OU X, et al. Non-obese histologically confirmed NASH patients with abnormal liver biochemistry have more advanced fibrosis[J]. Hepatol Int, 2019, 13(6): 766-776. DOI: 10.1007/s12072-019-09982-z. |
| [26] |
TOBARI M, HASHIMOTO E, TANIAI M, et al. Characteristics of non-alcoholic steatohepatitis among lean patients in Japan: Not uncommon and not always benign[J]. J Gastroenterol Hepatol, 2019, 34(8): 1404-1410. DOI: 10.1111/jgh.14585. |
| [27] |
CURCIC IB, BERKOVIC MC, KUNA L, et al. Obesity paradox in chronic liver diseases: Product of bias or a real thing?[J]. J Clin Transl Hepatol, 2019, 7(3): 275-279. DOI: 10.14218/JCTH.2019.00029. |
| [28] |
LONARDO A, NASCIMBENI F, BALLESTRI S, et al. Sex differences in nonalcoholic fatty liver disease: State of the art and identification of research gaps[J]. Hepatology, 2019, 70(4): 1457-1469. DOI: 10.1002/hep.30626. |
| [29] |
SHEEDFAR F, DI BIASE S, KOONEN D, et al. Liver diseases and aging: Friends or foes?[J]. Aging Cell, 2013, 12(6): 950-954. DOI: 10.1111/acel.12128. |
| [30] |
YOUNOSSI ZM, GOLABI P, de AVILA L, et al. The global epidemiology of NAFLD and NASH in patients with type 2 diabetes: A systematic review and meta-analysis[J]. J Hepatol, 2019, 71(4): 793-801. DOI: 10.1016/j.jhep.2019.06.021. |
| [31] |
BAZICK J, DONITHAN M, NEUSCHWANDER-TETRI BA, et al. Clinical model for NASH and advanced fibrosis in adult patients with diabetes and NAFLD: Guidelines for referral in NAFLD[J]. Diabetes Care, 2015, 38(7): 1347-1355. DOI: 10.2337/dc14-1239. |
| [32] |
SUNG KC, WILD SH, BYRNE CD. Resolution of fatty liver and risk of incident diabetes[J]. J Clin Endocrinol Metab, 2013, 98(9): 3637-3643. DOI: 10.1210/jc.2013-1519. |
| [33] |
LONARDO A, NASCIMBENI F, MANTOVANI A, et al. Hypertension, diabetes, atherosclerosis and NASH: Cause or consequence?[J]. J Hepatol, 2018, 68(2): 335-352. DOI: 10.1016/j.jhep.2017.09.021. |
| [34] |
AMPUERO J, ALLER R, GALLEGO-DURáN R, et al. The effects of metabolic status on non-alcoholic fatty liver disease-related outcomes, beyond the presence of obesity[J]. Aliment Pharmacol Ther, 2018, 48(11-12): 1260-1270. DOI: 10.1111/apt.15015. |
| [35] |
VILAR-GOMEZ E, MARTINEZ-PEREZ Y, CALZADILLA-BERTOT L, et al. Weight loss through lifestyle modification significantly reduces features of nonalcoholic steatohepatitis[J]. Gastroenterology, 2015, 149(2): 367-378. DOI: 10.1053/j.gastro.2015.04.005. |
| [36] |
ROMERO-GÓMEZ M, ZELBER-SAGI S, TRENELL M. Treatment of NAFLD with diet, physical activity and exercise[J]. J Hepatol, 2017, 67(4): 829-846. DOI: 10.1016/j.jhep.2017.05.016. |
| [37] |
DAVISON BA, HARRISON SA, COTTER G, et al. Suboptimal reliability of liver biopsy evaluation has implications for randomized clinical trials[J]. J Hepatol, 2020, 73(6): 1322-1332. DOI: 10.1016/j.jhep.2020.06.025. |
| [38] |
HOOKER JC, HAMILTON G, PARK CC, et al. Inter-reader agreement of magnetic resonance imaging proton density fat fraction and its longitudinal change in a clinical trial of adults with nonalcoholic steatohepatitis[J]. Abdom Radiol (NY), 2019, 44(2): 482-492. DOI: 10.1007/s00261-018-1745-3. |
| [39] |
HONG CW, MARSH A, WOLFSON T, et al. Reader agreement and accuracy of ultrasound features for hepatic steatosis[J]. Abdom Radiol (NY), 2019, 44(1): 54-64. DOI: 10.1007/s00261-018-1683-0. |
| [40] |
|










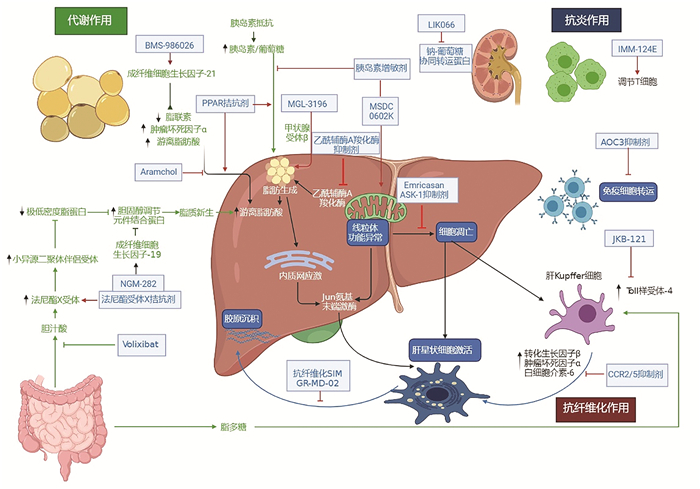




 DownLoad:
DownLoad: